Alcohol is a widely used substance and is present in many aspects of our everyday lives. From the food we eat to the beverages we drink, alcohol plays a big role in our culture. But do you know what the boiling point of alcohol is?
In this blog post, we’ll explore the boiling points of different types of alcohol and how they can be used in different applications.
We’ll also discuss why it is important to understand the boiling points of alcohol and how it can affect the outcome of your projects. Read on to learn more!
What is Alcohol?
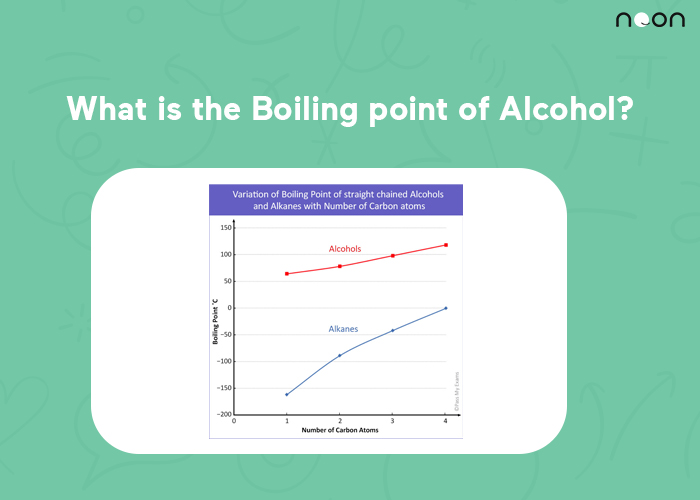
The boiling point of alcohol is the temperature at which the liquid will change into a gas. The boiling point of ethanol, the type of alcohol found in alcoholic beverages, is 78.3 degrees Celsius (173.14 degrees Fahrenheit). The alcohol formula is C2H6O.
You must be wondering, is alcohol polar or nonpolar? The answer is Polar.
When water boils, it turns into steam. The steam is made up of tiny water droplets that are suspended in the air.
Alcohol also boils and turns into vapor, but the vapor is made up of much larger molecules than steam.
These larger molecules make it more difficult for the vapor to escape from the liquid. That’s why alcohol has a lower boiling point than water.
The boiling point of a substance is affected by many factors, including atmospheric pressure and the presence of other chemicals.
For example, when you are at a higher altitude, the atmospheric pressure is lower and it takes less heat to make liquids boil. That’s why water boils at a lower temperature in Denver than it does at sea level.
How does alcohol affect the boiling point of water?
When water is heated, it begins to evaporate at 212 degrees Fahrenheit. However, the addition of alcohol lowers the boiling point of water.
The more alcohol that is present, the lower the boiling point will be. For example, if there is 50% alcohol and 50% water, the boiling point will be lower than 212 degrees Fahrenheit.
What is the boiling point of Alcohol?
The boiling point of alcohol depends on the type of alcohol. For example, ethanol has a boiling point of 78.37 degrees Celsius, whereas methanol has a boiling point of 64.68 degrees Celsius. The boiling points of other alcohols fall somewhere in between these two extremes.
The reason that different types of alcohol have different boiling points is the way that the molecules are structured.
The molecules of ethanol (and other similar alcohols) are held together by weak bonds, which means that they require less energy to break apart and turn into vapor.
Methanol (and other similar alcohols), on the other hand, have strong bonds holding their molecules together, meaning that more energy is required to break them apart and turn them into vapor.
So, if you want to know the boiling point of a particular type of alcohol, you just need to look up its molecular structure and find out how strong its bonds are.
Conclusion
The boiling point of alcohol depends on the type of alcohol and its concentration. While most pure types of alcohol have a boiling point that is lower than 100°C, it is important to note that alcoholic beverages such as wine, beer, and spirits can vary significantly in their boiling points due to their different levels of ethanol content.
By understanding the various factors that determine the boiling point of different types of alcohols, we can better understand why certain drinks require specific temperatures for proper preparation or distillation.
Noon is an application that can help you learn more about other subjects to ace your next exam.
With over 10,000 lectures on different subjects, Noon provides you with the opportunity to learn from some of the best teachers from all around the globe.
Not only does it have a wide range of topics covered, but the quality of each lecture is top-notch, making sure that you’re getting the most out of each learning experience.
In addition, Noon also comes with a number of tools for online teaching, making it the perfect assistant for any e-learning needs you may have. If you’re looking for a way to improve your grades and increase your knowledge base, then be sure to check out Noon today!


