Correct option is (A) ![]()
Oleum, or fuming sulfuric acid, is a solution of various compositions of sulfur trioxide in sulfuric acid, or sometimes more specifically to disulfuric acid (also known as pyrosulfuric acid
Formula of oleum is
(A)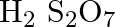
(B)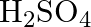
(C)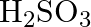
(D)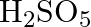
(A)
(B)
(C)
(D)
Formula of oleum is
(A)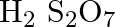
(B)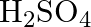
(C)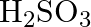
(D)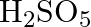
(A)
(B)
(C)
(D)
