If there is no current flowing in the conductors that are parallel to each other, the magnetic forces do not obey Newton's third law.
An electron enters with a velocity v = v0i into a cubical region in which there are uniform electric and magnetic fields. The orbit of the electron is found to spiral down inside the cube in the plane parallel to the x-y plane. Suggest a configuration of fields E and B that can lead to it.
The spiral route is formed by the fields E and B in their current configuration.
A charged particle of charge e and mass m is moving in an electric field E and magnetic field B. Construct dimensionless quantities and quantities of dimension [T]-1.
mv2/R = evB eB/m = v/R = ꞷ B = F/ev = [MA-1T-2] [ꞷ] = [eB/m]=[v/R] = [T-1]
A current-carrying loop consists of 3 identical quarter circles of radius R, lying in the positive quadrants of the x-y, y-z, and z-x planes with their centres at the origin, joined together. Find the direction and magnitude of B at the origin.
The quarter's vector sum of the magnetic field at the origin is given as \({{\vec{B}}_{net}}=\frac{1}{4}\left( \frac{{{\mu }_{0}}I}{2R} \right)(\widehat{i}+\widehat{j}+\widehat{k})\)
Describe the motion of a charged particle in a cyclotron if the frequency of the radio frequency (rf) field were doubled.
The time period of the radio frequency is halved when the frequency is doubled, resulting in a half revolution of the charges.
The magnetic force depends on v which depends on the inertial frame of reference. Does then the magnetic force differ from inertial frame to frame? Is it reasonable that the net acceleration has a different value in different frames of reference?
Because velocity varies depending on frame of reference, the net acceleration might have a different value in different frames of reference.
Show that a force that does no work must be a velocity dependent force.
\(dW=\vec{F}\cdot dl=0\) \(\vec{F}\cdot \vec{v}=0\)
Verify that the cyclotron frequency ꞷ = eB/m has the correct dimensions of [T]-1.
In a cyclotron, the particle follows a circular path, with magnetic force acting as a centripetal force. mv2/R = evB eB/m = v/R = ꞷ B = F/ev = [MLT-2]/[AT][LT-1] = [MA-1T-2] [ꞷ] = [eB/m] = [v/R] =...
A charged particle would continue to move with a constant velocity in a region wherein, a) E = 0, B ≠ 0 b) E ≠ 0, B ≠ 0 c) E ≠ 0, B = 0 d) E = 0, B = 0
a) E = 0, B ≠ 0 b) E ≠ 0, B ≠ 0 d) E = 0, B = 0
A cubical region of space is filled with some uniform electric and magnetic fields. An electron enters the cube across one of its faces with velocity v and a positron enters via opposite face with velocity –v. At this instant, a) the electric forces on both the particles cause identical acceleration b) the magnetic forces on both the particles cause equal accelerations c) both particles gain or lose energy at the same rate d) the motion of the centre of mass (CM) is determined by B alone
b) the magnetic forces on both the particles cause equal accelerations c) both particles gain or lose energy at the same rate d) the motion of the centre of mass (CM) is determined by B alone
Two identical current-carrying coaxial loops, carry current I in an opposite sense. A simple amperian loop passes through both of them once. Calling the loop as C, a)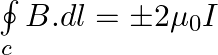 b) the value of
b) the value of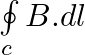 c) there may be a point on C where B and dl are perpendicular d) B vanishes everywhere on C
c) there may be a point on C where B and dl are perpendicular d) B vanishes everywhere on C
b) the value of \(\oint\limits_{c}{B.dl\) is independent of sense of C c) there may be a point on C where B and dl are perpendicular
Consider a wire carrying a steady current, I placed un a uniform magnetic field B perpendicular to its length. Consider the charges inside the wire. It is known that magnetic forces do not work. This implies that a) motion of charges inside the conductor is unaffected by B since they do not absorb energy b) some charges inside the wire move to the surface as a result of B c) if the wire moves under the influence of B, no work is done by the force d) if the wire moves under the influence of B, no work is done by the magnetic force on the ions, assumed fixed within the wire
b) some charges inside the wire move to the surface as a result of B d) if the wire moves under the influence of B, no work is done by the magnetic force on the ions, assumed fixed within the wire
The gyro-magnetic ratio of an electron in an H-atom, according to Bohr model is a) independent of which orbit it is in b) negative c) positive d) increases with the quantum number n
a) independent of which orbit it is in b) negative
A circular current loop of magnetic moment M is in an arbitrary orientation in an external magnetic field B. The work done to rotate the loop by 30o about an axis perpendicular to its plane is a) MB b) √3 MB/2 c) MB/2 d) zero
d) zero
In a cyclotron, a charged particle a) undergoes acceleration all the time b) speeds up between the dees because of the magnetic field c) speeds up in a dee d) slows down within a dee and speeds up between dees
a) undergoes acceleration all the time
An electron is projected with uniform velocity along the axis of a current-carrying long solenoid. Which of the following is true? a) the electron will be accelerated along the axis b) the electron path will be circular about the axis c) the electron will experience a force at 45o to the axis and hence execute a helical path d) the electron will continue to move with uniform velocity along the axis of the solenoid
d) the electron will continue to move with uniform velocity along the axis of the solenoid
A current circular loop of radius R is placed in the x-y plane with centre at the origin. Half of the lop with x > 0 is now bent so that it now lies in the y-z plane. a) the magnitude of magnetic moment now diminishes b) the magnetic moment does not change c) the magnitude of B at (0,0,z),z >> R increases d) the magnitude of B at (0,0,z),z >> R is unchanged
a) the magnitude of magnetic moment now diminishes
Biot-Savart law indicates that the moving electrons produce a magnetic field B such that a) B ┴ v b) B ‖ v c) it obeys inverse cube law d) it is along the line joining the electrons and point of observation
a) B ┴ v
Two charged particles traverse identical helical paths in an opposite sense in a uniform magnetic field a) they have equal z-components of momenta b) they must have equal charges c) they necessarily represent a particle-antiparticle pair d) the charge to mass ratio satisfy: (e/m)1 + (e/m)2 = 0
d) the charge to mass ratio satisfy: (e/m)1 + (e/m)2 = 0
There are two current-carrying planar coils made each from identical wires of length L. C1 is circular and C2 is square. They are so constructed that they have the same frequency of oscillation when they are placed in the same uniform B and carry the same current. Find a in terms of R.
The circular coil C1 has a radius of R, a length of L, and a number of turns per unit length of n1 = L/2R. The square C2 has a side, a perimeter, and a number of turns per unit length of n2 = L/4a....
Consider the plane S formed by the dipole axis and the axis of the earth. Let P be a point on the magnetic equator and in S. Let Q be the point of intersection of the geographical and magnetic equators. Obtain the declination and dip angle at P and Q.
The declination is zero, P is in the plane, S is in the north, and P is in the plane. The declination for point P is 0 since it is in the plane S created by the dipole axis and the earth's axis....
Assume the dipole model for earth’s magnetic field B which is given by Bv = vertical component of magnetic field = μ0/4π 2m cos θ/r3, BH = horizontal component of magnetic field = μ0/4π 2m sin θm/r3, θ = 90o latitude as measured from magnetic equator. Find loci of points for which i) |B| is minimum ii) dip angle is zero, and iii) dip angle is ±45o.
a) |B| is minimum at the magnetic equator. b) Angle of dip is zero when θ = π/2 c) When dip angle is ±45o θ = tan-1 is the locus.
What are the dimensions of χ, the magnetic susceptibility? Consider an H-atom. Guess an expression for χ, up to a constant by constructing a quantity of dimensions of χ, out of parameters of the atom: e, m, v, R and μ0. Here, m is the electronic mass, v is electronic velocity, R is Bohr radius. Estimate the number so obtained and compare with the value of | χ| equivalent to 10-5 for many solid materials.
χm = I/H = intensity of magnetisation/magnetising force χ is dimensionless as I and H has the same units χ = 10-4
Verify the Ampere’s law for the magnetic field of a point dipole of dipole moment Take C as the closed curve running clockwise along i) the z-axis from z = a > 0 to z = R; ii) along the quarter circle of radius R and centre at the origin, in the first quadrant of x-z plane; iii) along the x-axis from x = R to x = a and iv) along the quarter circle of radius a and centre at the origin in the first quadrant of the x-z plane.
Magnetic field = 0M/4(1/a2-1/R2) along the z-axis b) On the circular arc, the magnetic field at point A is = 0m/4R2. c) (d) The magnetic moment is 0
Use i) the Ampere’s law for H and ii) continuity of lines of B, to conclude that inside a bar magnet a) lines of H run from the N pole to S pole, while b) lines of B must run from the S pole to N pole.
The amperian loop is denoted by the letter C. We can find the angle between the two points by solving the above equation. cos is negative because it is greater than 90o.
In three moles of ethane (C2H6), calculate the following: (i) Number of moles of carbon atoms. (ii) Number of moles of hydrogen atom (iii) Number of molecules of ethane
(a) 1 mole ${{C}_{2}}{{H}_{6}}$ contains two moles of C- atoms. ∴∴ No. of moles of C- atoms in 3 moles of ${{C}_{2}}{{H}_{6}}$ = 2 * 3 = 6 (b) 1 mole ${{C}_{2}}{{H}_{6}}$ contains six moles of H-...
Calculate the atomic mass (average) of chlorine using the following data:
Average atomic mass of Cl. = $[(\text{Fractional abundance of }\!\!~\!\!\text{ }_{{}}^{35}Cl)(\text{molar mass of }\!\!~\!\!\text{ }_{{}}^{35}Cl)+(\text{fractional abundance of }\!\!~\!\!\text{...
Determine the molecular formula of an oxide of iron, in which the mass percent of iron and oxygen are 69.9 and 30.1, respectively.
Mass percent of Fe = 69.9% Mass percent of O = 30.1% No. of moles of Fe present in oxide =$ \frac{69.90}{55.85}$ = 1.25 No. of moles of O present in oxide =$\frac{30.1}{16.0}$ =1.88 Ratio of Fe to...
How much copper can be obtained from 100 g of copper sulphate (CuSO4)?
1 mole of $CuSO_{4}$ contains 1 mole of Cu. Molar mass of $CuSO_{4}$ = (63.5) + (32.00) + 4(16.00) = 63.5 + 32.00 + 64.00 = 159.5 grams 159.5 grams of $CuSO_{4}$ contains 63.5 grams of Cu....
Calculate the concentration of nitric acid in moles per litre in a sample which has a density, 1.41 g mL–1 and the mass per cent of nitric acid in it being 69%
Mass percent of HNO3 in sample is 69 % Thus, 100 g of HNO3 contains 69 g of HNO3 by mass. Molar mass of HNO3 = { 1 + 14 + 3(16)} g.mol^{-1}g.mol−1 = 1 + 14 + 48 = 63g mol^{-1}=63gmol−1 Now,...
Calculate the mass of sodium acetate 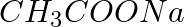 required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol–1.
required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol–1.
0.375 Maqueous solution of $CH_{3}COONa$ = 1000 mL of solution containing 0.375 moles of $CH_{3}COONa$ Therefore, no. of moles of $CH_{3}COONa$ in 500 mL = 0.1875 mole Molar mass of sodium acetate...
Calculate the amount of carbon dioxide that could be produced when (i) 1 mole of carbon is burnt in air. (ii) 1 mole of carbon is burnt in 16 g of dioxygen. (iii) 2 moles of carbon are burnt in 16 g of dioxygen.
(i) 1 mole of carbon is burnt in air. $C+{{O}_{2}}\to C{{O}_{2}}$ 1 mole of carbon reacts with 1 mole of O2 to form one mole of CO2. Amount of $CO_{2}$ produced = 44 g (ii) 1 mole of carbon...
Determine the empirical formula of an oxide of iron, which has 69.9% iron and 30.1% dioxygen by mass.
Percentage of Fe by mass = 69.9% [As said previously] By mass, 30.1 percent of O2 is present. [As said previously] Relative moles of Fe in iron oxide: = (69.9)x(55.85}/55.8569.9 = 1.25 Relative...
Calculate the mass per cent of different elements present in sodium sulphate 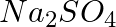
Now for Na2SO4. Molar mass of Na2SO4 = [(2 x 23.0) + (32.066) + 4(16.00)] =142.066 g Therefore, mass percent of the sodium element: = 32.379 = 32.4% Mass percent of the sulphur element: = 22.57...
Calculate the molar mass of the following: (i) 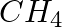 (ii)
(ii)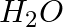 (iii)
(iii)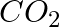
(i)Molecular weight of methane= (1 x Atomic weight of carbon) + (4 x Atomic weight of hydrogen) = [1(12.011 u) +4 (1.008u)] = 12.011u + 4.032 u = 16.043 u (ii)Molecular weight of water= (2 x...
Assertion (A): Among isomeric pentanes, 2, 2-dimethylpentane has the highest boiling point. Reason (R): Branching does not affect the boiling point. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (iii) is correct. Explanation: The lowest boiling point among isomeric pentanes is of 2,2-dimethylpentane, and further on branching, its boiling point decreases
Assertion (A): Nitration of benzene with nitric acid requires the use of concentrated sulphuric acid. Reason (R): The mixture of concentrated sulphuric acid and concentrated nitric acid produces the electrophile, NO2+. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (i) is correct Explanation: In nitration, benzene is treated with a nitrating mixture, which consists of conc. $HNO_3$ and $H_2SO_4$, and $H_2SO_4$ aids in the production of $NO_2 +$....
Assertion (A): Toluene on Friedel Crafts methylation gives o– and p–xylene. Reason (R): CH3-group bonded to benzene ring increases electron density at o– and p– position. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (i) is correct
Assertion (A): The compound cyclooctane has the following structural formula: It is cyclic and has conjugated 8π-electron system but it is not an aromatic compound.
Reason (R) : (4n + 2) π electrons rule does not hold good and the ring is not planar.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Option (i) is correct. Compounds with the following features are aromatic: planarity and complete delocalization of the electrons in the ring. The ring contains (4n+2) electrons, where n is an...
Match the reactions given in Column I with the reaction types in Column II.
(i) is d (ii) is a (iii) is b (iv) is c
Match the following reactants in Column I with the corresponding reaction products in Column II.
(i) is d (ii) is c (iii) is b (iv) is a
Match the hydrocarbons in Column I with the boiling points given in Column II.
(i) is b (ii) is c (iii) is a
Match the reagent from Column I which on reaction with CH3 —CH=CH2 gives some product given in Column II as per the codes given below :
(i) is d (ii) is a (iii) is e (iv) is c (v) is b
Suggest a route to prepare ethyl hydrogen sulphate (CH3–CH2–OSO2—OH) starting from ethanol (C2H5OH)
At about 140°C, ethanol is processed with sulphuric acid to produce hydrogen sulphate. The response is
Which of the following compounds are aromatic according to Huckel’s rule?
Huckel's rule states that it must meet the (4n+) rule. Aromatic compounds include B, C, D, and F.
The ring systems having the following characteristics are aromatic. (i) Planar ring containing conjugated π bonds. (ii) Complete delocalisation of the π−electrons in-ring system i.e. each atom in the ring has unhybridised p-orbital, and (iii) Presence of (4n+2)−electrons in the ring where n is an integer (n = 0, 1, 2,………..) [Huckel rule]. Using this information classifies the following compounds as aromatic/non-aromatic.
Aromatic compounds: A, E and F Non-Aromatic : B, C, D and G
An alkane C8H18 is obtained as the only product on subjecting a primary alkyl halide to Wurtz reaction. On monobromination, this alkane yields a single isomer of a tertiary bromide. Write the structure of alkane and the tertiary bromide.
Write hydrocarbon radicals that can be formed as intermediates during monochlorination of 2-methylpropane? Which of them is more stable? Give reasons.
In comparison to the 1° free radical, the 3° free radical is stabilised by 9 hyperconjugation structures, giving it greater stability.
Write the structures and names of products obtained in the reactions of sodium with a mixture of 1-iodo-2-methylpropane and 2-iodopropane.
When a combination of 1-iodo-2-methylpropane and 2-iodopropane is treated with salt, it produces three compounds as a result of intermolecular and intramolecular reactions:
The relative reactivity of 1°, 2°, 3° hydrogen’s towards chlorination is 1: 3.8: 5. Calculate the percentages of all mono-chlorinated products obtained from 2-methyl butane.
Number of hydrogen reactivity = number of mono-chlorinated compounds 1° H = 9 1 = 9 mono-chlorinated products 2° H = 2 3.8 = 7.6 mono-chlorinated products 3° H = 1 5 = 5 mono-chlorinated products...
Nucleophiles and electrophiles are reaction intermediates having electron-rich and electron-deficient centres’ respectively. Hence, they tend to attack electron-deficient and electron-rich centres respectively. Classify the following species as electrophiles and nucleophiles.
Nucleophiles I (vi), (vii), and (viii) Electrophiles are (ii), (iii), (iv), and (v).
Predict the major product (s) of the following reactions and explain their formation.
Step 1: Peroxide homolysis to produce free radicals Step 2: Bromine free radical formation ∙C6H5 + H-BR → C6H6 + Br Step 3: hydrogen bromide reacts with an alkyl radical.∙
Suggest a route for the preparation of nitrobenzene starting from acetylene?
I We can cycle acetylene using the intermolecular condensation technique and then treat it at high temperatures in a red hot iron tube. (ii) After being converted to benzene, the aliphatic molecule...
Why does the presence of a nitro group make the benzene ring less reactive in comparison to the unsubstituted benzene ring? Explain.
A nitrogen atom is linked to two extremely electronegative oxygen atoms in the Nitro group. This causes a net reduction in electron density around the nitrogen atom, giving nitrogen a positive charge.
Despite their – I effect, halogens are o- and p-directing in halo-arenes. Explain.
Halogens have a ns2p5 outer configuration, indicating that they may take one electron and are near to completing their octate; halogens have a significant affinity for attracting one electron,...
Arrange the following set of compounds in the order of their decreasing relative reactivity with an electrophile. Give reason.
How will you convert benzene into (i) p – nitrobromobenzene (ii) m – nitrobromobenzene
I Bromine undergoes electrophilic substitution with Br2 in the presence of anhydrous FeBr3 to generate bromobenzene. We get a p-nitrobromobenzene after treating it with conc. HNO3 and conc. H2SO4 at...
What will be the product obtained as a result of the following reaction and why?
Friedel Crafts alkylation with a Lewis acid is demonstrated in this process. The development of the carbocation will occur initially, followed by the production of a more stable secondary...
The intermediate carbocation formed in the reactions of HI, HBr and HCl with propane is the same and the bond energy of HCl, HBr and HI is 430.5 kJ mol-1, 363.7 kJ mol-1 and 296.8 kJ mol-1 respectively. What will be the order of reactivity of these halogen acids?
The ascending order of reactivity is HI >HBr>HCl. The rising order of halogen reactivity corresponds to the rise in bond energy.
Draw Newman and Sawhorse projections for the eclipsed and staggered conformations of ethane. Which of these conformations is more stable and why?
Because there is less C – H bond pair repulsion and the atoms are at their furthest distance from one other, the staggered arrangement is more stable than an eclipse.
Rotation around carbon-carbon single bond of ethane is not completely free. Justify the statement.
In ethane, the single bond is a – bond, which is a coaxial overlap of orbitals that allows the C C bond to rotate on its axis. However, due to the torsional strain that the bond receives as a result...
Alkynes on reduction with sodium in liquid ammonia form trans alkenes. Will the butene thus formed on the reduction of the 2-butyne show the geometrical isomerism?
The negative charge that has generated on one carbon attacks the proton from NH3 and causes another sodium atom to lose its electron, causing the atom to create a second negative charge. The second...
Why do alkenes prefer to undergo electrophilic addition reaction while arenes prefer electrophilic substitution reactions? Explain.
To produce a more stable saturated product, alkenes undergo an addition reaction. Hybridization shifts from sp2 to sp3 in this reaction. A substitution process maintains the resonance stability of...
The molecules having dipole moment are __________.(i) 2,2-Dimethylpropane (ii) trans-Pent-2-ene (iii) cis-Hex-3-ene (iv) 2, 2, 3, 3 – Tetramethylbutane.
Option (ii) and (iii) are the answers
Four structures are given in options (i) to (iv). Examine them and select the aromatic structures.
Option (i) and (iii) are the answers.
Which of the following is correct?
Option (i) and (iii) are the answers.
In an electrophilic substitution reaction of nitrobenzene, the presence of nitro group ________. (i) deactivates the ring by an inductive effect. (ii) activates the ring by an inductive effect. (iii) decreases the charge density at ortho and para position of the ring relative to meta position by resonance. (iv) increases the charge density at meta position relative to the ortho and para positions of the ring by resonance
Option (i) and (iii) are the answers.
For an electrophilic substitution reaction, the presence of a halogen atom in the benzene ring _______. (i) deactivates the ring by the inductive effect (ii) deactivates the ring by resonance (iii) increases the charge density at ortho and para position relative to meta position by resonance (iv) directs the incoming electrophile to meta position by increasing the charge density relative to ortho and para position.
The solutions are options (i) and (iii).
Which are the correct IUPAC names of the following compound?;(i) 5 – (2′, 2′–Dimethylpropyl)-decane (ii) 4 – Butyl – 2,2– dimethylnonane (iii) 2,2– Dimethyl – 4– pentyloctane (iv) 5 – neo-Pentyldecane
Option (i) and (iv) are the answers.
Which are the correct IUPAC names of the following compound?;(i) 5– Butyl – 4– isopropyldecane (ii) 5– Ethyl – 4– propyldecane (iii) 5– sec-Butyl – 4– iso-propyldecane (iv) 4–(1-methoxymethyl)– 5 – (1-methyl propyl)-decane
The solutions are options (iii) and (iv).
Which of the following alkenes on ozonolysis give a mixture of ketones only?
The solutions are options (iii) and (iv).
In the following questions, two or more options may be correct. Some oxidation reactions of methane are given below. Which of them is/are controlled oxidation reactions? (i) CH4 (g) + 2O2 (g)→ CO2 (g) + 2H2O (l) (ii) CH4 (g) + O2 (g) → C (s) + 2H2O (l) (iii) CH4 (g) + O2 (g) →(Mo O2 3) HCHO + H2O (iv) 2CH4 (g) + O2 (g) → (Cu/523/100 atm) 2CH3OH
The solutions are options (iii) and (iv).
Which of the following reactions of methane is incomplete combustion:
Option (iii) is the answer.
Arrange the following alkyl halides in decreasing order of the rate of β– elimination reaction with alcoholic KOH.(i) A > B > C (ii) C > B > A (iii) B > C > A (iv) A > C > B
The solution is option (iv).
Arrange the following carbanions in order of their decreasing stability. (A) H3C – C ≡ C– (B) H – C ≡ C– (C) H3C-CH–2 (i) A > B > C (ii) B > A > C (iii) C > B > A (iv) C > A > B
The solution is option (ii).
Arrange the following hydrogen halides in order of their decreasing reactivity with propane. (i) HCl > HBr > HI (ii) HBr > HI > HCl (iii) HI > HBr > HCl (iv) HCl > HI > HBr
The solution is Option (iii)
Which of the following will not show geometrical isomerism?
Option (iv) is the answer.
The addition of HBr to 1-butene gives a mixture of products A, B and C;The mixture consists of (i) A and B as major and C as minor products (ii) B as major, A and C as minor products (iii) B as minor, A and C as major products (iv) A and B as minor and C as major products
Option I is the correct response.
The correct IUPAC name of the following alkane is ;(i) 3,6 – Diethyl – 2 – methyl octane (ii) 5 – Isopropyl – 3 – ethyloctane (iii) 3 – Ethyl – 5 – isopropyloctane (iv) 3 – Isopropyl – 6 – ethyloctane
Option I is the correct response.
The increasing order of reduction of alkyl halides with zinc and dilute HCl is (i) R–Cl < R–I < R–Br ii) R–Cl < R–Br < R–I (iii) R–I < R–Br < R–Cl (iv) R–Br < R–I < R–Cl
Option (ii) is the correct response.
Arrange the halogens F2 , Cl2 , Br2 , I2 , in order of their increasing reactivity with alkanes. (i) I2 < Br2 < Cl2 < F2 (ii) Br2 < Cl2 < F2 < I2 (iii) F2 < Cl2 < Br2 < I2 (iv) Br2 < I2< Cl2 < F2
Option (i) is the solution
Arrange the following in decreasing order of their boiling points. (A) n–butane (B) 2–methylbutane (C) n-pentane (D) 2,2–dimethylpropane (i) A > B > C > D (ii) B > C > D > A (iii) D > C > B > A (iv) C > B > D > A
the answer is Option (iv)
Assertion (A): If BOD level of water in a reservoir is less than 5 ppm it is highly polluted. Reason (R): High biological oxygen demand means a low activity of bacteria in water. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (iii) is the answer.
Assertion (A): Excessive use of chlorinated synthetic pesticides causes soil and water pollution. Reason (R): Such pesticides are non-biodegradable. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct
Option (i) is the answer.
Assertion (A): Ozone is destroyed by solar radiation in the upper stratosphere. Reason (R): Thinning of the ozone layer allows excessive UV radiations to reach the surface of the earth. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (iv) is the answer.
Assertion (A): Carbon dioxide is one of the important greenhouse gases. Reason (R): It is largely produced by respiratory function of animals and plants. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (ii) is the answer.
Assertion (A): Photochemical smog is oxidising in nature. Reason (R): Photochemical smog contains NO2 and O3, which are formed during the sequence of reactions. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (i) is correct.
Assertion (A): The pH of acid rain is less than 5.6. Reason (R): Carbon dioxide present in the atmosphere dissolves in rain water and forms carbonic acid. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
Option (ii) is correct.
Assertion (A): Greenhouse effect was observed in houses used to grow plants and these are made of green glass. Reason (R): Greenhouse name has been given because glasshouses are made of green glass. (i) Both A and R are correct and R is the correct explanation of A. (ii) Both A and R are correct but R is not the correct explanation of A. (iii) Both A and R are not correct. (iv) A is not correct but R is correct.
correct is Option (iii)
Match the pollutants given in Column I with their effects given in Column II.
(i) are a and d (ii) is c (iii) is a (iv) is b
Match the activity given in Column I with the type of pollution created by it given in Column II.
(i) is e (ii) is d (iii) is a (iv) is b (v) is c
Match the pollutant(s) in Column I with the effect(s) in Column II.
(i) is d (ii) is e (iii) is a (iv) is c (v) is b
Match the terms given in Column I with the compounds given in Column II.
(i) are c and d (ii) are e and d (iii) is b (iv) is a
A person was using water supplied by the Municipality. Due to the shortage of water, he started using underground water. He felt a laxative effect. What could be the cause?
The laxative effect is the loosening of faeces and increased bowel movement induced by laxative drugs. Laxatives in high dosages might induce diarrhoea. This might be due to an excessive amount of...
Ozone is a gas heavier than air. Why does the ozone layer not settle down near the earth?
The ozone layer is heavier than oxygen and is thermodynamically unstable, yet the interaction between ozone production and dissociation has a dynamic equilibrium. As a result, they avoid settling...
How is ozone produced in stratosphere?
The action of ultraviolet light on atmospheric oxygen forms the ozone layer in the stratosphere. UV rays cause molecular oxygen to divide or dissociate into two oxygen atoms. O 2 (g) → O (g) + O (g)...
From where does ozone come in the photochemical smog?
Nitrogen oxides, volatile organic compounds (VOC), ozone, and PAN (peroxyacetyl nitrate) are all components of photochemical smog. In the presence of sunshine, nitrogen oxide in the smog dissociates...
Oxidation of Sulphur dioxide into Sulphur trioxide in the absence of a catalyst is a slow process but this oxidation occurs easily in the atmosphere. Explain how does this happen. Give chemical reactions for the conversion of SO2 into SO3.
The uncatalyzed oxidation of sulphur dioxide to sulphur trioxide is sluggish, but it may be oxidised quickly in the presence of a catalyst in the environment. There are substances in the atmosphere...
A factory was started near a village. Suddenly villagers started feeling the presence of irritating vapours in the village and cases of headache, chest pain, cough, dryness of throat and breathing problems increased. Villagers blamed the emissions from the chimney of the factory for such problems. Explain what could have happened. Give chemical reactions for the support of your explanation.
This sort of breathing difficulty can be caused by toxic nitrogen and sulphur oxides in the environment. These oxides are produced in industry by the oxidation of fossil fuels such as coal. N2 (g) +...
Why does water cover with excessive algal growth become polluted?
Algae breakdown by bacteria continually generates a nasty odour, and algae contaminate and make water unpleasant. The quantity of dissolved oxygen in the water drops as well, which might cause...
What is the importance of measuring BOD of a water body?
The quantity of oxygen required by bacteria to breakdown organic materials is known as biochemical oxygen demand (BOD). The higher the BOD of water, the more contaminated it is. The water with a...
What are the sources of dissolved oxygen in water?
Water absorbs oxygen from the atmosphere when it comes into direct contact with atmospheric air. During the day, green aquatic plants photosynthesise. Photosynthesis does not occur at night, but the...
What are biodegradable and non-biodegradable pollutants?
Biodegradable pollutants are ones that can be degraded by bacteria or other natural elements such as fruits, sewage, and so on. Bacteria have a difficult time decomposing non-biodegradable...
During an educational trip, a student of botany saw a beautiful lake in a village. She collected many plants from that area. She noticed that villagers were washing clothes around the lake and at some places waste material from houses was destroying its beauty. After a few years, she visited the same lake again. She was surprised to find that the lake was covered with algae, the stinking smell was coming out and its water had become unusable. Can you explain the reason for this condition of the lake?
The dumping of household garbage in the lake can give nutrients for algae and aquatic plants to develop fast. The breakdown of these, which is aided by bacteria, can also result in a bad odour. The...
What could be the harmful effects of improper management of industrial and domestic solid waste in a city?
Inadequate industrial and household waste management in a city can result in significant harm to both living and non-living objects. 1. If household trash is not properly disposed of, sewage pipes...
Based on chemical reactions involved, explain how chlorofluorocarbons cause thinning of the ozone layer in the stratosphere
The action of ultraviolet radiation causes chlorofluorocarbons to dissociate, releasing the free chlorine radicle. U V radiations + CF2Cl2 + Cl + CFC Now that this Chlorine radicle has formed, it is...
Dissolved oxygen in water is very important for aquatic life. What processes are responsible for the reduction of dissolved oxygen in water?
The process of eutrophication is when the amount of dissolved oxygen in the water decreases. It's a process in which nutrient-rich water bodies support a dense plant population, which then kills...
Ozone is a toxic gas and is a strong oxidizing agent even then its presence in the stratosphere is very important. Explain what would happen if ozone from this region is completely removed?
When ozone is removed from the atmosphere, UV radiation comes into direct contact with living organisms, causing harm such as skin cancer and a variety of other dangerous illnesses. The ozone in the...
Acid rain is known to contain some acids. Name these acids and where from they come in the rain?
Acid rain is caused by human activities that release sulphur and nitrogen oxides into the atmosphere. 2H2SO4 = 2SO2 (g) + O2 (g) + 2H2O (l) (aq) 4HNO3 (aq) = 4NO2 (g) + O2 (g) + 2H2O (l). Acid rain...
Greenhouse effect leads to global warming. Which substances are responsible for the greenhouse effect?
Greenhouse gases such as carbon dioxide, methane, ozone, chlorofluorocarbon compounds (CFCs), and water vapour are all responsible for the greenhouse effect, which leads to global warming.
The consequences of global warming may be _________. (i) increase in average temperature of the earth (ii) melting of Himalayan Glaciers. (iii) increased biochemical oxygen demand. (iv) eutrophication.Solution:
The solutions are (i), (ii).
The acids present in acid rain are _________. (i) Peroxyacetylnitrate (ii) H2CO3 (iii) HNO3 (iv) H2SO4
The solutions are (ii), (iii), and (iv).
Phosphate containing fertilisers cause water pollution. Addition of such compounds in water bodies causes __________. (i) enhanced growth of algae. (ii) the decrease in the amount of dissolved oxygen in the water. (iii) deposition of calcium phosphate. (iv) increase in the fish population.
The solutions are option (i) and (ii)
Which of the following conditions shows the polluted environment. (i) a pH of rainwater is 5.6. (ii) amount of carbon dioxide in the atmosphere is 0.03%. (iii) biochemical oxygen demand 10 ppm. (iv) eutrophication.
The solutions are option (iii) and (iv)
Which of the following practices will not come under green chemistry? (i) If possible, making use of soap made of vegetable oils instead of using synthetic detergents. (ii) Using H2O2 for bleaching purpose instead of using chlorine-based bleaching agents. (iii) Using a bicycle for travelling small distances instead of using petrol/diesel-based vehicles. (iv) Using plastic cans for neatly storing substances.
The solution is option (iV).
Which of the following statements is correct? (i) The ozone hole is a hole formed in the stratosphere from which ozone oozes out. (ii) The ozone hole is a hole formed in the troposphere from which ozone oozes out. (iii) The ozone hole is thinning of the ozone layer of the stratosphere at some places. (iv) Ozone hole means vanishing of ozone layer around the earth completely.
The solution is option (iii).
The pollutants which come directly in the air from sources are called primary pollutants. Primary pollutants are sometimes converted into secondary pollutants. Which of the following belongs to secondary air pollutants? (i) CO (ii) Hydrocarbon (iii) Peroxyacetyl nitrate (iv) NO
The solution is option (iii).
Dinitrogen and dioxygen are main constituents of air but these do not react with each other to form oxides of nitrogen because of _________. (i) the reaction is endothermic and requires very high temperature. (ii) the reaction can be initiated only in the presence of a catalyst. (iii) oxides of nitrogen are unstable. (iv) N2and O2 are unreactive.
The solution is option (i).
The gaseous envelope around the earth is known as the atmosphere. The lowest the layer of this is extended up to 10 km from sea level, this layer is _________. (i) Stratosphere (ii) Troposphere (iii) Mesosphere (iv) Hydrosphere
The solution is option (ii).
Which of the following statements about photochemical smog is wrong? (i) It has a high concentration of oxidising agents. (ii) It has a low concentration of the oxidising agent. (iii) It can be controlled by controlling the release of NO2 , hydrocarbons, ozone etc. (iv) Plantation of some plants like pinus helps in controlling photochemical smog.
The solution is option (ii).
Sewage containing organic waste should not be disposed of in water bodies because it causes major water pollution. Fishes in such polluted water die because of (i) Large number of mosquitoes. (ii) Increase in the amount of dissolved oxygen. (iii) The decrease in the amount of dissolved oxygen in the water. (iv) Clogging of gills by mud.
The solution is option (iii).
Which of the following statements is wrong? (i) Ozone is not responsible for the greenhouse effect. (ii) Ozone can oxidise sulphur dioxide present in the atmosphere to sulphur trioxide. (iii) The ozone hole is thinning of ozone layer present in the stratosphere. (iv) Ozone is produced in the upper stratosphere by the action of UV rays on oxygen.
The solution is option (i).
Biochemical Oxygen Demand, (BOD) is a measure of organic material present in water. BOD value less than 5 ppm indicates a water sample to be __________. (i) rich in dissolved oxygen. (ii) poor in dissolved oxygen. (iii) highly polluted. (iv) not suitable for aquatic life.
The solution is option (i).
Which of the following statements is not true about classical smog? (i) Its main components are produced by the action of sunlight on emissions of automobiles and factories. (ii) Produced in a cold and humid climate. (iii) It contains compounds of reducing nature. (iv) It contains smoke, fog and sulphur dioxide.
The solution is option (i).
Photochemical smog occurs in a warm, dry and sunny climate. One of the following is not amongst the components of photochemical smog, identify it. (i) NO2 (ii) O3 (iii) SO2 (iv) Unsaturated hydrocarbon
The solution is option (iii).
Which of the following gases is not a greenhouse gas? (i) CO (ii) O3 (iii) CH4 (iv) H2O vapour
Option (i) is the correct answer
A bar magnet of magnetic moment m and moment of inertia I is cut into two equal pieces, perpendicular to length. Let T be the period of oscillations of the original magnet about an axis through the midpoint, perpendicular to the length, in a magnetic field B. What would be the similar period T’ for each piece?
T stands for the time period. The moment of inertia is me. The magnet's mass is m. B stands for magnetic field. T = 2I/MB M' = M/2 magnetic dipole moment T' = T/2 is the time period.
Suppose we want to verify the analogy between electrostatic and magnetostatic by an explicit experiment. Consider the motion of i) electric dipole p in an electrostatic field E and ii) magnetic dipole m in a magnetic field B. Write down a set of conditions on E, B, p, m so that the two movements are verified to be identical.
pE sin θ = μB sin θ pE = μB E = cB pcB = μB p = μ/c
Three identical bar magnets are riveted together at the centre in the same plane as shown in the figure. This system is placed at rest in a slowly varying magnetic field. It is found that the system of magnets does not show any motion. The north-south poles of one magnet is shown in the figure. Determine the poles of the remaining two.
As the system is in equilibrium, the net torque and net force will be equal to zero.
Verify the Gauss’s law for magnetic field of a point dipole of dipole moment m at the origin for the surface which is a sphere of radius R.
P is the point at a distance r from O and OP, then the magnetic field is given as: dS is the elementary area of the surface P, then dS = r2 (r2 sin θ d θr) Solving the above we...
A ball of superconducting material is dipped in liquid nitrogen and placed near a bar magnet. i) In which direction will it move? ii) What will be the direction of it’s magnetic moment?
I The superconducting material will repel the bar magnet. ii) The magnetic moment will be directed from left to right.
From molecular view point, discuss the temperature dependence of susceptibility for diamagnetism, paramagnetism, and ferromagnetism.
The temperature has little effect on the temperature dependence of susceptibility for a diamagnetism. The temperature affects the temperature dependence of susceptibility for paramagnetism and...
Explain quantitatively the order of magnitude difference between the diamagnetic susceptibility of N2 and Cu.
nitrogen Density = 28 g/ 22400 cc copper Density = 8 g/ 22400 cc Ratio = 16 × 10-4 Diamagnetic susceptibility = density of nitrogen/density of copper = 1.6 × 10-4
A permanent magnet in the shape of a thin cylinder of length 10 cm has M = 106 A/m. Calculate the magnetisation current Im.
Intensity = 106 A/m l = 0.1 m M = IM/l IM = Ml = 105 A
A proton has spin and magnetic moment just like an electron. Why then its effect is neglected in magnetism of materials?
A comparison of a proton's and an electron's spinning is made by comparing their magnetic dipole moment, which is given as μp = eh/4πmp μe = eh/4πme μp/μe = me/mp = 1/1837 >> 1 μp << μe...
Essential difference between electrostatic shielding by a conducting shell and magneto static shielding is due to a) electrostatic field lines can end on charges and conductors have free charges b) lines of B can also end but conductors cannot end them c) lines of B cannot end on any material and perfect shielding is not possible d) shells of high permeability materials can be used to divert lines of B from the interior region
a) electrostatic field lines can end on charges and conductors have free charges c) lines of B cannot end on any material and perfect shielding is not possible d) shells of high permeability...
A long solenoid has 1000 turns per meter and carries a current of 1 A. It has a soft iron core of μr = 1000. The core is heated beyond the Curie temperature Tc a) the H field in the solenoid is unchanged but the B field decreases drastically b) the H and B fields in the solenoid are nearly unchanged c) the magnetisation in the core reverses direction d) the magnetisation in the core diminishes by a factor of about 108
a) the H field in the solenoid is unchanged but the B field decreases drastically d) the magnetisation in the core diminishes by a factor of about 108
The primary origin(s) of magnetism lies in a) atomic currents b) Pauli exclusion principle c) polar nature of molecules d) intrinsic spin of electron
a) atomic currents d) intrinsic spin of electron
S is the surface of a lump of magnetic material a) lines of B are necessarily continuous across S b) some lines of B must be discontinuous across S c) lines of H are necessarily continuous across S d) lines of H cannot all be continuous across S
a) lines of B are necessarily continuous across S d) lines of H cannot all be continuous across S
A paramagnetic sample shows a net magnetisation of 8 Am-1 when placed in an external magnetic field of 0.6T at a temperature of 4K. When the same sample is placed in an external magnetic field of 0.2T at a temperature of 16K, the magnetisation will be a) 32/3 Am-1 b) 2/3 Am-1 c) 6 Am-1 d) 2.4 Am-1
b) 2/3 Am-1
Consider the two idealized systems: i) a parallel plate capacitor with large plates and small separation and ii) a long solenoid of length L >> R, the radius of the cross-section. In i) E is ideally treated as a constant between plates and zero outside. In ii) magnetic field is constant inside the solenoid and zero outside. These idealised assumptions, however, contradict fundamental laws as below: a) case (i) contradicts Gauss’s law for electrostatic fields b) case (ii) contradicts Gauss’s law for magnetic fields c) case (i) agrees withd) case (ii) contradicts
b) In the case of magnetic fields, instance (ii) violates Gauss's law.
In a permanent magnet at room temperature a) magnetic moment of each molecule is zero b) the individual molecules have a non-zero magnetic moment which is all perfectly aligned c) domains are partially aligned d) domains are all perfectly aligned
d) all of the domains are exactly matched
The magnetic field of the earth can be modelled by that of a point dipole placed at the centre of the earth. The dipole axis makes an angle of 11.3o with the axis of the earth. At Mumbai, declination is nearly zero. Then, a) the declination varies between 11.3o W to 11.3o E b) the least declination is 0o c) the plane defined by dipole axis and the earth axis passes through Greenwich d) declination average over the earth must be always negative
a) The declination ranges from 11.3 degrees West to 11.3 degrees East.
A toroid of n turns, mean radius R and cross-sectional radius a carries current I. It is placed on a horizontal table taken as an x-y plane. Its magnetic moment m a) is non-zero and points in the z-direction by symmetry b) points along the axis of the toroid c) is zero, otherwise, there would be a field falling as 1/r3 at large distances outside the toroid d) is pointing radially outwards
c) is zero; otherwise, a field dropping as 1/r3 at great distances outside the toroid would exist.
A long solenoid ‘S’ has ‘n’ turns per meter, with diameter ‘a’. At the centre of this coil, we place a smaller coil of ‘N’ turns and diameter ‘b’ (where b < a). If the current in the solenoid increases linearly, with time, what is the induced emf appearing in the smaller coil. Plot graph showing nature of variation in emf, if current varies as a function of mt2 + C.
The solenoid's changing magnetic field is represented as: onI = B1(t) (t) The second coil's magnetic flux is 2 = onI(t).b2 As a result of the solenoid's changing magnetic field, the induced emf in...
A metallic ring of mass m and radius l (ring being horizontal) is falling under gravity in a region having a magnetic field. If z is the vertical direction, the z-component of the magnetic field is Bz = Bo (1+λ z). If R is the resistance of the ring and if the ring falls with a velocity v, find the energy lost in the resistance. If the ring has reached a constant velocity, use the conservation of energy to determine v in terms of m, B, λ and acceleration due to gravity g.
v = mgR/B02π2λ2l4
Find the current in the sliding rod AB (resistance = R). B is constant and is out of the paper. Parallel wires have no resistance. v is constant. Switch S is closed at time t = 0.
The angle between A and B = 0o Therefore, the emf is Bvd. -LdI(t)/dt + Bvd = IR LdI(t)/dt + RI (t) = Bvd Solving the equation, we get I = Bvd/R [1-e-Rt/2]
Find the current in the sliding rod AB (resistance = R). B is constant and is out of the paper. Parallel wires have no resistance. v is constant. Switch S is closed at time t = 0.
It = /R is the current induced in the loop. BA = 1/R.d/dt It = 1/R.d/dt It = 1/R.d/d vBd/R = it The angle formed by B and A is equal to zero. At t = o, the switch S is closed. Q(t) = Cv is the...
A rod of mass m and resistance R slides smoothly over two parallel perfectly conducting wires kept sloping at an angle θ with respect to the horizontal. The circuit is closed through a perfect conductor at the top. There is a constant magnetic field B along the vertical direction. If the rod is initially at rest, find the velocity of the rod as a function of time.
The angle formed by B and PQ is 90 dϕ = B.dA dϕ = B v d cos θ -ε = B v d cos θ I = -Bvd/R cos θ Using Newton's second law to solve the preceding problem, we get v as v = α g sin θ [1 – e...
A magnetic field B is confined to a region r a ≤ and points out of the paper (the z-axis), r = 0 being the centre of the circular region. A charged ring (charge = Q) of radius b, b > a and mass m lies in the x-y plane with its centre at the origin. The ring is free to rotate and is at rest. The magnetic field is brought to zero in time ∆t. Find the angular velocity ω of the ring after the field vanishes.
When the magnetic field is lowered in t, the magnetic flux across the conducting ring drops to zero from its maximum. E2b = induced emf According to Faraday's law of emf, The induced emf is equal to...
A rectangular loop of wire ABCD is kept close to an infinitely long wire carrying a current II ( ) t = o (1– /t T ) for 0 ≤ ≤ t T and I (0) = 0 for t > T. Find the total charge passing through a given point in the loop, in time T. The resistance of the loop is R.
If the instantaneous current is t, then I(t) = 1/R d/dt I(t) If q is the charge that passes during time t, dQ/dt = I(t) 1/R d/dt = dQ/dt When we integrate the equation, we obtain Q = 0L1L2/2R log...
Consider an infinitely long wire carrying a current I (t ), with dI dt =λ= constant. Find the current produced in the rectangular loop of wire ABCD if its resistance is R.
The strip has a width of dr and a length of l, and it is located inside the rectangular box at a distance of r from the current-carrying conductor's surface. The magnetic field along the length of...
i) When the position of the rotating conductor is assumed to be at the time interval t = 0 to t = π/4ꞷ We get current I = 1/2 Bl2ꞷ/λl sec2 ꞷt cos ꞷt = Blꞷ/2λ cos ꞷt ii) When the position of the rotating conductor is at time interval π/4ꞷ < t < 3π/4ꞷ We get current I = 1/2 Blꞷ/λ sin ꞷt iii) When the position of the rotating conductor is at time interval 3π/4ꞷ < t < π/ꞷ We get current I = 1/2 Blꞷ/ λ sin ꞷt
I If the spinning conductor's location is considered to be in the time period t = 0 to t = 4 We obtain current I = 1/2 Bl2/l sec2 t cos t = Bl/2 cos t = Bl/2 cos t = Bl/2 cos t = Bl/2 cos t = Bl/2...
A conducting wire XY of mass m and negligible resistance slides smoothly on two parallel conducting wires. The closed-circuit has a resistance R due to AC. AB and CD are perfect conductors. There is a magnetic field B = B t(k ). (i) Write down the equation for the acceleration of the wire XY. (ii) If B is independent of time, obtain v(t) , assuming v (0) = u0. (iii) For (b), show that the decrease in kinetic energy of XY equals the heat lost in R.
m = B.A = BA cos m The area vector is A, while the magnetic field vector is B. e1 = -dB(t)/dt lx e1 e2 = B(t) lv (t) The total emf in the circuit is equal to the emf owing to field change plus the...
A magnetic field B = Bo sin ωt k covers a large region where a wire AB slides smoothly over two parallel conductors separated by a distance d. The wires are in the x-y plane. The wire AB (of length d) has resistance R and the parallel wires have negligible resistance. If AB is moving with velocity v, what is the current in the circuit? What is the force needed to keep the wire moving at constant velocity?
Allow wire AB to travel with velocity v at time t = 0. x(t) = vt at time t AB = e1 = Blv Motional emf across (Bo sin t)vd = e1 (-j) d(B)/dt = e2 e2 = -B0 cos tx (t)d e2 = -B0 cos tx (t)d e2 = -B0...
There are two coils A and B separated by some distance. If a current of 2 A flows through A, a magnetic flux of 10-2 Wb passes through B (no current through B). If no current passes through A and a current of 1 A passes through B, what is the flux through A?
The current flowing through the coil is denoted by Ia. Mutual induction between A and B is known as Mab. The number of turns in coil A is Na. The number of turns in coil B is Nb. an is the flux...
A (current vs time) graph of the current passing through a solenoid is shown in Fig 6.9. For which time is the back electromotive force (u) a maximum. If the back emf at t = 3s is e, find the back emf at t = 7 s, 15s and 40s. OA, AB and BC are straight line segments.
We may deduce from the graph that when the rate of change of magnetic flux reaches its highest, the electromagnetic force, which is proportional to the rate of change of current, reaches its...
Find the current in the wire for the configuration. Wire PQ has negligible resistance. B, the magnetic field is coming out of the paper. θ is a fixed angle made by PQ travelling smoothly over two conducting parallel wires separated by a distance d.
F is the force acting on PQ's free charge particle. The motional emf is calculated by multiplying E along the PQ by the effective length of the PQ. As a result, the induced current will be vBd/R,...
Consider a closed loop C in a magnetic field such that the flux passing through the loop is defined by choosing a surface whose edge coincides with the loop and using the formula φ= B1dA1 + B2dA2 + …. Now if we chose two different surfaces S1 and S2 having C as their edge, would we get the same answer for flux. Justify your answer.
The magnetic flux lines that pass through are identical to those that flow through the surface. The magnetic field lines in an area A with magnetic flux B are represented by = B1dA1 + B2dA2. As a...
A magnetic field in a certain region is given by B = Bo cos ωt k and a coil of radius a with resistance R is placed in the x-y plane with its centre at the origin in the magnetic field. Find the magnitude and the direction of the current at (a, 0, 0) at t =π ω /2, t =π /2ω and t =3π/ω
The magnetic field is directed along the z-axis. B.A = BA cos = B.A cos = B.A cos = B.A cos = B.A cos Using the electromagnetic induction law of Faraday, R sin t = Boa2/R sin t = I = Boa2/R sin t =...
Consider a metallic pipe with an inner radius of 1 cm. If a cylindrical bar magnet of radius 0.8cm is dropped through the pipe, it takes more time to come down than it takes for a similar unmagnetised cylindrical iron bar dropped through the metallic pipe. Explain.
The magnetic flux across the pipe changes when a cylindrical bar magnet with a radius of 0.8 cm is dropped through it, causing eddy currents to form. The existence of eddy current causes the magnet...
Consider a metal ring kept (supported by a cardboard) on top of a fixed solenoid carrying a current I. The centre of the ring coincides with the axis of the solenoid. If the current in the solenoid is switched off, what will happen to the ring?
We already know that current was flowing through the solenoid, causing it to act like a magnet with the S pole on the upper side. As a result, the ring has no induced current. When the current is...
Consider a metal ring kept on top of a fixed solenoid such that the centre of the ring coincides with the axis of the solenoid. If the current is suddenly switched on, the metal ring jumps up. Explain.
The metal ring leaps up when the current is quickly turned on because the magnetic flux across the ring is enhanced.
A solenoid is connected to a battery so that a steady current flows through it. If an iron core is inserted into the solenoid, will the current increase or decrease? Explain.
The magnetic flux rises when an iron core is introduced into the solenoid. According to Lenz's law, as the flux increases, the current flow through the coil decreases.
A wire in the form of a tightly wound solenoid is connected to a DC source, and carries a current. If the coil is stretched so that there are gaps between successive elements of the spiral coil, will the current increase or decrease? Explain.
When the spiral coil is extended and there are gaps between consecutive components, the current rises. Reactance must drop in order for current to rise.
Consider a magnet surrounded by a wire with an on/off switch S. If the switch is thrown from the off position (open circuit) to the on position (closed circuit), will a current flow in the circuit? Explain
There will be no current induced since there is no change in the magnet or the circuit area. In addition, there has been no change in the angle
A circular coil expands radially in a region of the magnetic field and no electromotive force is produced in the coil. This can be because (a) the magnetic field is constant. (b) the magnetic field is in the same plane as the circular coil and it may or may not vary. (c) the magnetic field has a perpendicular (to the plane of the coil) component whose magnitude is decreasing suitably. (d) there is a constant magnetic field in the perpendicular (to the plane of the coil) direction.
(b) the magnetic field is in the same plane as the circular coil and it may or may not vary. (c) the magnetic field has a perpendicular (to the plane of the coil) component whose magnitude is...
The mutual inductance M12 of coil 1 with respect to coil 2 (a) increases when they are brought nearer. (b) depends on the current passing through the coils. (c) increases when one of them is rotated about an axis. (d) is the same as M21 of coil 2 with respect to coil 1.
(a) increases when they are brought nearer. (d) is the same as M21 of coil 2 with respect to coil 1.
An e .m.f is produced in a coil, which is not connected to anexternal voltage source. This can be due to (a) the coil is in a time-varying magnetic field. (b) the coil moving in a time-varying magnetic field. (c) the coil moving in a constant magnetic field. (d) the coil is stationary in an external spatially varying magnetic field, which does not change with time.
(a) the coil is in a time-varying magnetic field. (b) the coil moving in a time-varying magnetic field. (c) the coil moving in a constant magnetic field.
A metal plate is getting heated. It can be because (a) a direct current is passing through the plate. (b) it is placed in a time-varying magnetic field. (c) it is placed in a space varying magnetic field, but does not vary with time. (d) a current (either direct or alternating) is passing through the plate.
(a) The plate is receiving a direct current. (b) it is exposed to a magnetic field that changes over time. (c) it is put in a magnetic field that varies in space but not in time.
The self-inductance L of a solenoid of length l and area of cross-section A, with a fixed number of turns N increases as (a) l and A increase. (b) l decreases and A increases. (c) l increases and A decreases. (d) both l and A decrease.
(b) l decreases and A increases.
Same as problem 4 except the coil A is made to rotate about a vertical axis. No current flows in B if A is at rest. The current in coil A, when the current in B (at t = 0) is counterclockwise and the coil A is as shown at this instant, t = 0, is (a) constant current clockwise. (b) varying current clockwise. (c) varying current counterclockwise. (d) constant current counterclockwise.
(a) clockwise steady current
There are two coils A and B. A current starts flowing in B as shown, when A is moved towards B and stops when A stops moving. The current in A is counterclockwise. B is kept stationary when A moves. We can infer that (a) there is a constant current in the clockwise direction in A. (b) there is a varying current in A. (c) there is no current in A. (d) there is a constant current in the counterclockwise direction in A.
(d) In A, there is a counterclockwise current that is constant.
A cylindrical bar magnet is rotated about its axis. A wire is connected from the axis and is made to touch the cylindrical surface through a contact. Then (a) a direct current flows in the ammeter A. (b) no current flows through the ammeter A. (c) an alternating sinusoidal current flows through the ammeter A with a time period T=2π/ω. (d) a time-varying non-sinusoidal current flows through the ammeter A.
(b) There is no current flowing through ammeter A.
A loop, made of straight edges has six corners at A(0,0,0), B(L,O,0) C(L,L,0), D(0,L,0) E(0,L,L) and F(0,0,L). A magnetic field B = Bo (i + k)T is present in the region. The flux passing through the loop ABCDEFA (in that order) is (a) Bo L2 Wb. (b) 2 Bo L2 Wb. (c) 2 Bo L2 Wb. (d) 4 Bo L2 Wb.
(b) 2 Bo L2 Wb. is the correct answer
A square of side L meters lies in the x-y plane in a region, where the magnetic field is given by B = Bo (2i + 3j + 4k) T where Bo is constant. The magnitude of flux passing through the square is (a) 2 Bo L2 Wb. (b) 3 Bo L2 Wb. (c) 4 Bo L2 Wb. (d) √29 B Lo2 Wb
(c) 4 Bo L2 Wb. is the correct answer
Match the ions given in Column I with their nature-given in Column II.
(i) is a, b and d (ii) is b (iii) is b (iv) are c and d
Match Column I with Column II.
(i) is c (ii) is e (iii) is a (iv) is b (v) is d
Match the terms mentioned in Column I with the terms in Column II.
(i) is c (ii) is f (iii) is b (iv) is a (v) is d (vi) is e
Match the type of mixture of compounds in Column I with the technique of separation/purification given in Column II.
(i) is e (ii) is d (iii) is a (iv) is b (v) is c
Which of the two structures (A) and (B) given below is more stabilised by resonance? Explain. (A) CH3COOH (B) CH3COO-
Compound A's two resonant structures are not equal, but compound B, which has a negative charge on the oxygen atom, has two equivalent structures, making it more stable.
By mistake, an alcohol (boiling point 97°C) was mixed with a hydrocarbon (boiling point 68°C). Suggest a suitable method to separate the two compounds. Explain the reason for your choice.
Steam distillation can be used to separate the mixture. The boiling points of alcohol and hydrocarbon are almost same. Steam distillation is used to purify temperature-sensitive materials in...
Resonance structures of propenal are given below. Which of these resonating structures is more stable? Give a reason for your answer.
Because all of the atoms in structure I have a complete octet, while the carbon atom with a positive charge does not have a complete octet in structure II, structure I will be more stable.
Which of the following compounds will not exist as a resonance hybrid. Give the reason for your answer:
Conjugation is only feasible when an atom possesses any of the double bond's charge or alternate positions. Because of this, CH3OH will not exist as a resonance hybrid here.
Give three points of differences between the inductive effect and resonance effect.
1. In the inductive effect, the electron is only sent through the sigma bond, but in the resonance effect, the electron is transmitted through both the sigma and pi bonds. 2. It is feasible when the...
Identify the most stable species in the following set of ions giving reasons:
(i) Because the bromine atom destabilises the positive charge on a carbon atom, C+H3 will be more stable. Bromine possesses a lone pair of electrons and is an electron-withdrawing group. (ii) CCl3...
Draw the resonance structures of the following compounds.
Write structural formulae for compounds named as- (a) 1-Bromoheptane (b) 5-Bromoheptanoic acid
Name the compounds whose line formulae are given below:
(i)The compound's name is 3-ethyl-4-methyl-5-heptane-2-one. (ii) The chemical is called 1-nitro-cyclohexane-2-ene.
Three students, Manish, Ramesh and Rajni were determining the extra elements present in an organic compound given by their teacher. They prepared the Lassaigne’s extract (L.E.) independently by the fusion of the compound with sodium metal. Then they added solid FeSO4 and dilute sulphuric acid to a part of Lassaigne’s extract. Manish and Rajni obtained Prussian blue colour but Ramesh got a red colour. Ramesh repeated the test with the same Lassaigne’s extract but again got red colour only. They were surprised and went to their teacher and told him about their observation. A teacher asked them to think over the reason for this. Can you help them by giving the reason for this observation? Also, write the chemical equations to explain the formation of compounds of different colours
3NaCNS + FeSO4 + dilute sulphuric→ Fe(CNS)3 (Blood red colour) + 3Na+ Ramesh's organic molecule has both Nitrogen and Sulphur, resulting in the Blood-red colour of Fe(CNS)3, whereas Manish and...
Write structures of various carbocation that can be obtained from 2-methyl butane. Arrange these carbocations in order of increasing stability.
The following is the stability order: (III) > (II) > (I) > IV This is due to the fact that (III) is a tertiary carbocation, (II) is a secondary carbocation, and (I) and (IV) are primary...
The structure of triphenylmethylcation is given below. This is very stable and some of its salts can be stored for months. Explain the cause of the high stability of this cation.
Triphenylcarbocation is a tertiary carbocation with a positive charge on the carbon atom that is stabilised by resonance thanks to the three phenyl groups. The stability of the system improves as a...
Which of the following ions is more stable? Use resonance to explain your answer.
Inductive effect, hyperconjugation, resonance, and other factors influence the stability of carbocation. Because structure A has a carbocation that is both primary and allylic, whereas...
Draw the possible resonance structures for and predict which of the structures is more stable. Give the reason for your answer.
Structure C is more stable than structure A because all atoms' octets are complete in structure C, but in structure A, a C-atom with a positive charge does not have 8 electrons in its valence...
If a liquid compound decomposes at its boiling point, which method(s) can you choose for its purification. It is known that the compound is stable at low pressure, steam volatile and insoluble in water.
Because the liquid component decomposes near its boiling point, indicating that it is heat-sensitive, we purify it using "Steam distillation." For temperature-sensitive materials, this is done.
In DNA and RNA, the nitrogen atom is present in the ring system. Can the Kjeldahl method be used for the estimation of nitrogen present in these? Give reasons.
Because nitrogen cannot be transformed into ammonium sulphate at the conditions employed in the Kjeldahl technique, it cannot be used to estimate nitrogen levels in DNA and RNA. This test for DNA...
Which of the following selected chains is correct to name the given compound according to the IUPAC system.
a. The main(parent) chain should be the longest carbon chain feasible, including all carbons containing functional groups. With both functional groups in the main chain, the aforementioned option is...
Compounds with same molecular formula but differing in their structures are said to be structural isomers. What type of structural isomerism is shown by
The functional groups (thioether/sulphide) are the similar in both structures, but the atoms in the main chain are arranged differently. As a result, they can show chain isomerism.
Show the polarisation of carbon-magnesium bond in the following structure. CH3—CH2—CH2—CH2—Mg—X
CH3—CH2—CH2—δ-CH2—δ+Mg+ X- Because the electronegativity differential in the C-Mg bond is strongly polarised, carbon is more electronegative than magnesium. Because C is more electronegative, its...
Explain, how is the electronegativity of carbon atoms related to their state of hybridisation in an organic compound?
There are three forms of carbon hybridization in organic compounds: sp, sp2, and sp3. The electronegativity of carbon increases as the ‘s' character increases because ‘s' orbitals are closer to the...
