1. 3n 2. n 3. 2n 4. n/2 Solution: 2n When two moles of ethyl bromide are combined with Sodium metal in the presence of dry ether, n-butane is formed. The reaction described above is known as the...
The number of sigma and Pi bonds respectively present in a molecule of toluene is,
10σ and 3π bonds 12σ and 3π bonds 15σ and 3π bonds 6σ and 3π bonds Solution: 15σ and 3π bonds Toluene is C6H5CH3. No. of σ bonds is 15. No. of π bonds...
Which of the following is not basic alpha-amino acid?
1. Histidine 2. Lysine 3. Arginine 4. Alanine Solution: Alanine Except Alanine all other amino acids contain - NH2 or - N atom in the side chain.
Which of the following methods of refining of crude metals uses green logs of wood ?
1. Zone refining 2. Polling 3. Liquation processes 4. Vapour phase refining Solution: Polling Poling is a crude metal refining technique that involves stirring molten metal in a ladle with a green...
Enthalpy of fusion of water is 6.0 KJ/mol at 0 degree celsius. What is the enthalpy change when 0.0045 kg of water undergoes fusion?
1.5×10-2 kJ 1.5 kJ 150 kJ 1.5×10-3 kJ Solution: 1.5 kJ
One atom of an element ‘X’ weighs 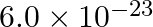 g. mass of 1 mole of atoms is
g. mass of 1 mole of atoms is
40 mol-1 32.31 mol-1 4.5 mol-1 36.13 mol-1 Solution: 36.13 mol-1 The mass of one atom of an element, A = 6.0 × 10−23 g We know, 1 mole of an atom is 6.023 × 1023 atoms. 1 mole of an atom = its...
O- acetyl salicylic acid is used as
1. Antimicrobial 2. tranquilizer 3. antioxidant 4. antirhuematic drug Solution: antirhuematic drug Aspirin, commonly known as acetylsalicylic acid (ASA), is a pain reliever, fever reducer, and...
Haloform test is NOT exhibited by
1. Ethanal 2. Propanal 3. Propanone 4. Acetophenone Solution: Propanal Haloform reaction is given by the compounds containing acetyl groups (CH3CO−). All the given options...
Oraganic compounds ‘A’ and ‘B’ react with sodium metal and liberate hydrogen gas. ‘A’ and ‘B’ react together to give ethyl methanoate, compound A and B respectively are
Solution: Option 1 is correct
Predict the product of following reaction
A. 2-hydroxy cyclohexanol B. Cyclohexene C. Benzene D. 2-Bromo cyclohexene Solution: Cyclohexene An alkene (cyclohexene) will be created in this case by dehydrating alcohol...
A solution contains 1.2 moles of solute and 13.8 moles of solvent. Hence the mole fraction of the solute is
1. 0.085 2. 0.09 3. 0.08 4. 0.075 Solution: 0.08 The mole fraction is given as: $ {{X}_{solute}}=\frac{{{n}_{solute}}}{{{n}_{solute}}+{{n}_{solvent}}} $ $...
Calculate the volume of bcc type unit cell having edge length 288 pm and 7.2 g cm-3 density? (mass of unit cell= 52g)
$ 1.\,\,7.22\,c{{m}^{3}} $ $ 2.\,\,1.38\times {{10}^{-1}}\,c{{m}^{3}} $ $ 3.\,\,2.38\times {{10}^{-2}}\,c{{m}^{3}} $ $ 4.\,\,3.023\times {{10}^{-2}}\,c{{m}^{3}} $ Solution: $ 1.\,\,7.22\,c{{m}^{3}}...
What type of glycosidic linkage is present in maltose ?
1. 1 → 6 β 2. 1 → 4 β 3. 1 → 6 α 4. 1 → 4 α Solution: 1 → 4 α Maltose is a disaccharide made up of alpha 1,4 glucose molecules linked together. Maltase is the enzyme that breaks it down. It acts on...
Which of the following pairs of solutions will be isotonic at the same temperature?
1M NaCl and 1M MgCl2 1.5M KCl and 2.5M Urea 1.5M AlCl3 and 2M Na2SO4 1M NaCl and 2M MgCl2 Solution: 1.5M AlCl3 and 2M Na2SO4 The osmotic pressure and molar concentration in isotonic solutions should...
Identify optically inactive compound among the following
1. 2 - chloropropanal 2. 2 - chloropentane 3. 2 - chloro,2-methylbutane 4. 2 - chlorobutane Solution: 2 - chloro,2-methylbutane 2-chloro-2-methylbutane is optically inactive as no chiral center is...
Which among the following complex ion is NOT diamagnetic?
$ 1.\,\,{{\left[ Ni{{(CN)}_{4}} \right]}^{2-}} $ $ 2.\,\,[Ni{{(CO)}_{4}}] $ $ 3.\,\,{{\left[ CO{{(N{{H}_{3}})}_{6}} \right]}^{3+}} $ $ 4.\,\,{{[NiC{{l}_{4}}]}^{2-}} $ Solution: $...
The standard enthalaphy of formation of ethane is -84.7 KJ/mol. What is the enthalapy change for the formation of 0.06 kg ethane?
1. -42.35 kJ 2. -169.4 kJ 3. -50.82 kJ 4. -236.7 kJ Solution: -169.4 kJ Given mass = 0.06 kg = 60 g Molar mass of ethane = 2(12) + 6(1) = 30 g number of moles of ethane in 60 grams = 60/30 = 2 We...
What happens during the action of catalyst?
1. Temperature increases 2. Temperature decreases 3. Ea increases 4. Ea decreases Solution: Ea decreases A catalyst is a material that speeds up a reaction without being consumed in the process....
Styrene and 1,3-butadiene on polymerisation forms
1. Buna-N 2. Neoprene 3. Butyl rubber 4. Buna-S Solution: Buna-S Styrene-butadiene rubber is another name for the rubber produced (SBR). Bu stands for butadiene, Na for sodium, and S for styrene in...
The temperature and pressure of 4 dm3 of an ideal gas are doubled. The volume of the gas now is
2 dm3 3 dm3 4 dm3 8 dm3 Solution: 4 dm3 From ideal gas equation, we have: $ \frac{{{P}_{1}}{{V}_{1}}}{{{T}_{1}}}=\frac{{{P}_{2}}{{V}_{2}}}{{{T}_{2}}} $ $ We\,have\,: $ $...
PCl5 exists but NCl5 doesn’t, because of
1. NCl5 is unstable 2. larger size of nitrogen 3. inertness of nitrogen 4. non-availability of vacant d- atomic orbitals Solution: non-availability of vacant d- atomic orbitals By employing the...
Which among following raections of water generates oxygen gas ?
1. Reaction with calcium oxide 2. Reaction with sodium 3. Reaction with ammonia 4. Photosynthesis reaction Solution: Photosynthesis reaction The overall balanced equation of photosynthesis reaction...
Nitronic acid with 50% sulphuric acid at room temperature forms an aldehyde. This reaction is known as
1. Gabriel phthalamide synthesis 2. Nef carbonyl synthesis 3. Etard reaction 4. Hoffmann degradation Solution: Nef carbonyl synthesis Nef carbonyl synthesis Acid hydrolysis of a salt of a...
The sodium fusion extract of aniline is boiled with ferrous sulphate and then acidified with concentrated sulphuric acid. The color of the complex formed is
1. Violet 2. Yellow 3. Black 4. Prussian blue Solution: Prussian blue The reaction equation is: $ FeS{{O}_{4}}+NaOH\,\,\to \,\,Fe{{(OH)}_{2}}+N{{a}_{2}}S{{O}_{4}} $ $ 6NaCN+Fe{{(OH)}_{2}}\,\,\to...
Which among the following elements exhibits only +3 oxidation state?
Nd La Dy Yb Solution: La We have Z = 57 for La. In +3 oxidation state Lanthanum(La) achieves the noble gas configuration. So, Lanthanum exhibits only +3 oxidation state.
Any galvanic cell working under standard condition, if the equation of the cell reaction is multiplied by 3 then
A. Eo increase three times B. Eo is unchanged C. Eo decreases three times D. Go remains unchanged Solution: Eo is unchanged $ Nernst's\text{ }equation\text{ }states~: $ $...
What type of hybridization is present in PCl5 molecule?
A. sp3d2 B. sp3d3 C. sp3d D. dsp2 Solution: sp3d2 PCl5 is expected to have a hybridization of sp3d and a shape of trigonal bipyramidal. But in solid-state PCl5 exists in ionic form...
Identify the amphoteric oxide from the following
B2O3 CO ZnO CaO Solution: ZnO Zinc oxide (ZnO) is known as amphoteric oxide because it has both acidic and basic properties.
Which of the following allotropic forms of sulphur exists in chair form?
1. Cyclo - sulphur 2. α - Sulphur 3. β - Sulphur 4. plastic sulphur Solution: Cyclo - sulphur
The standard electode potential of calomel electrode is increased by
decreasing the concentration of KCl solution Increasing the quality of Hg2Cl2 decreasing the quantity of calomel Increasing the concentration of KCl solution Solution: decreasing the concentration...
What is the charge on 0.05 mol of electrons?
1. 2412.5 C 2. 965 C 3. 4825 C 4. 9650 C Solution: 4825 C We have: Mass of one electron = 9.10×10−31 kg Charge of one electron = 1.602×10−19 coulomb $...
Which is most stable oxidation state of Vanadium (Atomic no. 23)?
1. +2 2. +3 3. +4 4. +5 Solution: +5 Ammonium metavanadate, NH4VO3, is the most common source of vanadium in the +5 oxidation state. This isn't particularly water soluble, thus it's normally...
When aniline forms 2,4,6- tribromo aniline on reaction with bromine water , it undergoes
1. nucleophilic addition 2. nucleophilic substitution 3. electrophilic substitution 4. electrophilic addition Solution: electrophilic substitution Bromination is the reaction that is taking place...
For the elementary reaction 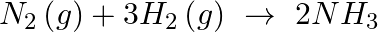 . Identify the correct relation from the following relations:
. Identify the correct relation from the following relations:
Solution: the correct option is A As the reaction progresses, the reactant concentration decreases and the product concentration rises. As a result, the rate of...
The decreasing order of thermal stability of hydrides of group 15 elements is
Solution: The correct option is 2. Hydride stability reduces as one moves along the group from NH3 to SbH3. This is because their bond dissociation enthalpy has decreased. The...
The magnitude of  is
is
1. Nature of solute only 2. Nature of the solute and the concentration of solution 3. Nature of the solvent and the concentration of solution 4. Nature of solvent only Solution: 3. Nature of the...
What is SI unit of Luminous intensity?
1. Ampere 2. 0C 3. Kelvin 4. Candela Solution: Candela The SI unit of luminous intensity is candela.
Two moles of an ideal gas are expanded from volume of 15.5 litre to 20 litre against a constant external pressure of 1 atmosphere. The amount of work done is
- 4.5 J 2×10-2 J - 506.5 J 225 J Solution: - 506.5 J The following equation can be used to describe the work done by a system against an external pressure: We know that: w = -Pext ∆V 1atm...
Which of following is a mineral of aluminium?
1. Dolomite 2. Siderite 3. China clay 4. Calamine Solution: China Clay $ Dolomite-[CaMg{{(C{{O}_{3}})}_{2}}] $ $ Siderite-[FeC{{O}_{3}}] $ $ China\text{ }Clay-[Al2{{O}_{3}}.2Si{{O}_{2}}.2{{H}_{2}}O]...
Neo-pentyl alcohol is a –
1. Tertiary alcohol 2. Secondary alcohol 3. Primary alcohol 4. Dihydric alcohol Solution: Primary alcohol The primary alcohols are those that have only one alkyl group linked to the carbon atom of...
Which among following element has highest chemical reactivity?
1. Be 2. Mg 3. Sr 4. Ba Solution: Ba Because of its high reactivity, barium (Ba) is only found in nature in conjunction with other elements. Sulfate and carbonate are the most common compounds...
Which of the following rate expression is true for alkaline hydrolysis of methyl bromide ?
Solution: Option 1. is the correct answer Consider methyl bromide's alkaline hydrolysis to yield methanol. $ C{{H}_{3}}-Br+NaOH\,\,\xrightarrow{\Delta }\,\,C{{H}_{3}}-OH+NaBr $...
Linkage Isomers Ionization Isomers Geometrical isomers Coordination isomers Solution: Ionization isomers When the same molecular formula produces distinct ions in a solution, this is known as...
Which among following gases is readily adsorbed by activated charcoal ?
H2 SO2 N2 O2 Solution: SO2 SO2 is an easily liquefiable gas and easily liquefiable gases are adsorbed to a greater extent than elemental gases like N2, O2, and H2.
Which one of the following is a neutral oxide?
$ 1.\,A{{l}_{2}}{{O}_{3}} $ $ 2.\,{{N}_{2}}O $ $ 3.\,N{{a}_{2}}O $ $ 4.\,S{{O}_{2}} $ Solution: $ 2.\,{{N}_{2}}O $ Oxides that are neither acidic nor basic are known as neutral oxides. In other...
A polymer obtained from the monomers ethylene glycol and dimethyl terephthalate is
1. Nylon-6 2. Terylene 3. Bakelite 4. Nylon-6,10 Solution: terylene Ethylene glycol (1,2 ethanediol) and terephthalic acid are the monomers of terylene (1,4 benzene dicarboxylic acid). Terylene...
Identify diamagnetic ion from following. (Atomic no.of Na=11, Cu=29, Fe=26, Cr=24)
Cu2+ Fe3+ Na+ Cr3+ Solution: Na+ Cu2+ = 1s22s22p63s23p63d9 Fe3+ = 1s22s22p63s23p63d5 Cr3+ = 1s22s22p63s23p63d3 Na+ = 1s22s22p6 All the given ions except Na+ have unpaired electrons in their...
Identify the oxidising agent in following redox reaction
$ C{{l}_{2}}+2B{{r}^{-}}\,\to \,2C{{l}^{-}}+B{{r}_{2}} $ $ 1.\,C{{l}^{-}} $ $ 2.\,B{{r}^{-}} $ $ 3.\,B{{r}_{2}} $ $ 4.\,C{{l}_{2}} $ Solution: $ 4.\,C{{l}_{2}} $ Br is the reducing agent because it...
Among the following, an artificial sweetening agent which does not contain – CO – NH – bonding in molecule is
1. Sucralose 2. Alitame 3. Aspartame 4. Saccharine Solution: Sucralose Alitame, Aspartame and Saccharine all three contain the CO-NH linkage whereas Sucrolose contains the C-O linkage....
Which of the following nitro-alkane doesn’t react with nitrous acid?
2-methyl-2-nitropropane 2-nitropropane Nitroethane 1-nitropropane Solution: 2-methyl-2-nitropropane The structure of 2-methyl-2-nitropropane is as follows: We can see that...
Pumice stone is an example of
Solid Sol Emulsion Aerosol Solid foam Solution: Solid Foam Pumice stone is an example of solid foam. In this type of colloid, the dispersion medium is solid and the dispersion phase is...
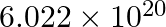 molecules of urea are present in 100 mL of its solution. The concentration of solution is
molecules of urea are present in 100 mL of its solution. The concentration of solution is
0.1 M 0.02 M 0.01 M 0.001 M Solution: 0.01 M Given: 6.02×1020 molecules of urea The volume of solution is 100/1000 = 0.1 L 1 mole of urea have 6.02×1023 molecules Number of moles present = 6.02×1020...
What products are expected from disproportionation reaction of orthophosphorus acid?
$ 1.\,\,{{H}_{3}}P{{O}_{3}}+P{{H}_{3}} $ $ 2.\,\,{{H}_{3}}P{{O}_{4}}+P{{H}_{3}} $ $ 3.\,\,P{{H}_{3}}+{{P}_{2}}{{O}_{5}} $ $ 4.\,\,{{H}_{3}}P{{O}_{3}}+{{P}_{2}}{{O}_{5}} $ Solution: $...
Identify the product B in following conversion!
$Chlorobenzene+{{H}_{2}}O\xrightarrow[\Pr essure]{Cu,\,673K}A\xrightarrow[373K]{conc.\,{{H}_{2}}S{{O}_{4}}}B$ 4-hydroxybenzene Sulphonic acid Benzene Sulphonic acid 2-hydroxybenzene Sulphonic acid...
What is the quantity of Gold chloride obtained when 4.5 g of gold and 2.1 g of Chlorine are sealed in a a tube and heated at 150 degreees C?
4.5 g 4.8 g 6.07 g 20.7 g Solution: 6.07 g Excess reagents are reactants that are not used up when a chemical reaction is completed. Because its quantity limits the amount of product generated, the...
What is the standard emf of following cell?
$ N{{i}_{(s)}}|(1M)\,Ni_{(aq)}^{2+}||(1M)\,Au_{(aq)}^{3+}|Au\left( s \right) $ $ if\,E_{Ni}^{\circ }=-0.25V,\,E_{Au}^{\circ }=1.50V $ -1.25 V 1.75 V 1.25 V -1.75 V Solution: 1.75 V $...
How many tertiary carbon atoms and primary carbon atoms respectively are present in 2-iodo-3, 3-dimethyl pentane?
2,4 0,4 2,3 1,3 Solution: 0,4 We can represent 2-iodo-3, 3-dimethyl pentane as follows: ${{H}_{3}}C-CH(I)-C{{(C{{H}_{3}})}_{2}}-C{{H}_{2}}-C{{H}_{3}}$ As we can see that there are zero carbon atoms...
5600 sec 360.0 sec 560.0 sec 3364 sec Solution: 560 sec $ Given: $ $ {{[R]}_{0}}=0.0210\,M~ $ $ [R]=0.0150\,M~ $ $ k=6\times {{10}^{-4}}\,{{\sec }^{-1}} $ $ For\text{ }a\text{ }first\text{...
Which of the following alcohols is not having Csp3-OH bond?
Phenyl Methanol 2-Methyl Propan-2-ol Propan-2-ol Vinyl Alcohol Solution: Vinyl Alcohol Vinyl Alcohol is represented as follows: The simplest enol is vinyl alcohol, commonly...
What is the number of hydroxyl group present in lactic acid?
Zero Three Two One Solution: One Lactic acid (2-hydroxy propionic acid) is a bifunctional molecule that has both a carboxylic acid and a hydroxyl group, making it useful in a...
Which of the following is not an octahedral complex?
$ 1.\,\,{{[Ir{{({{C}_{2}}{{O}_{4}})}_{2}}C{{l}_{2}}]}^{3-}} $ $ 2.\,\,{{[CoC{{l}_{2}}{{(en)}_{2}}]}^{+}} $ $ 3.\,\,{{\left[ Co{{(en)}_{2}}{{(N{{O}_{3}})}_{2}} \right]}^{+}} $ $ 4.\,\,\left[...
What is the oxidation state of iron in potassium ferrate?
+3 +4 +6 +2 Solution: +6 Potassium ferrate has the chemical formula K2FeO4. This purple salt is paramagnetic, and is a rare example of an iron(VI) compound. $...
What is the number of moles of Silver Chloride precipitated when excess of aqueous silver nitrate is treated with ![Rendered by QuickLaTeX.com [Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-7f8a1797a451d4bad014fffc46745e19_l3.png)
1 mole 2 mole 3 mole 4 mole Solution: 1 mole Only one mole of AgCI is precipitated with an excess of AgNO3 because only one Cl- is in the ionization sphere.
When slaked lime is passed through excess carbon dioxide, it forms
$ 1.\,\,CaC{{O}_{3}} $ $ 2.\,\,CaC{{l}_{2}} $ $ 3.\,\,Ca{{(HC{{O}_{3}})}_{2}} $ $ 4.\,\,C{{a}_{2}}HC{{O}_{3}} $ Solution: $ 1.\,\,CaC{{O}_{3}} $ Passing Carbon dioxide through slaked lime gives...
Which of the following is not found in hybridization?
Formation of sigma bonds Mixing and recasting of atomic orbitals Excitation of electrons Loss and gain of electrons Solution: Loss and gain of electrons The following ideas are important in...
Which of the following statements is true for pyran?
It is a saturated aliphatic compound, It is a homocyclic compound. It is a heterocyclic compound with oxygen atom in the ring The molecular formula of Pyran is C5H5S Solution: It is a heterocyclic...
Aluminium crystallizes in face centered cubic structure, having atomic radius 125pm. The edge length of unit cell of aluminium is
253.5 pm 353.5 pm 465 pm 250 pm Solution: 353.5 pm $ For\text{ }a~FCC~or~~CCP~unit\text{ }cell,we\,have: $ $ 4r=\surd 2\times a $ $ \Rightarrow a=\frac{4r}{\surd 2}=353.5pm $
Which of the following elements has six unpaired electrons in observed electronic observations?
Fe (Z = 26) Cr (Z = 24) Cu (Z = 29) Mn (Z = 25) Solution: Cr (Z = 24) The following is the electrical configuration of the given elements: $...
Which of the following is the strongest reducing agent?
Na Mg Li Ca Solution: Li Lithium has the highest ionization potential of all alkali metals, implying that its tendency to ionize to give Li+ ions should be the least, implying that Li should be the...
Which among the following electrical properties has SI unit siemens per meter?
Conductance Conductivity Resistance Resistivity Solution: Conductivity The ability of an electrolyte solution to conduct electricity is measured by its conductivity (or specific conductance)....
Aniline reacts with bromine water at room temperature to give
3-Bromoaniline 2-Bromoaniline 4-Bromoaniline 2,4,6-Tribromoaniline Solution: 2,4,6-Tribromoaniline This happens due to the highly activating nature of -...
Which of the following statements is not true for glyceraldehyde?
It is a sugar molecule It is optically active It contains two asymmetric carbon atoms It has carbonyl and hydroxyl group Solution: It contains two asymmetric carbon atoms ...
Copper crystallizes as face centered cubic lattice , with edge length of unit cell 361 pm. Calculate the radius of the copper atom.
108.6 pm 127.65 pm 181.6 pm 157.6 pm Solution: 127.65 pm For F.C.C, a√2 = 4r 361×√2 = 4r r = 127.65 pm
Which of the following reagents is used to avoid further oxidation of aldehydes?
$ 1.\,\,{{C}_{5}}{{H}_{5}}N{{H}^{+}}Cr{{O}_{3}}Cl $ $ 2.\,\,{{K}_{2}}C{{r}_{2}}{{O}_{7}}/dil.\,{{H}_{2}}S{{O}_{4}} $ $ 3.\,\,dil.\,HN{{O}_{3}} $ $ 4.\,\,Cr{{O}_{3}} $ Solution: $...
What is the molecular formula of allyl bromide?
$ 1.\,\,{{C}_{2}}{{H}_{4}}Br $ $ 2.\,\,{{C}_{2}}{{H}_{3}}Br $ $ 3.\,\,{{C}_{3}}{{H}_{5}}Br $ $ 4.\,\,{{C}_{3}}{{H}_{6}}Br $ Solution: $ 3.\,\,{{C}_{3}}{{H}_{5}}Br $ $ The\text{ }Allyl\text{...
What is the bond length of C-H bonds in alkanes?
154 pm 120 pm 133pm 112pm Solution: 112 pm
Which of the following solution will have highest freezing point depression?
M Glucose M Sucrose M Urea M KCl Solution: M KCl ΔTf = i Kf m ΔTf is the freezing point depression, i is the van’t Hoff factor, Kf is the molal freezing point depression constant for the solvent,...
Identify the polymer from following, that contains amide linkage
Terylene PHBV Nylon - 6,6 Dextron Solution: Nylon-6,6 Polyamides have the amide linkage. Example- Nylon 6-6, Nylon 6 Nylon 6, 6 is used in the manufacturing of sheets, brush bristles, and...
The volume of 400cm3 of chlorine gas at 400 mm of Hg is decreased to 200 cm3 at constant temperature. What is new pressure of the gas?
800 mm of Hg 1600 mm of Hg 200 mm of Hg 600 mm of Hg Solution: 800 mm of Hg This is an example of Boyle's law, which states that the volume of a gas is inversely proportional to the pressure at...
Which of the following compounds is used to avoid oxidation in food?
O-hydroxy benzoic acid Acetyl Salicylic acid Ethyl Salicylate Butylated hydroxy Anisole Solution; Butylated hydroxy Anisole Butylated hydroxyanisole (BHA) is an antioxidant made up of...
The H-H bond energy is 430 KJ/mol and Cl-Cl bond energy is 240 KJ/mol. 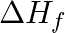 for HCl is 90 KJ/mol. Then HCl bond energy is
for HCl is 90 KJ/mol. Then HCl bond energy is
360 KJ/mol 213 KJ/mol 180 KJ/mol 425 KJ/mol Solution: 425 KJ/mol $ \frac{1}{2}{{H}_{2}}(g)+\frac{1}{2}C{{l}_{2}}(g)\,\,\to \,\,HCl $ $ \Delta {{H}_{f}}=-90KJ $ $ \Delta {{H}_{f}}=~B.E\text{...
Van’t Hoff factor (i) for the centimolal solution of ![Rendered by QuickLaTeX.com {{\text{K}}_{\text{3}}}\text{ }\!\![\!\!\text{ Fe(CN}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-1e79ee193daba828d4bfe9ad8e4b1525_l3.png) is 3.333. What is it’s percentage dissociation?
is 3.333. What is it’s percentage dissociation?
80% 70% 33.33% 77.7% Solution: 77.7% $ {{K}_{3}}[Fe{{(CN)}_{6}}]\to 3{{K}^{+}}+{{[Fe{{(CN)}_{6}}]}^{3-}} $ $ We\,get: $ $ i=\alpha n+(1-\alpha )=1+\alpha (n-1)=1+\alpha (4-1)~ $ $ Van't\text{...
What is the melting point of zinc?
473 K 1193 K 423 K 692 K Solution: 692 K The metal Zinc has a melting point of 692K.
In Merck’s method, hydrogen peroxide is obtained from?
$ 1.\,\,Ba{{O}_{2}}+{{H}_{2}}S{{O}_{4}} $ $ 2.\,\,N{{a}_{2}}{{O}_{2}}+{{H}_{2}}S{{O}_{4}} $ $ 3.\,\,Ba{{O}_{2}}+{{H}_{3}}P{{O}_{4}} $ $ 4.\,\,Ba{{O}_{2}}+{{H}_{2}}O+C{{O}_{2}} $ Solution: $...
An element crystallizes in bcc structure. The number of unit cells of an element in 4 g of it is ( given mass = 40)
0.1 NA / 2 2(0.1) NA 0.1 NA 2 NA Solution: 0.1 NA / 2 Given: Mass of element = 40 g and Z = 2 (bcc structure) The number of moles in 1 gram of element = 1/40 Similarly, number of moles in 4 gram of...
Which of the following is NOT correct in hybridisation?
There should be very little difference in energy of involving orbitals The shape of hybrid orbitals is the same as that of atomic orbitals The number of hybrid orbitals formed is equal to the...
Dumas method is used for the estimation of
Nitrogen Sulphur Oxygen Carbon Solution: Nitrogen In analytical chemistry, the Dumas method is a method for quantifying nitrogen in chemical substances based on a method proposed by Jean-Baptiste...
Which of the following compounds is obtained when 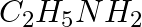 is treated with excess
is treated with excess 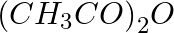 in presence of pyridine?
in presence of pyridine?
$ 1.\,\,{{C}_{2}}{{H}_{5}}N{{(COC{{H}_{3}})}_{2}} $ $ 2.\,\,{{\left( {{C}_{2}}{{H}_{5}} \right)}_{2}}NH $ $ 3.\,\,{{C}_{2}}{{H}_{5}}COOH $ $ 4.\,\,{{C}_{2}}{{H}_{5}}NHCOC{{H}_{3}} $ Solution: $...
Which among the following polymers is an example of addition polymers?
Dacron Unrealformaldehyde Polymer Nylon-6 Polythene Solution: Polythene The addition polymers are formed by the repeated addition of monomer molecules possessing double or triple bonds, e.g., the...
Which among the following is a first oxidation product of butan-2-ol?
Butanal Butanoic Acid Propanoic acid and carbon di-oxide Butan-2-one Solution: Butan-2-one Ketone is the first oxidation product of secondary alcohol. For example, butan-2-ol is oxidised to...
Which of the following compounds is obtained when benzene is treated with CO and HCl in presence of catalyst anhyfrous 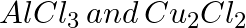 under high pressure?
under high pressure?
Toulene Benzoic Acid Benzaldehyde Acetophenone Solution: Benzaldehyde When the vapors of CO and HCI are passed into benzene in the presence of anhydrous AICI3/CuCI, benzaldehyde...
Which among the following oxides of nitrogen, the nitrogen atom contains one unpaired electron?
$ A)\,{{N}_{2}}{{O}_{4}} $ $ B)\,N{{O}_{2}} $ $ C)\,{{N}_{2}}{{O}_{5}} $ $ D)\,{{N}_{2}}{{O}_{3}} $ Solution: $ B)\,N{{O}_{2}} $ Among the oxides of nitrogen, only NO2 contains odd number of...
The rate of first order reaction is A->B is 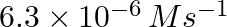 . if [A] = 0.3 M what is the arye
. if [A] = 0.3 M what is the arye
$ A)\,\,2.1\times {{10}^{-5}}\,{{s}^{-1}} $ $ A)\,\,1.2\times {{10}^{-5}}\,{{s}^{-1}} $ $ A)\,\,1.3\times {{10}^{-5}}\,{{s}^{-1}} $ $ A)\,\,1.6\times {{10}^{-5}}\,{{s}^{-1}} $ Solution: $...
Which of the following equations shows the relationship between heat of reaction at constant pressure and heat of reaction at constant volume if the temperature is not constant?
$ 1.\,\,\Delta H-\Delta n=\Delta URT $ $ 2.\,\,\Delta H-\Delta U=\Delta nRT $ $ 3.\,\,\Delta H=\Delta nRT $ $ 4.\,\,\Delta H=\Delta U-RT $ Solution: $ 2.\,\,\Delta H-\Delta U=\Delta nRT $...
Which of the following elements is refined by zone refining?
A. Gallium B. Bismuth C. Copper D. Zinc Solution: gallium The zone refining method is generally used to refine metalloids and ultra-pure metal is obtained. This is based on the idea that impurities...
Which of the following is not a tranquilizer?
A. Iproniazid B. Serotonin C. Veronal D. Prontosil Solution: Prontosil A tranquillizer is a medicine that is used to treat anxiety, fear, tension, agitation, and other mental problems, with the goal...
Whivh of the following metals react with dilute Sulphuric acid?
A. Fe B. Be C. Cu D. Hg Solution: Fe Chemical reaction: $Fe+{{H}_{2}}S{{O}_{4}}\to FeS{{O}_{4}}+{{H}_{2}}$ Iron produces iron sulphate and hydrogen gas when it reacts with dilute sulphuric acid....
