Chloride radical is detected by the chromyl chloride test. In this test, chromyl chloride gas (orange red color) is produced. Equation Involved – 4NaCl + K2Cr2O7 + 6H2SO4 → 4NaHSO4 + 2KHSO4 + 3H2O +...
What do you understand by lanthanide contraction
The lanthanide contraction is the decrease in the atomic or ionic radii with increase in the atomic number of lanthanides
What are lanthanide elements?
Lanthanide elements resembles a lot in properties with lanthanum. Lanthanide is group of 14 elements from atomic number 58 to 71. In these elements on increasing atomic number electron enters into...
Explain oxidization properties of potassium permanganate in acidic medium.
2KMnO4 + 8H2SO4 + 10KI → 6K2SO4 + 8H2O + 5I2 2KMnO4 + 5SO2 + 2H2O → 2MnSO4 + 2H2SO4 + K2SO4 2KmO4 + 16HCl → 2KCl + 2MnCl2 + 8H2O + 5Cl2 5COOH – COOH + [5O] → 10CO2 + 5H2O
Give two differences between double salt and complex salt.
Answer: Double salt Complex salt A double salt is a combination of two salt compounds. A complex salt is a molecular structure that is composed of one or more complex ions. Double salts can give...
Give two differences between DNA and RNA.
Answer: DNA RNA DNA – Deoxyribo Nucleic Acid RNA – Ribo Nucleic acid DNA consists of adenine (A), cytosine (C), guanine (G), and thymine (T) RNA consists of adenine (A), cytosine (C), guanine (G),...
Which of the following compounds has tetrahedral geometry? (a) ![Rendered by QuickLaTeX.com \left[\mathrm{Ni}(\mathrm{CN})_{4}\right]^{-2}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c5a91864ba7e1e0ed1b5d273d822f5e5_l3.png) (b)
(b) ![Rendered by QuickLaTeX.com \left[\mathrm{Pd}(\mathrm{CN})_{4}\right]^{2-}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-ffda616d3dbe7dc8925722e87005924e_l3.png) (c)
(c) ![Rendered by QuickLaTeX.com \left[\mathrm{PdCl}_{4}\right]^{2-}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-90cc6a4f4d42dee46e06d6f8ed516b7a_l3.png) (d)
(d) ![Rendered by QuickLaTeX.com \left[\mathrm{NiCl}_{4}\right]^{2}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-149ee5c95bd9cfcc4405704fba2e1bab_l3.png)
SOL: Correct option is D. $\left[\mathrm{NiCl}_{4}\right]^{2}$
Oxidation number of gold metal is (a)+1 (b) 0 (c) 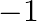 (d) all of these
(d) all of these
Sol: Correct option is B. 0
a) Consider the circuit in the figure. How much energy is absorbed by electrons from the initial state of no current to the state of drift velocity? b) Electrons give up energy at the rate of RI2 per second to the thermal energy. What time scale would one associate with energy in problem a) n = no of electron/volume = 1029/m3, length of circuit = 10 cm, cross-section = A = 1mm2
a) Current is given as I = V/R from the Ohm’s law Therefore, I = 1A But, I = ne Avd vd = I/neA When the values for the above parameters are substituted, vd = 1/1.6 × 10-4 m/s The KE = (KE of one...
In an experiment with a potentiometer, VB = 10V. R is adjusted to be 50 Ω. A student wanting to measure voltage E1 of a battery finds no null point possible. He then diminishes R to 10 Ω and is able to locate the null point on the last segment of the potentiometer. Find the resistance of the potentiometer wire and potential drop per unit length across the wire in the second case.
Equivalent resistance of the potentiometer = 50 Ohm + R’ Equivalent voltage across the potentiometer = 10 V Current through the main circuit I = 10/(50 Ohms +R’) Potential difference across wire of...
A room has AC run for 5 hours a day at a voltage of 220V. The wiring of the room consists of Cu of 1 mm radius and a length of 10 m. Power consumption per day is 10 commercial units. What fraction of it goes in the joule heating in wires? What would happen if the wiring is made of aluminium of the same dimensions?
Power consumption in a day = 10 units Power consumption per hour = 2 units Power consumption = 2 units = 2 kW = 2000 J/s Power consumption in resistors, P = VI Which gives I = 9A We know that...
Two cells of voltage 10V and 2V and internal resistances 10Ω and 5Ω respectively are connected in parallel with the positive end of the 10V battery connected to the negative pole of 2V battery. Find the effective voltage and effective resistance of the combination.
Kirchhoff’s law is applied at c, I1 = I + I2 Kirchhoff’s law is applied at efbae, 10 – IR – 10I2 = 0 10 = IR + 10I1 Kirchhoff’s law is applied at cbadc, -2-IR+5I2 = 0 2 = 5I2- RI I2 = I1 – I...
Suppose there is a circuit consisting of only resistances and batteries and we have to double all voltages and all resistances. Show that currents are unaltered.
Reff is the equivalent internal resistance of the battery Veff is the equivalent voltage of the battery Using Ohm’s law, I = Veff/(Reff + R) When the resistance and effective voltage are increased...
Two conductors are made of the same material and have the same length. Conductor A is a solid wire of diameter 1 mm. Conductor B is a hollow tube of outer diameter 2 mm and inner diameter 1 mm. Find the ratio of resistance RA to RB.
Resistance of wire R = ρ l/A Where A is the cross-sectional area of the conductor L is the length of the conductor ρ is the specific resistance RA = ρl/π(10-3 × 0.5)2 RB = ρl/ π[10-3)2 × (0.5 ×...
Two cells of same emf E but internal resistance r1 and r2 are connected in series to an external resistor R. What should be the value of R so that the potential difference across the terminals of the first cell becomes zero.
Effective emf of two cells = E + E = 2E Effective resistance = R + r1 + r2 Electric current is given as I = 2E/R+r1+r2 Potential difference is given as V1 – E – Ir1 = 0 Which f=gives R = r1 –...
The circuit in the figure shows two cells connected in opposition to each other. Cell E1 is of emf 6V and internal resistance 2Ω; the cell E2 is of emf 4V and internal resistance 8 Ω. Find the potential difference between the points A and B.
Applying Ohm’s law, equivalent emf of the two cells = 6 – 4 = 2V Equivalent resistance = 2 + 8 = 10 Ω Electric current, I = 6-4/2+8 = 0.2A When the loop is considered in the anti-clockwise...
Two cells of same emf E but internal resistance r1 and r2 are connected in series to an external resistor R. What should be the value of R so that the potential difference across the terminals of the first cell becomes zero.
Effective emf of two cells = E + E = 2E Effective resistance = R + r1 + r2 Electric current is given as I = 2E/R+r1+r2 Potential difference is given as V1 – E – Ir1 = 0 Which f=gives R = r1 –...
Let there be n resistors R1……..Rn with Rmax = max(R1……Rn) and Rmin = min(R1…….Rn). Show that when they are connected in parallel, the resultant resistance Rp < Rmin and when they are connected in series, the resultant resistance Rs > Rmax. Interpret the result physically.
The current is represented as I = E/R+nR when the resistors are connected in series. Current is expressed as 10I = E/(R+R/n) when the resistors are connected in parallel....
. A cell of emf E and internal resistance r is connected across an external resistance R. Plot a graph showing the variation of PD across R versus R.
The graphic depiction is as follows: The resistance r is connected across the external resistance R, and E is the cell's emf. V = ER/R+r is the connection between voltage and R....
While doing an experiment with potentiometer it was found that the deflection is one-sided and i) the deflection decreased while moving from one end A of the wire to the end B; ii) the deflection increased, while the jockey was moved towards the end B. i) Which terminal +ve or –ve of the cell E, is connected at X in case
i) and how is E1 related to E?
ii) Which terminal of the cell E1 is connected at X in case ii)?
The positive terminal of cell E1 is linked to E, and E is connected to X. Furthermore, E1 > E ii) cell E1's negative terminal is linked to X.
Power P is to be delivered to a device via transmission cables having resistance Rc. If V is the voltage across R and I the current through it, find the power wasted and how can it be reduced.
P = i2Rc is the power utilised by transmission lines. The resistance of connecting wires is denoted by Rc. P = VI is the formula for calculating power. Power...
Match the example given in Column I with the name of the reaction in Column II
Solution: (i) is e (ii) is d (iii) is a (iv) is b (v) is f (vi) is c
Match the reactions given in Column I with the suitable reagents given in Column II.
Solution: (i) is c (ii) is d (iii) is a (iv) is b
Match the acids given in Column I with their correct IUPAC names given in Column II.
Solution: (i) is b (ii) is e (iii) is d (iv) is a (v) is c
Match the common names given in Column I with the IUPAC names given in Column II
Solution: (i) is d (ii) is e (iii) is a (iv) is b (v) is c
Can Gatterman-Koch reaction be considered similar to Friedel Craft’s acylation? Discuss.
Solution: Both reactions resemble each other. In Friedel Craft’s acylation reaction, an aryl group or benzene is treated with an acid chloride in the presence of anhydrous AlCl3 and corresponding...
Ethylbenzene is generally prepared by acetylation of benzene followed by reduction and not by direct alkylation. Think of a possible reason.
Solution: This is due to the formation of polysubstituted products. To avoid the formation of polysubstituted products Friedel-craft’s alkylation reaction is not used for the preparation of...
Complete the following reaction sequence.
Solution:
Why are carboxylic acids more acidic than alcohols or phenols although all of them have a hydrogen atom attached to an oxygen atom (—O—H)?
Solution: Due to the resonance in carboxylic acids, the negative charge is at the more electronegative oxygen whereas, in alcohols or phenols, the negative charge is on a less electronegative atom....
. Identify the compounds A, B and C in the following reaction.
Solution: Compound A = CH3-MgBr Compound B = CH3-COOH Compound C = CH3COOCH3
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Solution: The oxygen atom in carbonyl compound pull more shared pair of electron towards itself and so, carbon acquires partial positive charge and oxygen acquires partial negative charge in...
Alkenes and carbonyl compounds both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Solution: Both the compounds carbon atom is attached to the electronegative atom oxygen. Thus the oxygen pulls more shared pair of electron towards them and a partial positive charge will be...
Arrange the following in decreasing order of their acidic strength. Explain the arrangement. C6H5COOH, FCH2COOH, NO2CH2COOH
Solution: NO2CH2COOH > FCH2COOH > C6H5COOH. NO2CH2COOH is most acidic among the given three compounds. Electron withdrawing groups like -NO2, increases the acidity of carboxylic acids by...
Compound ‘A’ was prepared by oxidation of compound ‘B’ with alkaline KMnO4. Compound ‘A’ on reduction with lithium aluminium hydride gets converted back to compound ‘B’. When compound ‘A’ is heated with compound B in the presence of H2SO4 it produces the fruity smell of compound C to which family the compounds ‘A’, ‘B’ and ‘C’ belong to?
Solution: Compound ‘A’ belongs to the carboxylic acid. Compound ‘B’ belongs to alcohol. Compound ‘C’ belongs to an ester group.
What product will be formed on reaction of propanal with 2-methyl propanal in the presence of NaOH? What products will be formed? Write the name of the reaction also.
Solution: When propanal reacts with 2-methyl propanal in the presence of NaOH, the mixture of aldehydes are formed. Both the reactants have an alpha-hydrogen and hence, can undergo cross aldol...
Arrange the following in decreasing order of their acidic strength and give the reason for your answer.
Solution: FCH2COOH > ClCH2COOH > C6H5CH2COOH > CH3COOH > CH3CH2OH. CH3CH2OH is least acidic among the given compounds. C6H5CH2COOH is more acidic than CH3COOH due to the resonance in...
Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Solution: Solution: The products formed on oxidation of 2, 5-dimethylhexan-3-one are the mixtures of ketone and carboxylic acids. Ketone is then further oxidized to carboxylic acids. Overall the...
Name the electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous AlCl3. Name the reaction also.
Solution: The electrophile produced in the reaction of benzene with benzoyl chloride in the presence of anhydrous AlCl3 is benzoylinium cation. The product formed in this reaction is benzophenone....
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzyl chloride and then benzaldehyde from it.
SOLUTION: Toluene is first converted to benzal chloride by side-chain chlorination, in presence of Chlorine gas and light. Benzal chloride on hydrolysis at 373K gives benzaldehyde.
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzyl chloride and then benzaldehyde from it.
Solution:
Write IUPAC names of the following structures.
Solution: (i) Ethane-1,2-dial. (ii) Benzene-1, 4-dicarbaldehyde. (iii) 3-Bromobenzaldehyde.
Give the structure of the following compounds. (i) 4-Nitropropiophenone (ii) 2-Hydroxycyclopentanecarbaldehyde (iii) Phenyl acetaldehyde
Give the IUPAC names of the following compounds
Solution: (i) 3-Phenylprop-2-ene-1-al. (ii) Cyclohexanecarbaldehyde (iii) 3-Oxopentan-1-al (iv) IUPAC name: But-2-enal
Power P is to be delivered to a device via transmission cables having resistance Rc. If V is the voltage across R and I the current through it, find the power wasted and how can it be reduced.
Power consumed by the transmission lines is given as P = i2Rc. Where Rc is the resistance of connecting cables. Power is given as P = VI. The transmission of power takes places either during low...
Arrange the bonds in order of increasing ionic character in the molecules: LiF, 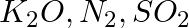 , and
, and 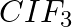 .
.
Solution: The difference in electronegativity between constituent atoms determines the ionic character of a molecule. As a result, the greater the difference, the greater the ionic character of a...
Why are alloys used for making standard resistance coils?
Alloys are used in the making of the standard resistance coils because they have less temperature coefficient of resistance and the temperature sensitivity is also less.
Explain with the help of suitable example polar covalent bond.
Solution: The bond pair of electrons are not shared equally when two unique atoms with different electronegativities join to form a covalent bond. The bond pair is attracted to the nucleus of an...
Define electronegativity. How does it differ from electron gain enthalpy?
Solution: "Electronegativity refers to an atom's ability to attract a bond pair of electrons towards itself in a chemical compound." Sr. No Electronegativity Electron affinity 1 A tendency to...
For wiring in the home, one uses Cu wires or Al wires. What considerations are involved in this?
The main considerations in the selection of the wires is the conductivity of the metal, cost of metal, and their availability.
Write the significance/applications of dipole moment.
Solution: There is a difference in electro-negativities of constituents of the atom in a heteronuclear molecule, which causes polarisation. As a result, one end gains a positive charge, while the...
Although both 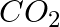 and
and 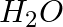 are triatomic molecules, the shape of the
are triatomic molecules, the shape of the 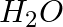 molecule is bent while that of
molecule is bent while that of 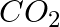 is linear. Explain this on the basis of dipole moment.
is linear. Explain this on the basis of dipole moment.
Solution: $CO_2$ has a dipole moment of 0 according to experimental results. And it's only possible if the molecule's shape is linear, because the dipole moments of the C-O bond are equal and...
What are the advantages of the null-point method in a Wheatstone bridge? What additional measurements would be required to calculate R unknown by any other?
The advantage of a null-point in the Wheatstone bridge is that the resistance of the galvanometer is not affected by the balance point. The R unknown is calculated by using Kirchhoff’s rule.
The relaxation time τ is nearly independent of applied E field whereas it changes significantly with temperature T. First fact is responsible for Ohm’s law whereas the second fact leads to a variation of ρ with temperature. Elaborate why?
Relaxation time is the time interval between two successive collisions of the electrons.It is defined asτ = mean free path/rms velocity of electrons usually, the drift velocity of the electrons is...
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions :(iii) Al and N.
Solution: Below is a list of Lewis symbols. To form a cation, a metal atom loses one or more electrons, while a nonmetal atom gains one or more electrons. Ionic bonds are formed between cations and...
Is the momentum conserved when charge crosses a junction in an electric circuit? Why or why not?
The momentum is not conserved when the charge crosses a junction in an electric circuit. This is because the drift velocity is proportional to the electric field.
Use Lewis symbols to show electron transfer between the following atoms to form cations and anions : (i) K and S (ii) Ca and O
Solution: Below is a list of Lewis symbols. To form a cation, a metal atom loses one or more electrons, while a nonmetal atom gains one or more electrons. Ionic bonds are formed between cations and...
Temperature dependence of resistivity ρ(T) of semiconductors, insulators, and metals is significantly based on the following factors:
a) number of charge carriers can change with temperature T
b) time interval between two successive collisions can depend on T
c) length of material can be a function of T
d) mass of carriers is a function of T
The correct answer is a) number of charge carriers can change with temperature T b) time interval between two successive collisions can depend on T
In a meter bridge, the point D is a neutral point.
a) the meter bridge can have no other neutral point for this set of resistances
b) when the jockey contacts a point on meter wire left of D, current flows to B from the wire
c) when the jockey contacts a point on a meter wire to the right of D, current flows from B to the wire through the galvanometer
d) when R is increased, the neutral point shifts to left
The correct answer is a) the meter bridge can have no other neutral point for this set of resistances c) when the jockey contacts a point on a meter wire to the right of D, current flows from B to...
Write the resonance structures for  , and
, and 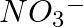
Solution: Resonance is the phenomenon that allows a molecule to be expressed in multiple ways, none of which fully explain the molecule's properties. The molecule's structure is called a resonance...
Explain the important aspects of resonance with reference to the 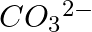 ion.
ion.
Solution: However, while the carbonate ion cannot be represented by a single structure, the properties of the ion can be described by two or more different resonance structures. The actual structure...
Define Bond length.
Solution: Bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule.
How do you express the bond strength in terms of bond order?
Solution: During the formation of a molecule, the extent of bonding that occurs between two atoms is represented by the bond strength of the molecule. As the bond strength increases, the bond...
Discuss the shape of the following molecules using the VSEPR model: 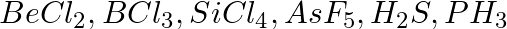
Solution: $BeCl_2$ The central atom does not have a lone pair, but it does have two bond pairs. As a result, its shape is AB2, or linear. $BCl_3$ The central atom has three bond pairs but no lone...
Define the octet rule. Write its significance and limitations
Solution: “Atoms can combine either by transferring valence electrons from one atom to another or by sharing their valence electrons in order to achieve the closest inert gas configuration by having...
Write Lewis symbols for the following atoms and ions: Sand 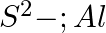 and
and 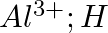 and
and 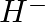
Solution: For S and S2- A sulphur atom has only 6 valence electrons, which is a very small number. As a result, the Lewis dot symbol for the letter S is The presence of a...
The measurement of an unknown resistance R is to be carried out using Wheatstone bridge. Two students perform an experiment in two ways. The first student take R2 = 10Ω and R1 = 5Ω. The other student takes R2 = 1000 Ω and R1 = 500 Ω. In the standard arm, both take R3 = 5 Ω. Both find R = R2/R1 R3 = 10 Ω within errors.
a) the errors of measurement of the two students are the same
b) errors of measurement do depend on the accuracy with which R2 and R1 can be measured
c) if the student uses large values of R2 and R1, the currents through the arms will be feeble. This will make determination of null point accurately more difficult
d) Wheatstone bridge is a very accurate instrument and has no errors of measurement
The correct answer is b) errors of measurement do depend on the accuracy with which R2 and R1 can be measured c) if the student uses large values of R2 and R1, the currents through the arms will be...
Write Lewis dot symbols for atoms of the following elements :c) B d) O
Solution: Boron atoms have only three valence electrons, which is a very small number. As a result, the Lewis dot symbols for B are as follows: The oxygen atom has only six valence...
Temperature dependence of resistivity ρ(T) of semiconductors, insulators, and metals is significantly based on the following factors:
a) number of charge carriers can change with temperature T
b) time interval between two successive collisions can depend on T
c) length of material can be a function of T
d) mass of carriers is a function of T
solution:The correct answer is a) number of charge carriers can change with temperature T b) time interval between two successive collisions can depend on T
Consider a simple circuit in the figure.stands for a variable resistance R’.
R’ can vary from R0 to infinity. r is internal resistance of the battery,
a) potential drop across AB is nearly constant as R’ is varied
b) current through R’ is nearly a constant as R’ is varied
c) current I depends sensitively on R’
d) I ≥V/r+R always
solution: The correct answer is a) potential drop across AB is nearly constant as R’ is varied d) I ≥V/r+R always
Kirchhoff’s junction rule is a reflection of
a) conservation of current density vector
b) conservation of charge
c) the fact that the momentum with which a charged particle approaches a junction is unchanged as the charged particle leaves the junction
d) the fact that there is no accumulation of charges at a junction
solution: The correct answer is b) conservation of charge d) the fact that there is no accumulation of charges at a junction
Which of the following characteristics of electrons determines the current in a conductor?
v a) drift velocity alone
b) thermal velocity alone
c) both drift velocity and thermal velocity
d) neither drift nor thermal velocity
solution: The correct answer is a) drift velocity alone
A metal rod of length 10 cm and a rectangular cross-section of 1 cm × 1/2 cm is connected to battery across opposite faces. The resistance will be
a) maximum when the battery is connected across 1 cm × 1/2 cm faces
b) maximum when the battery is connected across 10 cm × 1 cm faces
c) maximum when the battery is connected across 10 cm × 1/2 cm faces
d) same irrespective of the three faces
solution:The correct solution is a) maximum when the battery is connected across 1 cm × 1/2 cm faces
Two cells of emf’s approximately 5V and 10V are to be accurately compared using a potentiometer of length 400 cm.
a) the battery that runs the potentiometer should have voltage of 8V
b) the battery of potentiometer can have a voltage of 15V and R adjusted so that the potential drop across the wire slightly exceeds 10V
c) the first portion of 50 cm of wire itself should have a potential drop of 10V
d) potentiometer is usually used for comparing resistances and not voltages
Solution: The correct solution is b) the potentiometer's battery can be set to 15V and R adjusted so that the potential drop across the wire is a little higher than 10V.
A resistance R is to be measured using a meter bridge. Student chooses the standard resistance S to be 100Ω. He finds the null point at l1 = 2.9 cm . He is told to attempt to improve the accuracy. Which of the following is a useful way?
a) he should measure l1 more accurately
b) he should change S to 1000 Ω and repeat the experiment
c) he should change S to 3 Ω and repeat the experiment
d) he should give up hope of a more accurate measurement with a meter bridge
solution:The correct answer is c) he should change S to 3 Ω and repeat the experiment
Two batteries of emf ε1 and ε2 and internal resistances r1 and r2 respectively are connected in parallel as shown in the figure.a) the equivalent emf εeq of the two cells is between ε1 and ε2 that is ε1 < εeq < ε2
b) the equivalent emf εeq is smaller than ε1
c) the εeq is given by εeq = ε1 + ε2 always
d) εeq is independent of internal resistances r1 and r2
solution: The correct answer is a) the equivalent emf εeq of the two cells is between ε1 and ε2 that is ε1 < εeq < ε2
Consider a current-carrying wire in the shape of a circle. Note that as the current progresses along the wire, the direction of j changes in an exact manner, while the current I remain unaffected. The agent that is essentially responsible for is
a) source of emf
b) electric field produced by charges accumulated on the surface of wire
c) the charges just behind a given segment of wire which push them just the right way by repulsion
d) the charges ahead
solution: The correct answer is b) electric field produced by charges accumulated on the surface of wire
Write a test to differentiate between pentan-2-one and pentan-3-one.
Solution: One can differentiate between pentan-2-one and pentan-3-one by iodoform test. Pentan-2-one have a –CO-CH3 group and therefore forms a yellow precipitate of Iodoform. Pentan-2-one gives a...
Why is there a large difference in the boiling points of butanal and butane-1-ol?
Solution: Butanal has no intermolecular hydrogen bonding but butan-1-ol has intermolecular hydrogen bonding. This bonding in butan-1-ol makes it more stable at a higher temperature than butanal.
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A) :
Solution: Option (A) and (B) are the answers. Reason:
Benzophenone can be obtained by ____________. (i) Benzoyl chloride + Benzene + AlCl3 (ii) Benzoyl chloride + Diphenyl cadmium (iii) Benzoyl chloride + Phenyl magnesium chloride (iv) Benzene + Carbon monoxide + ZnCl2
Solution: Option (i) and (ii) are the answers Reason: Benzophenone can be obtained by the Friedel-Craft acylation reaction. The reaction is shown as
Through which of the number of the following reactions of carbon atoms can be increased in the chain? (i) Grignard reaction (ii) Cannizaro’s reaction (iii) Aldol condensation (iv) HVZ reaction
Solution: Option (i) and (iii) are the answers. Reason: Grigned reaction and aldol condensation is used to increase the number of carbon attom in the chain as follows:
Write Lewis dot symbols for atoms of the following elements :
a) Mg
b) Na
Solution: Only two valence electrons exist in the magnesium atom. As a result, the Lewis dot symbols for Mg are as follows: Only one valence electron exists in the sodium atom. As a...
Which of the following conversions can be carried out by Clemmensen Reduction? (i) Benzaldehyde into benzyl alcohol (ii) Cyclohexanone into cyclohexane (iii) Benzoyl chloride into benzaldehyde (iv) Benzophenone into diphenylmethane
Solution: Option (ii) and (iv) are the answers. Reason: The carbonyl group of aldehydes and ketones is reduced to CH2 group on treatment with zinc amalgam and concentrated hydrochloric acid...
Explain the formation of a chemical bond.
Answer: "A chemical bond is an attractive force that holds a chemical species' constituents together." For chemical bond formation, many theories have been proposed, including valence shell electron...
Treatment of compound with NaOH solution yields(i) Phenol (ii) Sodium phenoxide (iii) Sodium benzoate (iv) Benzophenone
Solution: Option (ii) and (iii) are the answers. Reason: Treatment of compound with NaOH yields sodium phenoxide and sodium by means of nucleophilic substitution reaction as follows
13. Which of the following compounds do not undergo aldol condensation?
Solution: Option (ii) and (iv) are the answers. reason: Aldehydes and ketones and having at least one alpha-hydrogen undergo a reaction in the presence of dilute alkali as catalyst to beta-hydroxy...
In Clemmensen Reduction carbonyl compound is treated with _____________. (i) Zinc amalgam + HCl (ii) Sodium amalgam + HCl (iii) Zinc amalgam + nitric acid (iv) Sodium amalgam + HNO3
Solution: Option (i) is the answer. Reason: From the above reaction carbonyl group is treated with Zn−Hg(Zinc Amalgum) and HCl
Which of the following compounds will give butanone on oxidation with alkaline KMnO4 solution? (i) Butan-1-ol (ii) Butan-2-ol (iii) Both of these (iv) None of these
Solution: Option (ii) is the answer.
Which is the most suitable reagent for the following conversion?(i) Tollen’s reagent (ii) Benzoyl peroxide (iii) I2 and NaOH solution (iv) Sn and NaOH solution
Solution: Option (iii) is the answer. Reason: This reaction is called as lodoform reaction.
Compound A and C in the following reaction are :_____________
Solution: Option (ii) is the answer. Reason:
Structure of ‘A’ and type of isomerism in the above reaction are respectively. (i) Prop–1–en–2–ol, metamerism (ii) Prop-1-en-1-ol, tautomerism (iii) Prop-2-en-2-ol, geometrical isomerism (iv) Prop-1-en-2-ol, tautomerism
Solution: Option (iv) is the answer. reason: Structure of A and the type of isomerism in the above reaction are Prop-1-en-2-ol, tautomerism respectively. Enol form tautomerises into keto...
Which product is formed when the compoundis treated with concentrated aqueous KOH solution?
Solution: Option (ii) is the answer. Reason: Benzaldhyde C6H5CHO on treatment with KOH yields the corresponding alcohol and acid. In this reaction, there is no alpha hydrogen atom present in...
Cannizaro’s reaction is not given by _____________.
Solution: Option (iv) is the answer. Reason: CH3CHO will not give Cannizzaro’s reaction because it contains a-hydrogen while other three compounds have no a-hydrogen. Hence, they will give...
The reagent which does not react with both, acetone and benzaldehyde. (i) Sodium hydrogen sulphite (ii) Phenyl hydrazine (iii) Fehling’s solution (iv) Grignard reagent
Solution: Option (iii) is the answer. Reason: Aromatic aldehydes and ketones does not respond to Fehling's test. Sodium hydrogen sulphite,phenyl hydrazine, grignard reaction are common for carbonyl...
Compound can be prepared by the reaction of _____________.
Solution: Option (ii) is the answer. Reason:
The correct order of increasing acidic strength is _____________. (i) Phenol < Ethanol < Chloroacetic acid < Acetic acid (ii) Ethanol < Phenol < Chloroacetic acid < Acetic acid (iii) Ethanol < Phenol < Acetic acid < Chloroacetic acid (iv) Chloroacetic acid < Acetic acid < Phenol < Ethanol
Solution: Option (iii) is the answer. Reason: The correct order of increasing acidic strength is Ethanol < Phenol < Acetic acid < Chloroacetic acid. Phenol is more acidic than ethanol...
Which of the following compounds is most reactive towards nucleophilic addition reactions?
Solution: Option (i) is the answer.
Addition of water to alkynes occurs in acidic medium and the presence of Hg2+ ions as a catalyst. Which of the following products will be formed on addition of water to but-1-one under these conditions.
Solution: Option (ii) is the answer. Reason: Addition of water to but-1-yne in the presence of H2SO4 and HgSO4 gives 2-butaone. The addition takes place by markovnikoff's rule....
Arrange the following compounds in increasing order of dipole moment. CH3CH2CH3, CH3CH2NH2, CH3CH2OH
Solution: CH3CH2CH3 < CH3CH2NH2 < CH3CH2OH The dipole moment of CH3CH2OH is greater than that of CH3CH2NH2. CH3CH2CH3 has the least dipole moment among the three given compounds because it is...
Predict the product of the reaction of aniline with bromine in a non-polar solvent such as CS2.
Solution: The products formed in the reaction of aniline with bromine in a non-polar solvent such as CS2 are 4-Bromoaniline and 2-Bromoaniline where 4-Bromoaniline is the major product.
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Solution: This reaction is carried out in a mild basic medium. This is an electrophilic substitution reaction. Aryldiazonium chloride reacts with aniline to form a yellow dye of p-Aminoazobenzene.
Explain why MeNH2 is a stronger base than MeOH?
Solution: MeNH2 is a stronger base than MeOH because of the lower electronegativity and the presence of the lone pair of electrons on the nitrogen atom in MeNH2.
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Solution: At high temperatures, benzene diazonium chloride is highly soluble in water, and at low temperatures, it is a very stable compound in water. Because it is unstable, it should be used as...
What is the best reagent to convert nitrile to primary amine?
Solution: LiAlH4 and Sodium/Alcohol are the best reagents for converting nitrile to primary amine. The nitriles can be converted into a corresponding primary amine through reduction.
Why is NH2 group of aniline acetylated before carrying out nitration?
Solution: The NH2 group of aniline is acetylated before nitration to control the nitration reaction and the formation of tarry oxidation products and nitro derivatives. P-nitroaniline is the main...
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
Solution: Option (i) and (ii) are the answers. Reason:...
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc. H2SO4 + conc. HNO3
(ii) Acetic anhydride/pyridine followed by conc. H2SO4 + conc. HNO3
(iii) Dil. HCl followed by reaction with conc. H2SO4 + conc. HNO3
(iv) Reaction with conc. HNO3 + conc.H2SO4
Solution: Option (i) and (ii) are the answers. Reason: In addition to the nitro derivatives, direct nitration of aniline produces tarry oxidation products. Furthermore, in a strongly acidic...
Which of the following reactions are correct?
Solution: Option (i) and (iii) are the answers. Reason:
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methyl benzylamine
(iv) Aniline
Solution: Option (i) and (ii) are the answers. Reason: Gabriel synthesis is used for the preparation of primary amines. Phthalimide on treatment with ethanolic potassium hydroxide forms potassium...
Arenium ion involved in the bromination of aniline is __________.
Solution: Option (i), (ii) and (iii) are the answers. Reason: Arenium ion involved in the bromination of aniline are as follows:
The product of the following reaction is __________.
Solution: Option (A) and (B) is the answer. Reason:
The reagents that can be used to convert benzene diazonium chloride to benzene are __________.
(i) 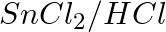
(ii) 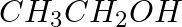
(iii) 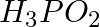
(iv) 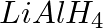
Solution: Option (ii) and (iii) are the answers. Reason: Hypophosphorous acid (phosphinic acid) and ethanol, for example, reduce diazonium salts to arenes, which are then oxidised to phosphorous...
Which of the following species are involved in the carbylamine test?
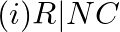
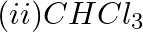
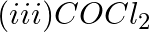
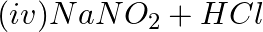
Solution: Option (i) and (ii) are the answers. Reason: Only RNC and CHCl3 are involved in carbylamine reaction.
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) Sn/HCl
(ii) Fe/HCl
(iii) H2-Pd
(iv) Sn/NH4OH
Solution: Option (i), (ii), and (iii) are the answers. Reason: They are reducing agents.
Which of the following cannot be prepared by Sandmeyer’s reaction?
(i) Chlorobenzene
(ii) Bromobenzene
(iii) Iodobenzene
(iv) Fluorobenzene
Solution: Option (iii) and (iv) are the answers. Reason: Sandmeyer's reaction is used for the preparation of chlorobenzene and bromobenzene.
Which of the following methods of preparation of amines will not give the same number of carbon atoms in the chain of amines as in the reactant?
(i) The reaction of nitrite with LiAlH4.
(ii) The reaction of the amide with LiAlH4 followed by treatment with water.
(iii) Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
(iv) Treatment of amide with bromine in the aqueous solution of sodium hydroxide.
Solution: Option (iv) is the answer. Reason: In Hoffmann Bromide degradation as the word, suggest, the amide is reduced to an amine with 1- carbonless, so this is the method in which we don't get...
Which of the following should be most volatile?
Solution: Option (ii) is the answer. Reason: The order of boiling points of isomeric amines is 1 amine > 2 amines > 3 amines. 3 amines have no intermolecular association because there are no H...
Among the following amines, the strongest Brönsted base is __________.
Solution; Option (iv) is the answer. Reason: Option (iv) is the strongest Bronsted base as there is no delocalization of lone pair of electron of the atom which is not possible in aniline and in...
The correct decreasing order of basic strength of the following species is _______. H2O, NH3, OH–, NH2– (i) NH2– > OH – > NH3 > H2O (ii) OH– > NH2– > H2O > NH3 (iii) NH3 > H2O > NH2– > OH– (iv) H2O > NH3> OH– > NH2–
Solution: Option (i) is the answer. Reason: NH3 is more basic than H2O, therefore NH2− (Conjugate base of weak acid NH3) is a stronger base than OH−.
Among the following amines, the strongest Brönsted base is __________.
Solution; Option (iv) is the answer. Reason: Option(iv)is the strongest Bronsted base as there is no delocalisation of lone pair of electron of N atom which is not possible in aniline and in...
Which of the following compounds is the weakest Brönsted base?
Solution: Option (iii) is the answer. Reason: A Bronsted Lowry base is a proton acceptor or hydrogen ion acceptor. Amines have a stronger tendency to accept protons and are strong Bronsted bases....
Which of the following compound will not undergo an azo coupling reaction with benzene diazonium chloride.
(i) Aniline
(ii) Phenol
(iii) Anisole
(iv) Nitrobenzene
Solution: Option (iv) is the answer. Reason: Diazonium cation is a weak electrophile and hence it reacts with electron-rich compounds containing electron-donating groups such as −OH, -$NH_2$ and...
The best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is
(i) Hoffmann Bromamide reaction
(ii) Gabriel phthalimide synthesis
(iii) Sandmeyer reaction
(iv) Reaction with 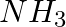
Solution: Option (ii) is the answer. Reason: Best method for preparing primary aminos form alkyl halides without changing the number of carbon atoms in the chain is Gabriel synthesis. Because this...
The reaction Ar + N2Cl– → (Cu/HCl)– ArCl + N2 + CuCl is named as _________.
(i) Sandmeyer reaction
(ii) Gatterman reaction
(iii) Claisen reaction
(iv) Carbylamine reaction
Solution: Option (ii) is the answer. Reason: Diazonium salts in the presence of copper powder and halogen acid give aryl halide. Gattermann reaction is a variation of Sandmeyer reaction in which...
Acid anhydrides on reaction with primary amines give ____________.
(i) amide
(ii) imide
(iii) secondary amine
(iv) imine
Solution: Option (i) is the answer Reason: When acid anhydrides react with primary amines, they produce amide. The H atom of the amino group is replaced with an acyl group in this nucleophilic...
The most reactive amine towards dilute hydrochloric acid is ___________.
Solution: Option (ii) is the answer. Reason: The reactivity of amines is proportional to their basicity. If the R group is, the order of basicity is secondary amine ...
Reduction of aromatic nitro compounds using Fe and HCl gives __________.
(i) aromatic oxime
(ii) aromatic hydrocarbon
(iii) aromatic primary amine
(iv) aromatic amide
Solution: Option (iii) is the answer. Reason: Reduction of nitro aryl compounds in presence of Fe and HCl gives aromatic primary amines.
In the nitration of benzene using a mixture of conc. 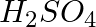 and conc.
and conc. 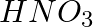 ,the species which initiates the reaction is __________.
,the species which initiates the reaction is __________.
(i) 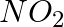
(ii) 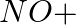
(iii) 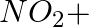
(iv) 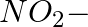
Solution: Option (iii) is the answer. Reason:
The gas evolved when methylamine reacts with nitrous acid is __________.
(i) 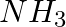 (ii)
(ii) 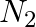 (iii)
(iii) 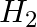 (iv)
(iv) 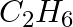
Solution: Option (ii) is the answer. Reason:
Methylamine reacts with HNO2 to form _________.
(i) 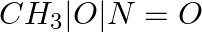 (ii)
(ii) 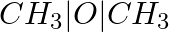 (iii)
(iii) 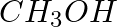 (iv)
(iv) 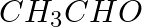
Solution: Option (iii) is the answer. Reason:
The correct increasing order of basic strength for the following compounds is _________.
(i) II < III < I
(ii) III < I < II
(iii) III < II < I
(iv) II < I < III
Solution: Option (iv) is the answer. Reason: Electron donating: group increases the basicity while electron-withdrawing group decreases the basicity of...
Hoffmann Bromamide Degradation reaction is shown by __________.
(i) 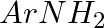
(ii) 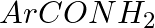
(iii) 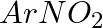
(iv) 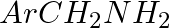
Solution: Option (ii) is the answer. Reason: Where the aryl amide is converted to arylamine in the presence of $Br_2$ and $NaOH$ .
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanolamine is _____.
(i) excess H2 (ii) Br2 in aqueous NaOH (iii) iodine in the presence of red phosphorus (iv) LiAlH4 in ether
Solution: Option (iv) is the answer. Reason:
Amongst the given set of reactants, the most appropriate for preparing 2° amine is _____.
(i) 2° R—Br + NH3
(ii) 2° R—Br + NaCN followed by 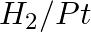
(iii) 1° R—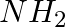 + RCHO followed by %H_2/Pt
+ RCHO followed by %H_2/Pt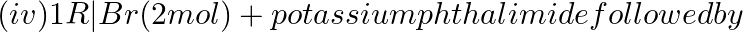 H_3O+$/heat
H_3O+$/heat
Solution: Option (iii) is the answer. Reason:
The source of nitrogen in Gabriel synthesis of amines is _____________.
(i) Sodium azide, NaN3
(ii) Sodium nitrite, NaNO2
(iii) Potassium cyanide, KCN
(iv) Potassium phthalimide
Solution: Option (iv) is the answer. Reason: Gabriel synthesis :The reaction is given to the image.Source of nitrogen atom is Gabriel synthesis is Potassium phthalamide.
To prepare a 1° amine from an alkyl halide with simultaneous addition of one 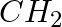 group in the carbon chain, the reagent used as a source of nitrogen is ___________.
group in the carbon chain, the reagent used as a source of nitrogen is ___________.
(i) Sodium amide, NaNH2
(ii) Sodium azide, NaN3
(iii) Potassium cyanide, KCN
(iv) Potassium phthalimide
Solution: Option (iii) is the answer. Reason:
6. Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine? (i) H2 (excess)/Pt (ii) LiAlH4 in ether (iii) Fe and HCl (iv) Sn and HCl
Solution: Option (ii) is the answer. Reason: LiAlH4/ether reduces aryl nitro compounds to azo compounds 2C6H5NO2→LiAIH4C6H5N=N-C6H5
5. Benzylamine may be alkylated as shown in the following equation : C6H5CH2NH2 + R—X → C6H5CH2NHR Which of the following alkyl halides is best suited for this reaction through SN1 mechanism? (i) CH3Br (ii) C6H5Br (iii) C6H5CH2Br (iv) C2H5 Br
Solution: Option (iii) is the answer. Reason: C6H5CH2Br is best suited for this reaction through SN1 mechanism as the carbocation (C6H5CH2) formed is resonance...
Which of the following is the weakest Brönsted base?
Solution: Option (A) is the answer. Reason: Aniline is the weakest Bronsted base due to delocalization of lone pair of electron...
Amongst the following, the strongest base in aqueous medium is ____________.
(i) 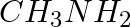
(ii) 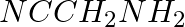
(iii) 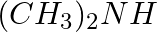
(iv) 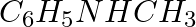
Solution: Option (iii) is the answer. Reason: Due to the electron releasing nature of the alkyl group, it (R) pushes electrons towards nitrogen and thus makes the uncharged electrons pair available...
The correct IUPAC name for 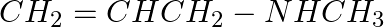 is (i) Allylmethylamine (ii) 2-amino-4-pentene (iii) 4-aminopent-1-ene (iv) N-methylprop-2-en-1-amine
is (i) Allylmethylamine (ii) 2-amino-4-pentene (iii) 4-aminopent-1-ene (iv) N-methylprop-2-en-1-amine
Solution: Option (iv) is the answer. Reason: $CH_2=CHCH_2-NHCH_3$ N−methylprop−2−en−1−amine.
Which of the following is a 3° amine?(i) 1-methylcyclohexylamine (ii) Triethylamine (iii) tert-butylamine (iv) N-methyl aniline
Solution: Option (ii) is the answer. Reason: Triethylamine is a 3° amine because of amonia in which each hydrogen atom is substituted by an methyl group.
A fix number of spherical drops of a liquid of radius ‘  ‘ coalesce to form a large drop of radius ‘
‘ coalesce to form a large drop of radius ‘  and volume ‘
and volume ‘ 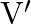 . If ‘
. If ‘ 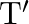 is the surface tension then energy
is the surface tension then energy
A) is niether released nor absorbed
B)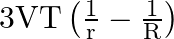 is released.
is released.
C) 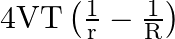 is released.
is released.
D) 3 VT 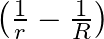 is absorbed.
is absorbed.
Correct option is B. $\begin{array}{l} \Delta \mathrm{U}=(\mathrm{T})(\Delta \mathrm{A}) \\ \mathrm{A}(\text { initial })=\left(4 \pi \mathrm{r}^{2}\right) \mathrm{n} \\ \mathrm{A}(\text { final...
A note produces 4 beat/s with a tuning fork of frequency 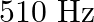 and 6 beat/s with a fork of frequency
and 6 beat/s with a fork of frequency 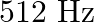 . The frequency of the note is
. The frequency of the note is
A)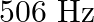
B)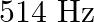
C)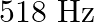
D)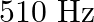
Correct option is A. It produces 4 beats/s with a fork of frequency $510 \mathrm{~Hz}$. Hence its frequency can be 514 or $506 \mathrm{~Hz}$. It also produces 6 beats/s with a fork of frequency $512...
When a beam of unpolarised monochromatic light is incident on a plane glass plate at a polarising angle, then which one of the following statements is correct?
A) Reflected and refracted rays are completely polarised with their planes of polarisation perpendicular to each other.
B) Reflected light is partially polarised but refracted light is plane polarised.
C) Reflected and refracted rays are completely polarised with their planes of polarisation parallel to each other.
D) Reflected light is plane polarised light but transmitted light is partially polarised.
Correct option is D.
Five capacitors each of capacitance ‘ 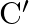 are connected as shown in the figure. The ratio of equivalent capacitance between
are connected as shown in the figure. The ratio of equivalent capacitance between 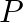 and
and 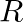 and the equivalent capacitance between
and the equivalent capacitance between 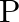 and
and 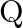 is
is
A)$2: 3$ B)$1: 1$ C)$3: 1$ D)$5: 2$ Correct option is A. For the both cases the equivalent circuits are shown in figure. For (1) the equivalent capacitance between $\mathrm{P}$ and $\mathrm{R}$ is...
What is the stopping potential, when a metal surface with work function 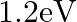 is illuminated with light of energy
is illuminated with light of energy 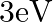 ?
?
A) 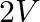
B)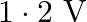
C)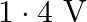
D)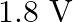
Correct option is D. $\begin{array}{l} e V s=E-W_{0} \\ \therefore V_{s}=\frac{E}{e}-\frac{W_{0}}{e}=\frac{3 e}{e}-\frac{1.2 e V}{e}=1.8 V \end{array}$
A smooth sphere of mass ‘M’ moving with velocity ‘u’ directly collides elastically with another sphere of mass ‘ 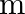 ‘ at rest. After collision, their final velocities are V’ and
‘ at rest. After collision, their final velocities are V’ and 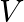 respectively. The value of
respectively. The value of 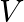 is given by
is given by
A)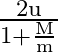
B) 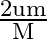
C)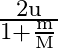
D)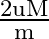
Correct option is C. Using Newton's law of collision, $\begin{array}{l} \frac{\mathrm{V}-\mathrm{v}}{\mathrm{u}-0}=-\mathrm{e} \\ \frac{\mathrm{V}-\mathrm{v}}{\mathrm{u}-0}=-1 \quad \quad...
A body performs S.H.M. due to force 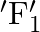 , with time period
, with time period 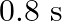 . If force is changed to
. If force is changed to 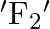 , it executes S.H.M. with time period
, it executes S.H.M. with time period 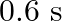 . Now both the forces act simultaneously in the same direction on the same body. New periodic time is
. Now both the forces act simultaneously in the same direction on the same body. New periodic time is
A) 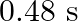
B)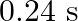
C)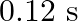
D)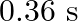
Correct option is A. $\ln \mathrm{SHM}, F=\frac{4 \pi^{2} m}{T^{2}} y$ or $T^{2} \propto \frac{1}{F}$ or $T \propto \frac{1}{\sqrt{F}}: \operatorname{So} 0.8 \propto \frac{1}{\sqrt{F_{1}}}$ and $0.6...
An ideal gas occupies a volume ‘ 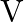 ‘ at a pressure ‘
‘ at a pressure ‘ 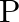 ‘ and absolute temperature
‘ and absolute temperature 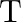 . The mass of each molecule is ‘
. The mass of each molecule is ‘ 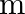 ‘. If ‘
‘. If ‘ 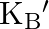 is the Boltzmann’s constant, then the density of gas is given by expression
is the Boltzmann’s constant, then the density of gas is given by expression
A)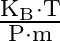
B)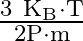
C)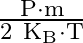
D)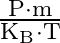
Correct option is D. The ideal gas law is given by $\mathrm{PV}=\mathrm{nRT}$, where $\mathrm{n}$ is the number of moles of a gas. $\mathrm{n}=$ mass of the gas $/$ molecular weight of the gas....
The depth of an ocean is 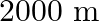 . The compressibility of water is
. The compressibility of water is 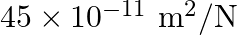 and density of water is
and density of water is 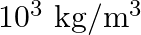 . At the bottom of the ocean, the fractional compression of water will be
. At the bottom of the ocean, the fractional compression of water will be 
A)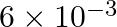
B)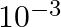
C)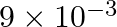
D)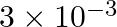
Correct option is C. compressibility is given as $\kappa=\frac{\frac{\Delta V}{V}}{\Delta P}$ $\Delta \mathrm{V}=\kappa \times \Delta \mathrm{P} \times \mathrm{V}$ Substituting values $\Delta...
For an ideal gas, if the ratio of Molar specific heats 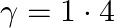 , then the specific heat at constant pressure
, then the specific heat at constant pressure 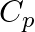 , specific heat at constant volume
, specific heat at constant volume 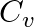 and corresponding molecule are respectively
and corresponding molecule are respectively
A)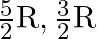 , monoatomic.
, monoatomic.
B)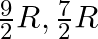 , polyatomic.
, polyatomic.
C)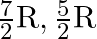 , non-rigid diatomic.
, non-rigid diatomic.
D) 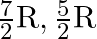 , rigid diatomic.
, rigid diatomic.
Correct option is D. Given, $\gamma=1.4=\frac{7}{5}$ We know, $C_p-C_v=R$ $\gamma=\frac{7}{5}=\frac{C_p}{C_v}$ $\therefore C_p=\frac{7}{5}C_v$ $\frac{7}{5}C_v-C_v=R$ $\frac{2}{5}C_v=R$...
A uniform wire has length ‘ 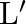 and weight ‘
and weight ‘ 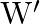 . One end of the wire is attached rigidly to a point in the roof and weight ‘
. One end of the wire is attached rigidly to a point in the roof and weight ‘ 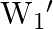 is suspended from its lower end. If ‘
is suspended from its lower end. If ‘ 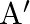 is the cross-sectional area of the wire then the stress in the wire at a height
is the cross-sectional area of the wire then the stress in the wire at a height 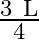 from its lower end is
from its lower end is
A)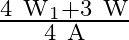
B)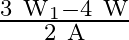
C)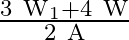
D)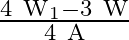
Correct option is A. $\text { stress }=\frac{\text { Tension }}{\text { Area }}$ Tension at height $\frac{3 \mathrm{~L}}{4}$ from lower end $\text { is } \frac{3}{4} \mathrm{w}+\mathrm{w}_{1}$ So,...
A small mass ‘ 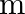 ‘ is suspended at the end of a wire having (negligible mass) length ‘L’ and cross-sectional area ‘
‘ is suspended at the end of a wire having (negligible mass) length ‘L’ and cross-sectional area ‘  ‘. The frequency of oscillation for the S.H.M. along the vertical line is [
‘. The frequency of oscillation for the S.H.M. along the vertical line is [ 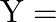 Young’s modulus of material of the wire ]
Young’s modulus of material of the wire ]
A)![Rendered by QuickLaTeX.com \frac{1}{2 \pi}\left[\frac{\mathrm{YAL}}{\mathrm{m}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-f3925ead959d4f80e07f99802608a95a_l3.png)
B)![Rendered by QuickLaTeX.com \frac{1}{2 \pi}\left[\frac{\mathrm{YA}}{\mathrm{mL}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-5b0a91f33eeb0af7f68b0f0b5612fc04_l3.png)
C)![Rendered by QuickLaTeX.com \frac{1}{2 \pi}\left[\frac{\mathrm{mA}}{\mathrm{YL}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-03f502b7ace2644cbea7275b6e92f850_l3.png)
D)![Rendered by QuickLaTeX.com \frac{1}{2 \pi}\left[\frac{\mathrm{YL}}{\mathrm{mA}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-bc772f47ba021ba390af8b01ac204fb0_l3.png)
Correct option is B. Let $F$ be the force applied on the end of wire and $x$ be the extension in the length of wire, then longitudinal strain $=\mathrm{x} / \mathrm{L}$ Normal stress $=$ F/A Young's...
Pascal’s law is not applied in
A) a hydraulic jack.
B) hydraulic breaks.
C) a hydraulic press.
D) an autuomiser.
Correct option is D.
A body of mass 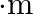 ‘ is moving along a circle of radius ‘
‘ is moving along a circle of radius ‘ 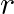 ‘ with linear speed ‘v’. Now, to change the linear speed to
‘ with linear speed ‘v’. Now, to change the linear speed to 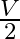 and to move it along the circle of radius ‘
and to move it along the circle of radius ‘ 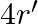 , required change in the centripetal force of the body is
, required change in the centripetal force of the body is
A) decrease by 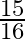
B) decrease by 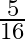
C) increase by 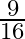
D) increase by 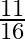
Correct answer is A. $\begin{array}{l} F_{1}=\frac{m v^{2}}{r} \\ F_{2}=\frac{m}{4 r} \cdot \frac{v^{2}}{4}=\frac{1}{16} \frac{m v^{2}}{r}=\frac{F_{1}}{16} \\ \therefore...
In communication with the help of antenna, the range covered (for the line of sight propagation) is initially ‘ 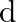 ‘. If the height of antenna is doubled, the range covered would become
‘. If the height of antenna is doubled, the range covered would become
A)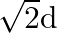
B) 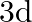
C) 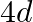
D) 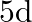
Correct option is A. Range of antenna $=d \sqrt{2 \mathrm{hr}}, \mathrm{h}=$ height of antenna, $\mathrm{R}=$ radius of earth if $\mathrm{h}$ is doubled i.e., $\mathrm{h}^{\prime}=2 \mathrm{~h}$,...
The electric field intensity at a point near and outside the surface of a charged conductor of any shape is 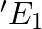 ‘. The electric field intensity due to uniformly charged infinite thin plane sheet is ‘
‘. The electric field intensity due to uniformly charged infinite thin plane sheet is ‘ 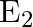 ‘. The relation between
‘. The relation between 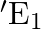 ‘ and ‘
‘ and ‘ 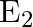 ‘ is:
‘ is:
A) 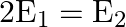
B) 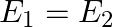
C) 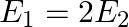
D) 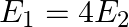
Correct option is C. Electric field outside the conductor $\mathrm{E}_{1}=\frac{\sigma}{\epsilon_{0}}$ Electric field due to uniformly charged infinitely thin plate $\mathrm{E}_{2}=\frac{\sigma}{2...
The relation between force ‘  ‘ and density ‘
‘ and density ‘  ‘ is
‘ is 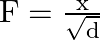 . The dimensions of
. The dimensions of 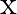 are
are
A) ![Rendered by QuickLaTeX.com \left[\mathrm{L}^{-1 / 2} \mathrm{M}^{3 / 2} \mathrm{~T}^{-2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-d0e831cace6d7fb698114df4b7d45d13_l3.png)
B) ![Rendered by QuickLaTeX.com \left[\mathrm{L}^{-1 / 2} \mathrm{M}^{1 / 2} \mathrm{~T}^{-2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-ca1924b4df824bd3aef6e1f25d91c1cf_l3.png)
C) ![Rendered by QuickLaTeX.com \left[\mathrm{L}^{-1} \mathrm{M}^{3 / 2} \mathrm{~T}^{-2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-3c203a52dfdc89b96eab16074bfa6721_l3.png)
D) ![Rendered by QuickLaTeX.com \left[\mathrm{L}^{-1} \mathrm{M}^{1 / 2} \mathrm{~T}^{-2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-8d414b1c70e8f1f0cecdb8869f66c412_l3.png)
Correct option is A. $\text { The dimensions of } \mathrm{x} \text { are...
In suspended type of moving coil galvanometer
A) coil is stationary.
B) magnet is stationary.
C) magnet and coil are stationary.
D) magnet and coil are moving.
Correct option is B.
A ring and a disc roll on the horizontal surface without slipping with same linear velocity. If both have same mass and total kinetic energy of the ring is 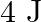 then total kinetic energy of the disc is
then total kinetic energy of the disc is
A)3 J
B)4 J
C)5 J
D)6 J
Correct option is A. Total kinetic energy of the body $\mathrm{K} \cdot \mathrm{E}_{\text {total }}=\left(\frac{1}{2} \mathrm{mv}^{2}\right)_{\text {translational }}+$ $\left(\frac{1}{2}...
The electron in the hydrogen atom is moving with a speed of 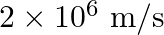 in an orbit of radius
in an orbit of radius 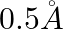 . The magnetic moment of the revolving electron is
. The magnetic moment of the revolving electron is
A)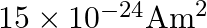
B)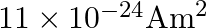
C)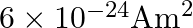
D)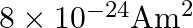
Correct option is D. $\text { Magnetic moment, } \mathrm{m}_{0}=\frac{\text { evr }}{2}$ $=\frac{1.6 \times 10^{-19} \times 2\times 10^{6} \times 0.5 \times 10^{-10}}{2}=8 \times 10^{-24}...
A mass ‘ 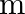 ‘ suspended from a spring stretches it by
‘ suspended from a spring stretches it by 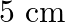 when on the surface of the earth . The mass is then taken on to a height of
when on the surface of the earth . The mass is then taken on to a height of 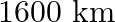 above earth’s surface and again suspended from the same spring. At this altitude the extension of the spring is
above earth’s surface and again suspended from the same spring. At this altitude the extension of the spring is  (Radius of earth
(Radius of earth 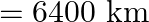 )
)
A)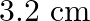
B) 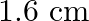
C) 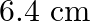
D)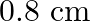
Correct option is A. $\begin{array}{l} g=\frac{G M}{r^{2}} \\ \therefore \frac{g^{\prime}}{g}=\frac{r^{2}}{r^{2}} \\ R=6400 \mathrm{~km}, r^{\prime}=6400+1600=8000 \mathrm{~km} \\...
For a photocell, the work function is ‘ 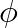 ‘ and the stopping potential is ‘
‘ and the stopping potential is ‘ 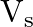 ‘. The wavelength of the incident radiation is
‘. The wavelength of the incident radiation is
A)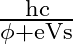
B)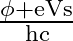
C)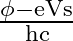
D)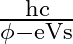
Correct option is A. $\begin{array}{l} h v=\phi+\frac{1}{2} m v_{\max }^{2} \text { or } \frac{h c}{\lambda}=\phi+e V_{s} \\ \therefore \lambda=\frac{h c}{\phi+e V_{s}} \end{array}$
The period of oscillation of a mass M suspended from a spring of negligible mass is 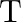 . If along with it another mass
. If along with it another mass 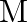 is also suspended, the period of oscillation will now be
is also suspended, the period of oscillation will now be
A)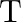
B)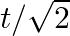
C)
D)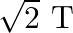
Correct option is D$\sqrt{2} \mathrm{~T}$ $\mathrm{T}=2 \pi \sqrt{\frac{\mathrm{M}}{\mathrm{k}}}, \mathrm{T}^{\prime}=2 \pi \sqrt{\frac{2 \mathrm{M}}{\mathrm{k}}}=\sqrt{2} \mathrm{~T}$
A Plano convex lens is made of glass of refractive index 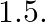 The of curvature of its convex surface is R. Its focal length:
The of curvature of its convex surface is R. Its focal length:
A) 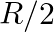
B) 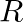
C) 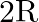
D) 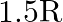
Correct option is C) ${2} \mathrm{R}$ Given, Radius of curvature, $\mathrm{R}$. Refractive index $=1.5$ Focal length of lens, $f=\frac{\mathrm{R}}{(\mu-1)}=\frac{\mathrm{R}}{(1.5-1)}=2 \mathrm{R}$
If 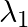 and
and 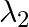 are the wavelengths of the first members of the Lyman and Paschen series respectively, then
are the wavelengths of the first members of the Lyman and Paschen series respectively, then  is:
is:
A) 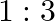
B) 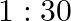
C) 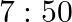
D) 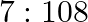
Correct option is D) $7: 108$ For first line of Lyman series, $\begin{array}{l} \mathrm{m}_{1}=1 \text { and }_{\mathrm{n} 2}=3 \\ \therefore...
A solenoid has core of a material with relative permeability 500 and its windings carry a current of 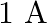 . The number of turns of the solenoid is 500 per metre. The magnetization of the material is nearly.
. The number of turns of the solenoid is 500 per metre. The magnetization of the material is nearly.
A) 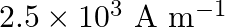
B) 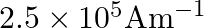
C)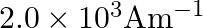
D)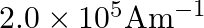
Correct option is B) $2.5 \times 10^{5} \mathrm{Am}^{-1}$ Here, $\mathrm{n}=500$ turns $/ \mathrm{m}$ $\mathrm{I}=1 \mathrm{~A}, \mu_{\mathrm{r}}=500$ Magnetic intensity, $\mathrm{H}=\mathrm{nI}=500...
Water rises in a capillary tube to a certain height such that the upward force due to surface tension is balanced by 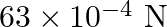 force due to the weight of the water. The surface tension of water is
force due to the weight of the water. The surface tension of water is 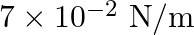 . The inner diameter of the capillary tube is nearly
. The inner diameter of the capillary tube is nearly 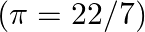
A)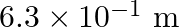
B)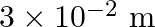
C)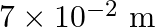
D)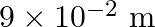
Correct answer is B. Upwards force due to surface tension, $F_{u}=(2 \pi R) \times T$ where, $2 \pi R \rightarrow$ circumference of tube $F_{u}=(2 \pi R) \times T$ Because surface tension is force...
When an alternating emf is applied across a capacitor 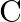 , the graph of capacitive reactance
, the graph of capacitive reactance 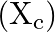 with frequency (
with frequency ( 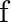 ) of the source of alternating signal is
) of the source of alternating signal is
Correct answer is B.
A proton moving in perpendicular magnetic field possess energy ‘ 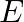 ‘. The magnetic field is increased four times. But the proton is constrained to move in the path of same radius. The kinetic energy will increase
‘. The magnetic field is increased four times. But the proton is constrained to move in the path of same radius. The kinetic energy will increase
A) 16 times.
B) 8 times.
C) 2 times.
D) 4 times.
Correct option is A. Radius, $\begin{aligned} R &=\frac{m v}{9 B}=\frac{\sqrt{m} \sqrt{m} v^{2}}{9 B} \\ &=\frac{\sqrt{m} \sqrt{E}}{9 B} \end{aligned}$ if $B \rightarrow 4 B$....
The moment of inertia of a ring about an axis passing through the centre and perpendicular to its plane is ‘I’. It is rotating with angular velocity 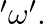 Another identical ring is gently placed on it so that their centres coincide. If both the rings are rotating about the same axis then loss is kinetic energy is
Another identical ring is gently placed on it so that their centres coincide. If both the rings are rotating about the same axis then loss is kinetic energy is
A) 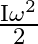
B) 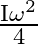
C) 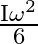
D) 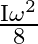
Correct option is B $\frac{\mathrm{I} \omega^{2}}{4}$ Initial angular momentum, $\mathrm{L}_{1}=\mathrm{I} \omega$ $\mathrm{K}_{1}=\frac{1}{2} \mathrm{I} \omega^{2}$ When, second ring is put on it...
A force 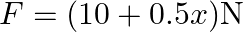 acts on a particle in
acts on a particle in 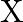 direction, where
direction, where 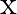 is in meters. Find the work done by this force during a displacement from
is in meters. Find the work done by this force during a displacement from 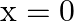 to
to 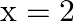 .
.
A) 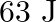
B) 
C)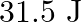
D)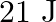
Correct option is D. Force $\mathrm{F}=10+0.5 \mathrm{x}$ Work done $\mathrm{W}=\int \mathrm{F} \mathrm{dx}$ $\begin{array}{l} \mathrm{W}=\int_{0}^{2}(10+0.5 \mathrm{x}) \mathrm{dx} \\...
The effective length of a magnet is 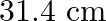 and its pole strength is
and its pole strength is 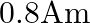 . The magnetic moment, if it is bent in the form of a semicircle is
. The magnetic moment, if it is bent in the form of a semicircle is 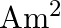
A) 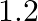
B)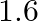
C) 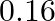
D) 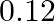
Correct option is C $0.16$ The effective length of magnet $=31.4 \mathrm{~cm}$ Pole strength $\mathrm{m}=0.8 \mathrm{Am}$ Length of semi-circle $=\pi \frac{\mathrm{D}}{2}=1$ where, $\mathrm{D}=$...
A particle of mass 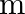 is rotating in a plane in circular path of radius
is rotating in a plane in circular path of radius 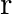 . Its angular momentum is
. Its angular momentum is 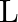 . The centripetal force acting on the particle is.
. The centripetal force acting on the particle is.
A) 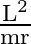
B) 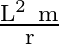
C) 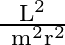
D) 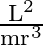
Correct option is D) $\frac{\mathrm{L}^{2}}{\mathrm{mr}^{3}}$ Centripetal force, $F=\frac{\mathrm{mv}^{2}}{\mathrm{r}}=\frac{\mathrm{m}}{\mathrm{r}} \frac{\mathrm{L}^{2}}{\mathrm{~m}^{2}...
The mutual inductance between two planar concentric rings of radii 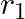 and
and 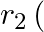 with
with 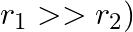 placed in air is given by
placed in air is given by
A) 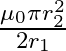
B) 
C) 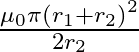
D) 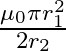
The correct option is $\mathbf{A}$$\frac{\mu_{0} \pi r_{2}^{2}}{2 r_{1}}$ Magnetic field due to the larger coil at its centre is $B=\frac{\mu_{0} I}{2 r_{1}}$ where $\mathrm{I}$ is the current in...
Which of the following reaction does NOT yield an amine
(1) (2) (3) (4) Correct Answer: Option (4) Explanation: The given reaction in the fourth option does not yield an amine whereas the rest of the reactions...
Identity ‘Z’ in the following series of reaction
1.Butan-1-ol 2.2-chlorobutane 3.Butan-2-ol 4.But-2-ene Correct Answer: 3.Butan-2-ol Explanation: Identification of Z - Butan-2-ol
Which of the following compounds is obtained when t-butyl bromide is treated with alcoholic ammonia?
(1) (2) (3) (4) Correct Answer: Option (3) Explanation:
Identify the product Y in the following reaction
1.Gluconic acid 2.Saccharic acid 3.n-Hexane 4.Glucoxime Correct Answer: 2. Saccharic acid Explanation: Identification of Y - Saccharic acid
Identify ‘A’ in the following reaction
1. 2.R-NH-OH 3.R-COOH 4.R-NH2 Correct Answer: 2.R-NH-OH Explanation: Identification of A - R-NH-OH
Which among the following statements about terpenes is NOT true?
1.Terpenes occur in essential oils 2.Terpenes include vitamin A, E and K 3.Terpenes consist of isoprene units 4.Terpenes are saturated hydrocarbons Correct Answer: 4. Terpenes are saturated...
How many pi bonds and sigma bound are present in following molecule?
1.5 pi,14 sigma - bonds 2.3 pi,17 sigma - bonds 3.3 pi, 17 sigma - bonds 4.2 pi, sigma - bonds Correct Answer: 3.3 pi, 17 sigma - bonds Explanation: 3 pi, 17 sigma - bonds are...
A resistance wire connected in the left gap of a metre bridge balances a 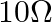 resistance in the right gap at a point which divides the bridge wire in the ratio
resistance in the right gap at a point which divides the bridge wire in the ratio 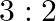 . If the length of the resistance wire is
. If the length of the resistance wire is 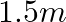 , then the length of
, then the length of 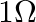 of the resistance wire is:
of the resistance wire is:
(1) 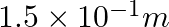
(2) 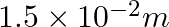
(3) 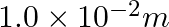
(4) 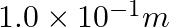
Correct option: (4) Explanation: Using the given ratio of wire, we have, $x \times 2 \lambda^{3}=10 \times 3 \lambda$ $x=15 \Omega$ $\therefore 15 \Omega \rightarrow 1.5 m$ $\therefore 1 \Omega...
The increase in the width of the depletion region in a p-n junction diode is due to:
(1) both forward bias and reverse bias
(2) increase in forward current
(3) forward bias only
(4) reverse bias only
Correct option: (4) Explanation: The absence of electrons and holes in the depletion region causes the region's width to increase. This only happens in the case of reverse bias in a diode.
In Young’s double slit experiment, if the separation between coherent sources is halved and the distance of the screen from the coherent sources is doubled, then the fringe width becomes :
(1) four times
(2) one-fourth
(3) double
(4) half
Correct option: (1) Explanation: In YDSE, the fringewidth is given by $\beta=\frac{\lambda D}{d} \quad \beta^{\prime}=\frac{\lambda 2 D}{d / 2}=4 \beta$
Aluminium crystallizes in face centered cubic structure, having atomic radius 125pm. The edge length of unit cell of aluminium is
253.5 pm 353.5 pm 465 pm 250 pm Solution: 353.5 pm $ For\text{ }a~FCC~or~~CCP~unit\text{ }cell,we\,have: $ $ 4r=\surd 2\times a $ $ \Rightarrow a=\frac{4r}{\surd 2}=353.5pm $
A ray is incident at an angle of incidence i on one surface of a small angle prism (with angle of prism A) and emerges normally from the opposite surface. If the refractive index of the material of the prism is  , then the angle of incidence is nearly equal to:
, then the angle of incidence is nearly equal to:
(1) 
(2) 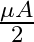
(3) 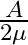
(4) 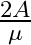
Correct option: (1) Explanation: As we know, $1 \sin i=\mu . \sin A$ For small angle approximation $\sin \theta=\theta$ $i=\mu A$
A 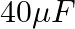 capacitor is connected to a
capacitor is connected to a 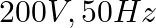 ac supply. The rms value of the current in the circuit is, nearly:
ac supply. The rms value of the current in the circuit is, nearly:
(1) 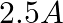
(2) 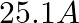
(3) 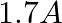
(4) 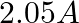
Correct option: (1) Explanation: As we know, $I_{r m s}=\frac{V_{r m s}}{Z}=\frac{200}{X_{C}}=200.2 \pi f . C$ Calculating the value by substituting the value, $=200 \times 2 \times 3.14 \times 50...
The solids which have the negative temperature coefficient of resistance are :
(1) semiconductors only
(2) insulators and semiconductors
(3) metals
(4) insulators only
Correct option: (2) Insulators have a negative temperature coefficient because their resistance decreases as the temperature rises. As the temperature rises, the semiconductor material's resistivity...
Which of the following is Rosenmund reduction?
(1) (2) (3) (4) Correct Answer: (3) Explanation: Rosenmund reduction reaction -
A charged particle having drift velocity of 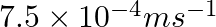 in an electric field of
in an electric field of 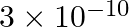
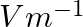 has a mobility in
has a mobility in 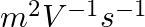 of :
of :
(1) 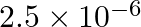
(2) 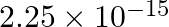
(3) 
(4) 
Correct option: (4) As we know that, $v _{ d }=\mu E \Rightarrow \mu=\frac{ V _{ d }}{ E }$ Calculating, $=\frac{7.5 \times 10^{-4}}{3 \times 10^{-10}}$ $\mu=2.5 \times 10^{6}$ $(\mu=$ mobility...
The capacitance of a parallel plate capacitor with air as medium is 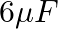 . With the introduction of a dielectric medium, the capacitance becomes
. With the introduction of a dielectric medium, the capacitance becomes 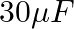 . The permittivity of the medium is :
. The permittivity of the medium is : 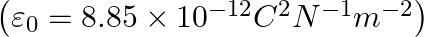
(1) 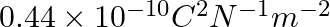
(2) 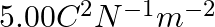
(3) 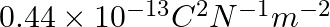
(4) 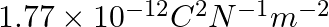
Correct option: (1) Explanation: Given: $C_{\text {air }}=6 \mu F , C_{\text {medium }}=30 \mu F$ As we know, $C_{\text {air }}=\frac{\varepsilon_{0} A}{d}, C_{\text {medium }}=\frac{\varepsilon_{0}...
An iron rod of susceptibility 599 is subjected to a magnetising field of 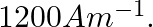 The permeability of the material of the rod is :
The permeability of the material of the rod is : 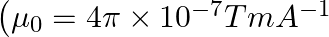 )
)
(1) 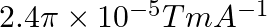
(2) 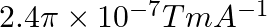
(3) 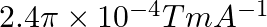
(4) 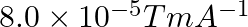
Correct option (3) Explanation: Given: $x _{ m }=599, \mu_{0}=4 \pi \times 10^{-7}$ $H =1200 A / m , \mu=?$ As we know, $\mu=\mu_{0}\left(1+ x _{ m }\right)$ Now, $=4 \pi \times 10^{-7}(1+599)$ $=4...
Two cylinders A and B of equal capacity are connected to each other via a stop cock. A contains an ideal gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is :
(1) isochoric
(2) isobaric
(3) isothermal
(4) adiabatic
Correct option (4) When cock is removed, dQ = 0 for the thermally insulated system. Because dw = 0 in vacuum Walls that are thermally insulated du + dw = dQ So du also a zero.
Identify the correct statement from the following :
(1) Blister copper has blistered appearance due to evolution of CO2.
(2) Vapour phase refining is carried out for Nickel by Van Arkel method.
(3) Pig iron can be moulded into a variety of shapes.
(4) Wrought iron is impure iron with 4% carbon.
Correct option: (3) Explanation: Because of the evolution of $SO_2$, not $CO_2$, blister copper has a blistered appearance. The Van Arkel method is used to obtain ultra-pure Titanium through vapor...
An alkene on ozonolysis gives methanal as one of the product. Its structure is
Correct option: (2) On ozonolysis, the structure in option B produces methane, as shown in the reaction above.
When carbolic acid is heated with concentrated nitric acid in presence of concentrated sulphuric acid it forms
1.benzoic acid 2.picric acid 3.phthalic acid 4.benzene sulphonic acid Correct Answer: 2. picric acid Explanation: When carbolic acid is heated with concentrated nitric acid in presence of...
For the following reaction, what is the value of ∆S(total) at 298k?
Fe2O3(s) + 3 CO(g) 2Fe(s) + 3CO2(g) o = -29.8 KJ and So =15JK-1. 1.29.8 J 2.100.0 J 3.298.0J 4.115.OJ Correct Answer: 4.115.OJ Explanation: The value of ∆S(total) at 298k is...
What is the oxidation number of carbons in glucose?
1.-6 2.+6 3.+3 4.Zero Correct Answer: 4. Zero Explanation: Zero is the oxidation number of carbons in glucose.
