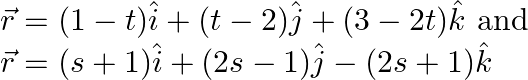Given: \[\frac{{{x}^{2}}}{{{a}^{2}}}+\frac{{{y}^{2}}}{{{b}^{2}}}=1\]…(i) Since, \[25>9\] So, above equation is of the form, \[\frac{{{x}^{2}}}{{{a}^{2}}}+\frac{{{y}^{2}}}{{{b}^{2}}}=1\]…(ii)...
Find the(v) length of the latus rectum of each of the following ellipses. ![Rendered by QuickLaTeX.com \[\frac{{{x}^{2}}}{{{a}^{2}}}+\frac{{{y}^{2}}}{{{b}^{2}}}=1\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-d32e615a6517b76e65b04fd25ae8bf0a_l3.png)
![Rendered by QuickLaTeX.com \[\frac{{{x}^{2}}}{{{a}^{2}}}+\frac{{{y}^{2}}}{{{b}^{2}}}=1\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-d32e615a6517b76e65b04fd25ae8bf0a_l3.png)