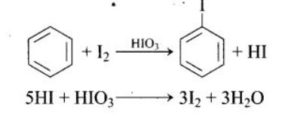
Arenes’ iodination can be reversed due to the production of HI. An oxidising agent, such as HNO3 or HIO4, oxidises HI to speed up the process and stabilise the result.

Arenes’ iodination can be reversed due to the production of HI. An oxidising agent, such as HNO3 or HIO4, oxidises HI to speed up the process and stabilise the result.