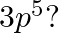(II) (a) ![]()
Complete electronic configuration: ![]()
Total no. electrons in the atom ![]() the element’s atomic number is 11
the element’s atomic number is 11
(b) ![]()
Complete electronic configuration: ![]()
Total no. electrons in the atom ![]()
![]() the element’s atomic number is 7
the element’s atomic number is 7
(c) ![]()
Complete electronic configuration: ![]()
Total no. electrons in the atom ![]()
![]() the element’s atomic number is 17
the element’s atomic number is 17
(II) What are the atomic numbers of elements whose outermost electrons are represented by (a) 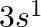 (b)
(b) 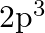 and (c)
and (c) 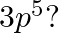
(II) What are the atomic numbers of elements whose outermost electrons are represented by (a) 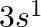 (b)
(b) 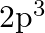 and (c)
and (c)