Blog
How you distinguish between the following pairs of terms:
(i) Hexagonal close-packing and cubic close-packing? Ans: A 2-d hexagonal close-packing contains two types of triangular voids (a and b) as shown in figure 1. Let us call this 2-D structure as layer...
‘Stability of crystal is reflected in the magnitude of its melting point’. Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules?
Ans: Higher the melting point, greater are the intermolecular forces of attraction between the atoms of a molecule and greater is the stability of that molecule. A substance with higher melting...
Niobium crystallises in body-centered cubic structure. If density is 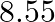
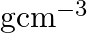 . Calculate the atomic radius of niobium using its atomic mass
. Calculate the atomic radius of niobium using its atomic mass 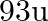 .
.
Ans: It is given that the density of niobium, $\mathrm{d}=8.55 \mathrm{gcm}^{-3}$ Atomic mass, $M=93 \mathrm{gmol}^{-1}$ As the lattice is bcc type, the number of atoms per unit cell, $z=2$ Let the...
A cubic solid is made of two elements 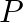 and
and 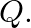 Atoms of
Atoms of 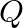 are at the corners of the cube and
are at the corners of the cube and 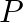 at the body-centre. What is the formula of the compound? What are the coordination numbers of
at the body-centre. What is the formula of the compound? What are the coordination numbers of 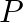 and
and 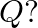
Ans: It is given that the atoms of $\mathrm{Q}$ are present at the corners of the cube. Therefore, number of atoms of $Q$ in one unit cell $=8 \times(1 / 8)=1$ It is also given that the atoms of...
Silver crystallises in the fcc lattice. If edge length of the cell is 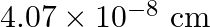 and density is
and density is 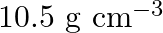 , calculate the atomic mass of silver.
, calculate the atomic mass of silver.
Ans: It is given that the edge length, $\mathrm{a}=4.07 \times 10^{-8} \mathrm{~cm}$ and density is $\mathrm{d}=$ $10.5 \mathrm{~g} \mathrm{~cm}^{3}$ As the lattice is foc type, the number of atoms...
Unit of electric potential is i) joule/coulomb ii) joule-coulomb iii) volt/metre iv) joule/coulomb-metre.
Correct option is (i)
Distinguish between electric potential and electric potential energy.
Electric potential at a place in an electric field is the amount of effort done to bring the unit positive charge from infinity to that point, whereas electric potential energy is the energy...
Stability of crystal is reflected in the magnitude of its melting point’. Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules?
Ans: Higher the melting point, greater are the intermolecular forces of attraction between the atoms of a molecule and greater is the stability of that molecule. A substance with higher melting...
Niobium crystallises in body-centered cubic structure. If density is 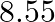
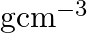 . Calculate the atomic radius of niobium using its atomic mass
. Calculate the atomic radius of niobium using its atomic mass 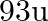 .
.
Ans: It is given that the density of niobium, $\mathrm{d}=8.55 \mathrm{gcm}^{-3}$ Atomic mass, $M=93 \mathrm{gmol}^{-1}$ As the lattice is bcc type, the number of atoms per unit cell, $z=2$ Let the...
A cubic solid is made of two elements 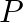 and
and 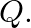 Atoms of
Atoms of 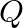 are at the corners of the cube and
are at the corners of the cube and 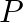 at the body-centre. What is the formula of the compound? What are the coordination numbers of
at the body-centre. What is the formula of the compound? What are the coordination numbers of 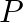 and
and 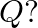
Ans: It is given that the atoms of $\mathrm{Q}$ are present at the corners of the cube. Therefore, number of atoms of $Q$ in one unit cell $=8 \times(1 / 8)=1$ It is also given that the atoms of...
Silver crystallises in the fcc lattice. If edge length of the cell is 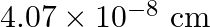 and density is
and density is 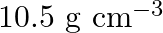 , calculate the atomic mass of silver.
, calculate the atomic mass of silver.
Ans: It is given that the edge length, $\mathrm{a}=4.07 \times 10^{-8} \mathrm{~cm}$ and density is $\mathrm{d}=$ $10.5 \mathrm{~g} \mathrm{~cm}^{3}$ As the lattice is foc type, the number of atoms...
Niobium crystallises in body-centered cubic structure. If density is 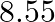
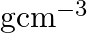 . Calculate the atomic radius of niobium using its atomic mass
. Calculate the atomic radius of niobium using its atomic mass 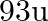 .
.
Ans: It is given that the density of niobium, $\mathrm{d}=8.55 \mathrm{gcm}^{-3}$ Atomic mass, $M=93 \mathrm{gmol}^{-1}$ As the lattice is bcc type, the number of atoms per unit cell, $z=2$
