Blog
Calculate the overall complex dissociation equilibrium constant for the 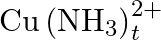 ion, given that
ion, given that 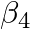 for this complex is
for this complex is 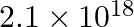 .
.
Ans: We are given the overall stability constant $\left(\beta_{s}\right)=2.1 \times 10^{13}$. The overall complex dissociation equilibrium constant is the reciprocal of the overall stability...
Write the formulas for the following coordination compounds: (i) Tetraamminediaquacobalt (III) chloride
Ans: The formula of Tetraamminediaquacobalt (III) chloride is $$ \left[\mathrm{Co}\left(\mathrm{H}_{2} \mathrm{O}\right)\left(\mathrm{NH}_{3}\right)_{4}\right] \mathrm{Cl}_{3} $$ (ii) Potassium...
A solution of glucose in water is labelled as 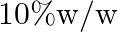 , that would be the molality and mole fraction of each component in the solution? If the density of solution is
, that would be the molality and mole fraction of each component in the solution? If the density of solution is 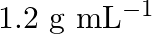 then what shall be the molarity of the
then what shall be the molarity of the
Solution 10 percent $w / w$ solution of glucose in water means $10 g$ glucose and $90 \mathrm{~g}$ of water. $10 \mathrm{~g}$ of glucose $=\frac{10}{180}=0.0555$ moles And $90 \mathrm{~g}$ of...
Density of solution 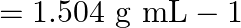 given Volume of
given Volume of
solution $-\frac{\text { mass }}{\text { density }}=\frac{100}{1.504}=66.5 \mathrm{~mL}$ Molarity of solution $$ \begin{array}{l} =\frac{\text { moles of sloution } \times 1000}{\text { Volume of...
Concentrated nitric acid used in laboratory work is 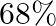 nitric acid by mass in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solution is
nitric acid by mass in aqueous solution. What should be the molarity of such a sample of the acid if the density of the solution is 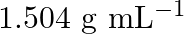 ?
?
Solution 4: $68 \%$ nitric acid by mass means that $68 \mathrm{~g}$ mass of nitric acid is dissolved in $100 \mathrm{~g}$ mass the solution. Molar mass of $\mathrm{HNO}_{3}=63 \mathrm{~g}...
Define the following terms:
(i) Mole fraction
(ii) Molality
(iii) Molarity
Solution 3: (i) Mole Fraction: It is defined as the ratio of the number of moles of the solute to the total number of moles in the solution. If $\mathrm{A}$ is the number of moles of solute...
Give an example of a solid solution in which the solute is a gas.
Solution Solution of hydrogen in palladium and dissolved gases in minerals
Define the term solution. How many types of solutions are formed? Write briefly about each type with an example.
Solution: A Solution is a homogeneous mixture of two or more chemically non-reacting substances. Types of solution: There are nine types of solution. Types of Solution Examples Gaseous solutions:...
Calculate the osmotic pressure in pascals exerted by solution prepared by dissolving 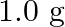 of polymer of molar mass 185,000 in
of polymer of molar mass 185,000 in 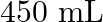 of water at
of water at 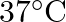 .
.
Solution $$ \begin{array}{l} \text { Given } \mathrm{V}=450 \mathrm{~mL}=0.45 \mathrm{~L} \\ =37^{\circ} \mathrm{C}=310 \mathrm{~K} \\ \mathrm{R}=8.314 \mathrm{kPaL} \mathrm{k}^{-1}...
Boiling point of water at 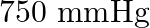 is
is 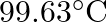 . How much sucrose is to be added to
. How much sucrose is to be added to 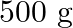 of water such that it boils at
of water such that it boils at 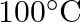 .
.
Solution Given $\Delta \mathrm{Tb}=100-99.63=3.37^{\circ}$ Mass of water w1 $=500 \mathrm{~g}$ Molar mass of water, $\mathrm{M}_{1}=18 \mathrm{~g} \mathrm{~mol}^{-1}$ Molar mass of sucrose,...
How can you determine the atomic mass of an unknown metal if you know its density and the dimension of its unit cell? Explain.
Ans: By knowing the density of an unknown metal and the dimension of its unit cell, the atomic mass of the metal can be determined. Let 'a' be the edge length of a unit cell of a crystal, 'd' be the...
What is meant by the term coordination number?
Ans: The number of nearest neighbors of any constituent particle present in the crystal lattice is called its coordination number.
