Option (i) and (ii) are the answers.
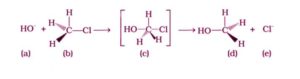
Alkyl halide is the main reactant in the given reaction. A transient condition is found here, in which one bond is broken and another is created synchronously, that is, in one step. As a result, it follows the SN2 mechanism.
The nucleophile approaches the carbon at an angle of 180 degrees to the leaving group in this process. As a result, the reactant and product have a configuration that is diametrically opposed.
