If you’ve ever wondered what an electron is or what its charge is, this blog is for you. In this blog, we’ll take a look at what electrons are and what their charge is. We’ll also discuss how electrons are important in physics, and how they’re used in technology. So read on to learn about this fascinating atom!
What is an Electron?

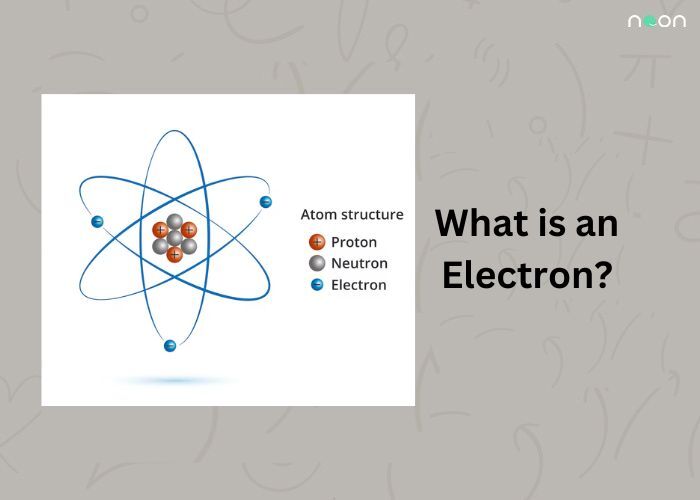
Electrons are the building blocks of atoms and molecules. They are the smallest particle of an atom that possesses the element’s nucleus. Electrons orbit around the nucleus in shells called energy levels. The higher the number, the more electrons are in that shell.
Each atom has a set number of protons in its nucleus, and those protons determine how many electrons a particular atom can have. For example, the atom with the number 10 nucleus can have 10, 11, 12, or 13 electrons in its shell. So, knowing what an electron is and what it does is key to understanding how atoms and molecules work.
Discovery of Atom

In 1896, J.J. Thomson observed that electricity flowed through cathode ray tubes and found that all the atoms in a material contained subatomic particles called electrons. He also suggested the atom’s plum pudding concept – a particle with a positive charge on one end and a negative charge on the other (hence its name, lepton).
It was not until 1911 that scientists discovered another particle, the neutron. Electron discovery marked an important development in our understanding of matter and energy at the atomic level.
What is the charge of an Electron?

An electron has a negative charge, which makes it an electron donor. Charges are calculated by units known as coulombs(C), and the charge of an electron is 1.6 x 10–19 Coulomb.
Every atom has a nucleus that contains protons (the nucleus of an atom is the heaviest atom). Protons are held together by a weak force, and electrons orbit the nucleus. When an electron is orbiting the nucleus of an atom, it is in a shell called an orbit.
The number of electrons in an orbit determines the chemical element of the atom. For example, the number of electrons in an orbit that is in the second shell of an atom (2s orbit) determines the element that the atom is.
Conclusion
In this blog post, we have answered the question of what an electron is and what its charge is. We hope that this has provided you with a basic understanding of this fundamental particle of matter. If you have any questions or comments, please feel free to leave them below and we’ll get back to you.
Thanks for reading! If you’re looking to learn more about other subjects that will help you ace your next exam, then the Noon App is a great app. With over 10,000 lectures on different topics, it’s easy to find the information and resources you need to improve your knowledge. Teachers from all over the world have put their skills and experience into this app, so you can be sure that what you learn will be of high quality. So why wait? Sign up today and get started learning!


