a) Zinc sulfate reacts with NaOH in both feasible amounts and excess. b) Copper sulfate reacts with $NH_4OH$ in both feasible amount and excess.
a) What do you understand by amphoteric oxide?
(b) Give the balanced equations for the reaction with two different amphoteric oxides with caustic alkali.
(c) Name the products formed.
Answer: (a) Amphoteric oxides are substances that react with both acids and alkalis to produce salt and water as byproducts. (b) ZnO + 2NaOH → Na2ZnO2 + H2O Al2O3 +...
What is observed when hot concentrated caustic soda solution is added to (a) Zinc (b) Aluminium? Write balanced equations.
Answer: a) In the presence of hot concentrated caustic soda or hot concentrated sodium hydroxide, zinc reacts to form zincate sodium and hydrogen. Zn + 2NaOH → Na2ZnO2 + H2 b) Aluminum reacts with...
How will you distinguish lead carbonate and zinc carbonate in solution?
Solution: They can be identified by dissolving them in dilute nitric acid and then dissolving them in excess ammonium hydroxide. When lead carbonate is dissolved in weak nitric acid, and then...
Distinguish by adding: sodium hydroxide solution and ammonium hydroxide solution to
(a) Calcium salt solution and lead salt solution
(b) Lead nitrate solution and zinc nitrate solution
Solution: a) The interaction of sodium hydroxide and ammonium hydroxide with calcium salt produces a milky white precipitate Ca(OH)2, whereas the reaction of sodium hydroxide and ammonium hydroxide...
Distinguish by adding: sodium hydroxide solution and ammonium hydroxide solution to
(c) Copper salt solution and ferrous salt solution
(d) Fe(II) salt solution and Fe(III) salt solution
Solution: (c) The combination of sodium hydroxide and ammonium hydroxide with copper salt produces a pale blue-colored precipitate, whereas the reaction with ferrous salt solution produces a dirty...
Distinguish by adding: sodium hydroxide solution and ammonium hydroxide solution to (e) Ferrous nitrate and lead nitrate
Answer: (e) The reaction of ammonium hydroxide with lead nitrate produces a chalky white insoluble precipitate, but the reaction with ferrous nitrate does not produce any precipitate.
You are provided with two reagent bottles marked A and B. One of which contains 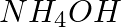 solution and the other contains NaOH solution. How will you identify them by a chemical test?
solution and the other contains NaOH solution. How will you identify them by a chemical test?
Solution: Calcium salts, such as calcium chloride, can be used to distinguish between reagent bottles A and B Ca(NO3)2. On adding NaOH to Ca(NO3)2, a white precipitate Ca...
What do you observe when freshly precipitated aluminum hydroxide reacts with caustic soda solution? Give a balanced equation.
Answer: In a reaction between newly precipitated aluminum hydroxide and caustic soda solution, sodium meta aluminate is formed as a white salt. Al(OH)3 + NaOH → NaAlO2 {Sodium meta aluminate} +...
Select the correct answers:
(c) A metal which produces hydrogen on reacting with alkali as well as with acid.
(i) Iron (ii) Magnesium
(iii) Zinc (iv) Copper
(d) The salt solution which does not react with ammonium hydroxide is
(i) Calcium nitrate (ii) Zinc nitrate
(iii) Lead nitrate (iv) Copper nitrate
Solution: Copper also reacts with dil HCl and produces hydrogen and it also reacts with NaOH. (c) (iii) Zn + 2NaOH → Na2ZnO2 {Sodium zincate} (Colourless) + H2 Zn + HCl →...
Select the correct answers:
(a) Colour of an aqueous solution of copper sulphate is
(i) Green (ii) Brown
(iii) Blue (iv) Yellow
(b) Colour of the precipitate formed on adding NaOH solution to iron (II) sulphate solution is
(i) White (ii) Brown
(iii) Green (iv) Pale blue
Solution: (a) (iii) An aqueous solution of copper sulfate is blue. (b) (iii) FeSO4 {Dirty green} + 2NaOH → Fe(OH)2 ↓ {gelatinous ppt.} + Na2SO4
Name
(a) A yellow monoxide that dissolves in hot and concentrated caustic alkali.
(b) A white, insoluble oxide that dissolves when fused with caustic soda or caustic potash.
(c) A compound containing zinc in the anion.
Answer: (a) PbO- Lead Oxide (b) ZnO- Zinc Oxide (c) K2ZnO2
On adding dilute ammonia solution to the colorless solution of a salt, a white gelatinous precipitate appears. This precipitate, however, dissolves in the addition of an excess of ammonia solution. Identity (choose from Na, Al, Zn,Pb, Fe)
(a) Which metal salt solution was used?
(b) What is the formula of the white gelatinous precipitate obtained?
Solution: (a) Zinc metal salt solution was used. $ZnCl_2$(b) The formula of white gelatinous precipitate is $Zn(OH)_2$
Name the chloride of a metal that is soluble in excess of ammonium hydroxide. Write an equation for the same.
Answer: Zinc chloride (ZnCl2) is soluble in excess of ammonium hydroxide. ZnCl2 + 2NH4OH → Zn(OH)2 ↓ {White gelatinous ppt.} + 2NH4Cl With an excess of NH4OH ppt. dissolves Zn(OH)2 + 2NH4Cl +...
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess.
(c) 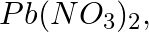
(d) 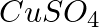 ? Write balanced equations for these reactions.
? Write balanced equations for these reactions.
Solution: Caustic soda is Sodium hydroxide (NaOH) (c) Pb(NO3)2 + 2NaOH → Pb(OH)2 ↓ {White ppt. (colourless)}+ 2NaNO3 In excess of alkali, the white precipitate of Pb(OH)2 becomes soluble. Pb(OH)2 +...
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess.
(a) 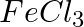 , (b)
, (b) 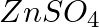 Write balanced equations for these reactions.
Write balanced equations for these reactions.
Solution: Caustic soda is Sodium hydroxide (NaOH) (a) FeCl3 + 3NaOH → Fe(OH)3 ↓ {Reddish-brown ppt.}+ 3NaCl In excess of alkali, the reddish-brown ppt. of Fe(OH)3 remains insoluble. (b) ZnSO4 +...
What happens when ammonia solution is added first dropwise and then in excess to the following solutions: (i) CuSO4 (ii) ZnSO4 (iii)FeCl3. Write balanced equations for these reactions.
Solution: (i) CuSO4 + 2NH4OH → Cu(OH)2 ↓ + (NH4)2SO4 With an excess of NH4OH, ppt. dissolves Cu(OH)2 + (NH4)2SO4 + 2NH4OH → [Cu(NH3)4]SO4 + 4H2O ii) ZnSO4 + 2NH4OH → Zn(OH)2 ↓ + (NH4)2SO4 With an...
What happens when ammonia solution is added first dropwise and then in excess to the following solutions: (iii)FeCl3. Write balanced equations for these reactions.
Answer: FeCl3 + 3NH4OH → Fe(OH)3 ↓ + 3NH4Cl
Write balanced equations for Q.2 (g) and (i).
Solution: For 2(g): For 2(i): PbO + 2NaOH → Na2PbO2 + H2O (Yellow) Sodium Plumbate (Colourless, soluble)
Name
(i) a colored metallic oxide that dissolves in alkalis to yield colorless solutions.
(j) a colourless cation, not a representative element.
Answer: (i) PbO (j) Ammonium ion
Name
(g) a metal that evolves into a gas that burns with a pop sound when boiled with alkali solutions.
(h) Two bases that are not alkalis but dissolve in strong alkalis.
Answer: (g) Aluminium (h) Zn(OH)2 and Al(OH)3
Name:
(e) Two colourless metal ions.
(f) Two coloured metal ions.
Answer: (e) Na+, Ca2+ (f) Fe2+, Mn2+
Name:
(c) a strong alkali.
(d) a weak alkali.
Answer: (c) NaOH : A strong alkali dissociates completely to form OH- ions. (d) NH4OH: A weak alkali only partially dissociates to form OH- ions.
Name:
(a) a metallic hydroxide soluble in excess of 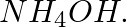
(b) a metallic oxide soluble in excess of caustic soda solution.
Answer: (a) Cu(OH)2 : Copper (II) hydroxide is amphoteric in nature and soluble in excess ammonium hydroxide. (b) ZnO: It is amphoteric which means that it dissolves in both acidic and basic...
Write the probable color of the following salts?
(a) Ferrous salts (b) Ammonium salts
(c) Cupric salts (d) Calcium salts
(e) Aluminium salts
Answer: (a) Ferrous salts – Light green (b) Ammonium salts – Colourless (c) Cupric salts – Blue (d) Calcium salts – Colourless (e) Aluminium salts...
