Solution:
Inductive Effect: The inductive impact alludes to the extremity delivered in a particle because of higher electronegativity of one molecule contrasted with another.Atoms or gatherings which lose electron towards a carbon iota are said to have +1 Effect.
Those iotas or gatherings which draw electron away from a carbon molecule are said to have – I Effect.
Commomexamples of – I impact are:
NO2, F, Cl, Br, I, OH and so forth
Instances of +1 impact are (Electron delivering)
(CH3)2C—, (CH3)2CH—, CH3CH2—CH3—and so forth
Electromeric impact: The electromeric impact alludes to the extremity delivered in a numerous reinforced compound as it is drawn nearer by a reagent.
![]()
The particle A has lost its portion in the electron pair and B has acquired this offer.
Subsequently A gets a positive charge and B a negative charge. It is a brief impact and happens just within the sight of a reagent.
(a) – I-impact as displayed beneath:
As the quantity of halogen particles diminishes, the generally speaking – I-impact diminishes and the corrosive strength diminishes appropriately.
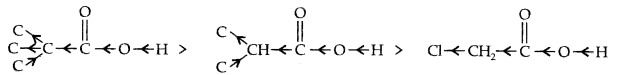
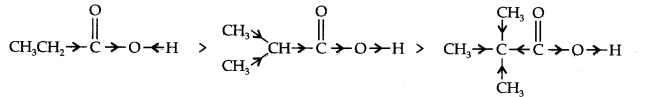
(b) +I-impact as displayed beneath:
As the quantity of alkyl bunches builds, the +I-impact increments and the corrosive strength
diminishes as needs be.