Blog
Calculate the vapour pressure of water for this solution and its relative lowering.
Solution 9: \begin{tabular}{l} \hline $\mathrm{P} \square=23.8 \mathrm{~mm} \mathrm{Hg}$ \\ $\mathrm{W} 2=50 \mathrm{~g}, \mathrm{M}_{2}($ urea $)=60 \mathrm{~g} \mathrm{~mol}^{-1}$ \\ $\mathrm{w}...
Heny’s law constant for 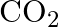 in water is
in water is 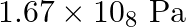 at
at 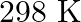 . Calculate the quantity of
. Calculate the quantity of 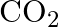 in
in 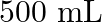 of soda water when packed under
of soda water when packed under 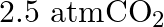 pressure at
pressure at 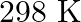 .
.
Solution 7: $$ \mathrm{KH}=1.67 \times 10_{8} \mathrm{~Pa} $$ $$ \begin{array}{l} \hline P_{A}^{o}=450 \mathrm{~mm}, P_{B}^{o}=700 \mathrm{~mm}, P_{\text {total }}=600 \mathrm{~mm} \\ \text { As...
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complexes: (i) ![Rendered by QuickLaTeX.com \mathrm{K}_{5}\left[\mathrm{Co}\left(\mathrm{C}_{2} \mathrm{O}_{4}\right)_{3}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-32cbfc6fdf62516d53f060567ba2033d_l3.png)
Ans: The complex will be $\left[\mathrm{Co}\left(\mathrm{C}_{2} \mathrm{O}_{4}\right)_{8}\right]^{3}$. Oxidation state of cobalt will be $=x-6=-3$ The oxidation state is $=+3$ Coordination number...
Discuss the nature of bonding in metal carbonyls.
Ans: The carbon metal linkages in metal carbonyls are characterized by both s and p. M-Ca bond consists of a donation into an empty metal orbital of a lone pair of electrons on the carbonyl carbon....
What is spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
Ans: In $\left[\mathrm{Cr}\left(\mathrm{NH}_{3}\right)_{6}\right]^{3 v}$ the oxidation state of $\mathrm{Cr}$ is in $+3$ oxidation state, which will cause the configuration of $\mathrm{Cr}$ as...
Using IUPAC norms Write the systematic names of the following:
(i) $\quad\left[\mathrm{CO}\left(\mathrm{NH}_{3}\right)_{6}\right] \mathrm{Cl}_{3}$ Ans: The IUPAC name of the compound is Hexaamminecobalt(III) chloride. (ii)...
Calculate the mass of urea (NH2CONH2) required in making 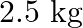 of
of 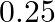 molal aqueous solution.
molal aqueous solution.
Solution 4: $0.25$ molal aqueous solution to urea means that Moles of urea $=0.25$ mole Mass of solvent $(\mathrm{NH} 2 \mathrm{CONH} 2)=60 \mathrm{~g} \mathrm{~mol}^{-1}$ $\therefore 0.25$ mole of...
Calculate the molarity of each of the following solution: (a) 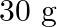 of
of 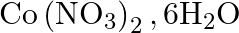 in
in 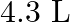 of
of
solution (b) $30 \mathrm{~mL}$ of $0.5 \mathrm{M} \mathrm{H}_{2} \mathrm{SO}_{4}$ diluted to $500 \mathrm{~mL}$. Solution 3: (a) Molar mass of $\mathrm{Co}\left(\mathrm{NO}_{3}\right)_{2}, 6...
Calculate the mole fraction of benzene in solution containing 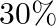 by mass in carbon tetrachloride.
by mass in carbon tetrachloride.
Solution 2: $30 \%$ by mass of $\mathrm{C} 6 \mathrm{H} 6$ in $\mathrm{CCl} 4 \Rightarrow 30 \mathrm{~g} \mathrm{C} 6 \mathrm{H} 6$ in $100 \mathrm{~g}$ solution $\therefore$ Number of moles of...
Calculate the mass percentage of benzene 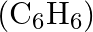 and carbon tetrachloride
and carbon tetrachloride 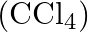 if
if 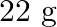 of benzene is dissolved in
of benzene is dissolved in 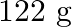 of carbon tetrachloride.
of carbon tetrachloride.
Solution Mass percentage of $$ \begin{array}{l} C_{6} H_{6}=\frac{\text { Mass of } \mathrm{C}_{6} \mathrm{H}_{6}}{\text { Total mass of the solution }} \times 100 \% \\ =\frac{\text { Mass of }...
Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous.
(i) Tetra phosphorus decoxide $\left(\mathrm{P}_{4} \mathrm{O}_{20}\right)$ (ii)Ammonium phosphate $\left(\mathrm{NH}_{4}\right)_{3} \mathrm{PO}_{4}$ (iii) $\mathrm{SiC}$ (iv) $\mathrm{I}_{2}$ (v)...
What makes a glass different from a solid such as quartz? Under what conditions could quartz be converted into glass?
Ans: The arrangement of the constituent particles makes glass different from quartz. In glass, the constituent particles have short range order, but in quartz, the constituent particles have both...
