(i) Balance the given equation: $N_{ 2 }\;(g) \; + \; 3H_{ 2 } \;(g) \; \rightarrow \; 2NH_{ 3 }\;(g) $ Thus, 1 mole (28 g) of N2 reacts with 3 mole (6 g) of H2 to give 2 mole (34 g)...
Dinitrogen and dihydrogen react with each other to produce ammonia according to the following chemical equation: N2 (g) + H2(g)→ 2NH3 (g) (i) Calculate the mass of
How are 0.50 mol 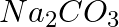 and 0.50 M
and 0.50 M 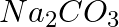 different?
different?
Molar mass of $Na_{ 2 }CO_{ 3 }$ = (2 × 23) + 12 + (3 × 16) = $106 g mol^{ -1 }$ 1 mole of $Na_{ 2 }CO_{ 3 }$ means 106 g of $Na_{ 2 }CO_{ 3 }$ Therefore, 0.5 mol of $Na_{ 2 }CO_{ 3 }$...
If 10 volumes of dihydrogen gas reacts with five volumes of dioxygen gas, how many volumes of water vapour would be produced?
2H2(g) +O2(g) →2H2O(g) 2 volumes of dihydrogen react with 1 volume of dioxygen to produce two volumes of vapour. Hence, 10 volumes of dihydrogen will react with five volumes of dioxygen to...
Which one of the following will have the largest number of atoms? (i) 1 g Au (s) (ii) 1 g Na (s) (iii) 1 g Li (s) (iv) 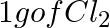 (g)
(g)
(i) 1 g Au (s) = $\frac{ 1 }{ 197 }$ mol of Au (s) = $\frac{ 6.022 \; \times \; 10^{ 23 } }{ 197 }$ atoms of Au (s) = $3.06 \times \; 10^{ 21 }$ atoms of Au (s) (ii) 1 g Na (s) = $\frac{ 1...
Calculate the molarity of a solution of ethanol in water, in which the mole fraction of ethanol is 0.040 (assume the density of water to be one).
Mole fraction of $C_{ 2 }H_{ 5 }OH$ = $\frac{Number \; of \; moles \; of \; C_{ 2 }H_{ 5 }OH}{Number \; of \; moles \; of \; solution}$ $0.040 = \frac{n_{C_{ 2 }H_{ 5 }OH}}{n_{C_{ 2 }H_{ 5 }OH} \; +...
What will be the mass of one 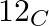 atom in g?
atom in g?
1 mole of carbon atoms =$ 6.023 \; \times \; 10^{ 23 }$atoms of carbon = 12 g of carbon Therefore, mass of $1 _{}^{ 12 }\textrm{ C }$ atom = $\frac{ 12 \; g }{ 6.022 \; \times \; 10^{ 23 }}$...
How many significant figures should be present in the answer of the following calculations? (i)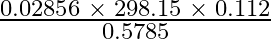 (ii) 5 × 5.365 (iii) 0.012 + 0.7864 + 0.0215
(ii) 5 × 5.365 (iii) 0.012 + 0.7864 + 0.0215
(i) $\frac{ 0.02856 \; \times \; 298.15 \; \times \; 0.112}{ 0.5785 }$ Least precise no. of calculation = 0.112 Therefore, no. of significant numbers in the answer = No. of significant numbers in...
Use the data given in the following table to calculate the molar mass of naturally occurring argon isotopes:
Molar mass of Argon: =$ [( 35.96755 \; \times \; \frac{ 0.337 }{ 100 }) + ( 37.96272 \; \times \; \frac{ 0.063 }{ 100 })+ ( 39.9624 \; \times \; \frac{ 99.600 }{ 100 })]$ =$ [0.121 + 0.024 + 39.802]...
Calculate the number of atoms in each of the following (i) 52 moles of Ar (ii) 52 u of He (iii) 52 g of He
(i) 52 moles of Ar 1 mole of Ar = $6.023 \; \times \; 10^{ 23 }$atoms of Ar Therefore, 52 mol of Ar = 52 × $6.023 \; \times \; 10^{ 23 }$atoms of Ar = $3.131 \; \times \; 10^{ 25 }$ atoms of Ar...
Calcium carbonate reacts with aqueous HCl to give CaCl2 and CO2 according to the reaction, CaCO3 (s) + 2 HCl (aq) → CaCl2(aq) + CO2 (g) + H2O(l) What mass of CaCO3 is required to react completely with 25 mL of 0.75 M HCl?
0.75 M of HCl ≡ 0.75 mol of HCl are present in 1 L of water ≡ $[(0.75 mol)\times(36.5 g mol–1 )]$ HCl is present in 1 L of water ≡ 27.375 g of HCl is present in 1 L of water Thus, 1000 mL of...
Chlorine is prepared in the laboratory by treating manganese dioxide (MnO2) with aqueous hydrochloric acid according to the reaction: 4 HCl (aq) + MnO2(s) → 2H2O (l) + MnCl2(aq) + Cl2 (g) How many grams of HCl react with 5.0 g of manganese dioxide?
1 mol of $MnO_{2}$ = 55 + 2 × 16 = 87 g 4 mol of HCl = 4 × 36.5 = 146 g 1 mol of $MnO_{2}$ reacts with 4 mol of HCl 5 g of $MnO_{ 2 }$will react with: =$ \frac{146 \; g}{87 \; g} \; \times \; 5...
In a reaction A + B2 → AB2 Identify the limiting reagent, if any, in the following reaction mixtures. (i) 300 atoms of A + 200 molecules of B (ii) 2 mol A + 3 mol B (iii) 100 atoms of A + 100 molecules of B (iv) 5 mol A + 2.5 mol B (v) 2.5 mol A + 5 mol B
Reagent limitation: It establishes the magnitude of a reaction. It is the first to be consumed in a reaction, causing the process to come to a halt and limiting the number of products produced. (i)...
If the speed of light is 3.0 × 10^8 m s^(–1), calculate the distance covered by light in 2.00 ns
Time taken = 2 ns = $2 \times10^{ -9 }$ s Now, Speed of light =$3 \times10^{ 8 } ms^{ -1 }$ So, Distance travelled in 2 ns = speed of light * time taken =$(3 \times10^{ 8 })(2 \times10^{ -9 })$ = $6...
The following data are obtained when dinitrogen and dioxygen react together to form different compounds:;(a) Which law of chemical combination is obeyed by the above experimental data? Give its statement. (b) Fill in the blanks in the following conversions: (i) 1 km = …………………. mm = …………………. pm (ii) 1 mg = …………………. kg = …………………. ng (iii) 1 mL = …………………. L = …………………. dm3
(a) If the mass of N2 is set at 28 g, the mass of O2 that will combine with it is 32 grammes, 64 grammes, 32 grammes, and 80 grammes. O2 has a mass-to-number ratio of 1: 2: 1: 5. As a result, the...
Round up the following upto three significant figures: (a) 34.216 (b) 10.4107 (c)0.04597 (d)2808
(a) The number after round up is: 34.2 (b) The number after round up is: 10.4 (c)The number after round up is: 0.0460 (d)The number after round up is: 2808
How many significant figures are present in the following? (a) 0.0027 (b) 209 (c) 6005 (d) 136,000 (e) 900.0 (f) 2.0035
(i) 0.0027: 2 significant numbers. (ii) 209: 3 significant numbers. (iii) 6005: 4 significant numbers. (iv) 136,000:3 significant numbers. (v) 900.0: 4 significant numbers. (vi) 2.0035: 5...
Express the following in the scientific notation: (i) 0.0048 (ii) 234,000 (iii) 8008 (iv) 500.0 (v) 6.0012
(a$) 0.0048= 4.8 \times10^{-3}$ (b) $234,000 = 2.34 \times10^{5}$ (c) $8008= 8.008 \times10^{3}$ (d) $500.0 = 5.000 \times10^{2}$ (e) $6.0012 = 6.0012 \times10^{0}$
A sample of drinking water was found to be severely contaminated with chloroform, CHCl3, supposed to be carcinogenic in nature. The level of contamination was 15 ppm (by mass). (i) Express this in per cent by mass. (ii) Determine the molality of chloroform in the water sample.
(a) $1 ppm = 1 part out of 1 million parts.$ Mass percent of 15 ppm chloroform in H2O= $\frac{15}{{{10}^{6}}}\times 100$ $\approx 1.5 \times{10}^{-3}%$ $ (b)$100 grams of the sample is having 1.5 ×...
What do you mean by significant figures?
The meaningful numbers that are known with certainty are known as significant figures. Significant numbers imply that the tested value is unknown. For example, if the experiment yielded 15.6 mL, 15...
Match the following prefixes with their multiples:
What is the SI unit of mass? How is it defined?
Kilogram is the SI unit of mass (kg) Mass: “Mass is defined as the mass equivalent to the mass of the international kilogramme prototype.”
Pressure is determined as force per unit area of the surface. The SI unit of pressure, pascal is as shown below: 1Pa = 1N m–2 If mass of air at sea level is 1034 g cm–2, calculate the pressure in pascal
As per definition, pressure is force per unit area of the surface. $P = \frac{F}{A}$ =$ \frac{1034\;g\;\times \;9.8\;ms^{-2}}{cm^{2}}\times \frac{1\;kg}{1000\;g}\times \frac{(100)^{2}\;cm^{2}}{1...
If the density of methanol is 0.793 kg 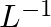 , what is its volume needed for making 2.5 L of its 0.25 M solution?
, what is its volume needed for making 2.5 L of its 0.25 M solution?
$Molar mass of CH_{3}OH$ = (1 * 12) + (4 * 1) + (1 * 16) =$32 g mol^{-1}$ = $0.032 kg mol^{-1}$ Molarity of the solution = $\frac{0.793\;kg\;L^{-1}}{0.032\;kg\;mol^{-1} }$ = 24.78 mol{L}^{-1}...
What is the concentration of sugar in mol
in mol  if its 20 g are dissolved in enough water to make a final volume up to 2L?
if its 20 g are dissolved in enough water to make a final volume up to 2L?
Molarity (M) is as given by, = $\frac{Number\;of\;moles\;of\;solute}{Volume\;of\;solution\;in\;Litres}$ =$\frac{\frac{Mass\;of\;sugar}{Molar\;mass\;of\;sugar}}{2\;L}$ =...
In three moles of ethane (C2H6), calculate the following: (i) Number of moles of carbon atoms. (ii) Number of moles of hydrogen atom (iii) Number of molecules of ethane
(a) 1 mole ${{C}_{2}}{{H}_{6}}$ contains two moles of C- atoms. ∴∴ No. of moles of C- atoms in 3 moles of ${{C}_{2}}{{H}_{6}}$ = 2 * 3 = 6 (b) 1 mole ${{C}_{2}}{{H}_{6}}$ contains six moles of H-...
Calculate the atomic mass (average) of chlorine using the following data:
Average atomic mass of Cl. = $[(\text{Fractional abundance of }\!\!~\!\!\text{ }_{{}}^{35}Cl)(\text{molar mass of }\!\!~\!\!\text{ }_{{}}^{35}Cl)+(\text{fractional abundance of }\!\!~\!\!\text{...
Determine the molecular formula of an oxide of iron, in which the mass percent of iron and oxygen are 69.9 and 30.1, respectively.
Mass percent of Fe = 69.9% Mass percent of O = 30.1% No. of moles of Fe present in oxide =$ \frac{69.90}{55.85}$ = 1.25 No. of moles of O present in oxide =$\frac{30.1}{16.0}$ =1.88 Ratio of Fe to...
How much copper can be obtained from 100 g of copper sulphate (CuSO4)?
1 mole of $CuSO_{4}$ contains 1 mole of Cu. Molar mass of $CuSO_{4}$ = (63.5) + (32.00) + 4(16.00) = 63.5 + 32.00 + 64.00 = 159.5 grams 159.5 grams of $CuSO_{4}$ contains 63.5 grams of Cu....
Calculate the mass of sodium acetate 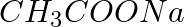 required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol–1.
required to make 500 mL of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol–1.
0.375 Maqueous solution of $CH_{3}COONa$ = 1000 mL of solution containing 0.375 moles of $CH_{3}COONa$ Therefore, no. of moles of $CH_{3}COONa$ in 500 mL = 0.1875 mole Molar mass of sodium acetate...
Calculate the amount of carbon dioxide that could be produced when (i) 1 mole of carbon is burnt in air. (ii) 1 mole of carbon is burnt in 16 g of dioxygen. (iii) 2 moles of carbon are burnt in 16 g of dioxygen.
(i) 1 mole of carbon is burnt in air. $C+{{O}_{2}}\to C{{O}_{2}}$ 1 mole of carbon reacts with 1 mole of O2 to form one mole of CO2. Amount of $CO_{2}$ produced = 44 g (ii) 1 mole of carbon...
Determine the empirical formula of an oxide of iron, which has 69.9% iron and 30.1% dioxygen by mass.
Percentage of Fe by mass = 69.9% [As said previously] By mass, 30.1 percent of O2 is present. [As said previously] Relative moles of Fe in iron oxide: = (69.9)x(55.85}/55.8569.9 = 1.25 Relative...
Calculate the mass per cent of different elements present in sodium sulphate 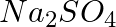
Now for Na2SO4. Molar mass of Na2SO4 = [(2 x 23.0) + (32.066) + 4(16.00)] =142.066 g Therefore, mass percent of the sodium element: = 32.379 = 32.4% Mass percent of the sulphur element: = 22.57...
Calculate the molar mass of the following: (i) 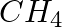 (ii)
(ii)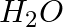 (iii)
(iii)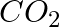
(i)Molecular weight of methane= (1 x Atomic weight of carbon) + (4 x Atomic weight of hydrogen) = [1(12.011 u) +4 (1.008u)] = 12.011u + 4.032 u = 16.043 u (ii)Molecular weight of water= (2 x...
A box contains some identical red coloured balls, labelled as A, each weighing 2 grams. Another box contains identical blue coloured balls, labelled as B, each weighing 5 grams. Consider the combinations AB, AB2, A2B and A2B3 and show that the law of multiple proportions is applicable.
AB ab2 A,B A2B3 Mass of A (in g) 2 2 4 415 Mass of B (in g) 5 10 5 According to the law of multiple proportions, Masses of B combines with a constant mass of A are 10g, 20g, 5g, 15g Simple...
Assertion (A): Combustion of 16 g of methane gives 18 g of water. Reason (R): In the combustion of methane, water is one of the products. (i) Both A and R are true but R is not the correct explanation of A. (ii) A is true but R is false. (iii) A is false but R is true. (iv) Both A and R are false.
Correct Answer: (iii) A is false but R is true Explanation: Combustion of 16 g of methane gives 36 g of water.
Assertion (A): Significant figures for 0.200 is 3 whereas for 200 it is 1. Reason (R): Zero at the end or right of a number are significantly provided they are not on the right side of the decimal point. (i) Both A and R are true and R is the correct explanation of A. (ii) Both A and R are true but R is not a correct explanation of A. (iii) A is true but R is false. (iv) Both A and R are false.
Correct Answer: (iii) A is true but R is false Explanation: Zero at the end of a number without the decimal point is not significantly based on the rate of accuracy.
The reactant which is entirely consumed in the reaction is known as limiting reagent. In the reaction 2A + 4B → 3C + 4D, when 5 moles of A react with 6 moles of B, then (i) which is the limiting reagent? (ii) calculate the amount of C formed?
(i) The reactant B is the the limiting reagent. (ii) Calculation: Let us consider that the reactant B got completely consumed as it is the limiting reagent. 4 mol of B gives 3 mol of C 6 mol of B...
The density of 3 molal solutions of NaOH is 1.110 g mL–1. Calculate the molarity of the solution.
Calculation: 3 molal solution of NaOH = 3 moles of NaOH dissolved in water 3 mole of NaOH = 120g Density of solution = 1.110gmL-1 Volume = mass/density Volume = 1120g/1.110gmL-1 Volume =1.009L...
Calculate the average atomic mass of hydrogen using the following data :
Isotope % Natural abundance Molar mass 1H 99.985 1 2H 0.015 2 Calculation: Average atomic mass of Hydrogen \[=099.985\times 1+\frac{0.015\times 2}{100}\] ...
45.4 L of dinitrogen reacted with 22.7 L of dioxygen and 45.4 L of nitrous oxide was formed. The reaction is given below:
2N2(g) + O2(g) → 2N2O(g) Which law is being obeyed in this experiment? Write the statement of the law? Answer: The Gay-Lussac’s law is used in the given reaction. The Gay-Lussac’s law states...
Which of the following terms is unitless?
(i) Molality (ii) Molarity (iii) Mole fraction (iv) Mass per cent Answer: Correct Answers: (iii) Mole fraction; (iv) Mass per cent Explanation: Unit of Molality - Molal or moles per kg Unit...
16 g of oxygen has the same number of molecules as in
(i) 16 g of CO (ii) 28 g of N2 (iii) 14 g of N2 (iv) 1.0 g of H2 Answer: Correct Answers: (iii) 14 g of N2; (iv) 1.0 g of H2 Explanation: The no. of molecules in N2 is 0.5 × 6.023 × 1023 The...
What will be the molality of the solution containing 18.25 g of HCl gas in 500 g of water? (i) 0.1 m (ii) 1 M (iii) 0.5 m (iv) 1 m
Correct Answer: (iv) 1 m Explanation: According to molality of a substance which is the number of mass of solute per mass of the solvent in liters, the molality of the solution containing 18.25 g of...
If the concentration of glucose (C6H12O6) in the blood is 0.9 g L-1, what will be the molarity of glucose in the blood? (i) 5 M (ii) 50 M (iii) 0.005 M (iv) 0.5 M
Correct Answer: (iii) 0.005 M Explanation: According to the molarity of a substance which is defined as the number of moles per volume of the solution in litres, the molarity of the glucose in blood...
The number of atoms present in one mole of an element is equal to Avogadro number. Which of the following element contains the greatest number of atoms? (i) 4g He (ii) 46g Na (iii) 0.40g Ca (iv) 12g He
Correct Answer: (iv) 12g He Explanation: The number of atoms present in 4g of Helium – 1 NA The number of atoms present in 46g of Sodium - 2 NA The number of atoms present in 0.04g of Calcium – 0.01...
If 500 mL of a 5M solution is diluted to 1500 mL, what will be the molarity of the solution obtained? (i) 1.5 M (ii) 1.66 M (iii) 0.017 M (iv) 1.59 M
Correct Answer: (ii) 1.66 M Explanation: According to the Using M1V1= M2V2 formula, the value of M2 is 1.66 M.
What will be the molarity of a solution, which contains 5.85 g of NaCl(s) per 500 mL? (i) 4 mol L-1 (ii) 20 molL-1 (iii) 0.2 molL-1 (iv) 2molL-1
Correct Answer: (iii) 0.2 molL-1 Explanation: The Molarity is defined as the number of moles per volume of the solution in litres. Given, Mass = 5.85g Molar mass = 58.5 g/mol Volume of the solution...
A measured temperature on the Fahrenheit scale is 200 °F. What will this reading be on a Celsius scale? (i) 40 °C (ii) 94 °C (iii) 93.3 °C (iv) 30 °C
Correct Answer: (iii) 93.3 °C Explanation: According to the relationship between the temperature in Fahrenheit and Celsius scale, the temperature on Celsius scale is 93.3 °C.
Two students performed the same experiment separately and each one of them recorded two readings of mass which are given below. The correct reading of mass is 3.0 g. Based on given data, mark the correct option out of the following statements. Student Readings
(i) (ii) A 3.01 2.99 B 3.05 2.95 (i) Results of both the students are neither accurate nor precise. (ii) Results of student A are both precise and accurate. (iii) Results of student B are neither...
