1.3.24 x 10-3Ms-1 2.3.24 x 10-6 Ms-1 3.6.48 x 10-6 Ms-1 4.2 x 10-3 Ms-1 Correct Answer: 3.6.48 x 10-6 Ms-1 Explanation: The rate of reaction when concentration of reactant is 2 x 10-3M is...
Calcite crystals used in Nicol’s prism are formed of
1.CaC2 2.CaCO3 3.CaCL2 4.CaO Correct Answer: 2.CaCO3 Explanation: Calcite crystals used in Nicol's prism are formed of CaCO3
H2 molecule is more stable than Li2 molecule, because
1.in H2 molecule 15 molecular orbitals are shielded by electrons are shielded by electrons. 2.In H2 bond order is one. 3.In Li2 molecule 15 molecular orbitals are shielded by electrons. 4.In Li2...
Which of the following monomers is used in manufacture of Neoprene rubber?
1.1,3-Butadien 2.styrene 3.2-chlorobuta-1,3-diene 4.Isobutylene Correct Answer: 3.2-chlorobuta-1,3-diene Explanation: 2-chlorobuta-1,3-diene monomer is used in the manufacture of Neoprene...
The unit of atomic mass, amu is replaced by u, here u stands for
1.unified mass 2.united mass 3.unique mass 4.universal mass Correct Answer: 1. unified mass Explanation: u stands for unified mass.
What is the lowest oxidation state possessed by phosphorus in its oxyacid?
1.+4 2.+2 3.+5 4.+1 Correct Answer: 4. +1 Explanation: +1 is the lowest oxidation state possessed by phosphorus in its oxyacid
What happen during bessemerization process of copper from copper pyrites?
1.Au and Ag metals are deposited as anode mud. 2.Impurities as As and Sb are removed as volatile oxides. 3.Cu is obtained by auto reduction of Cu2O and CuS. 4.Iron is removed in the form of slag....
What is the common unit of conductivity if the dimension is expressed in centimeter?
1.Ω cm-1 2.Ω-1 cm-1 3.Ω cm 4.Ω-1 cm Correct Answer: 2. Ω-1 cm-1 Explanation: Ω-1 cm-1 is the common unit of conductivity if the dimension is expressed in centimeter.
Blurring of vision is a side effect caused by the use of
1.antibiotics 2.antacides 3.tranquilizers 4.analgesics Correct Answer: 3. tranquilizers Explanation: Blurring of vision is a side effect caused by the use of tranquillizers....
What is the boiling point of heavy water?
1.100.4oC 2.101.4OC 3.273OC 4.100OC Correct Answer: 2.101.4OC Explanation: 101.4OC is the boiling point of heavy water.
What is effective atomic number of Fe in [Fe(CN)6]4- (At.no. of Fe =26)
1.34 2.26 3.36 4.35 Correct Answer: 3.36 Explanation: 36 is effective atomic number of Fe in [Fe(CN)6]4-
Which among the following elements has lowest density and is lightest?
1.Scandium 2.Cobalt 3.cpper 4.iron Correct Answer: 1. Scandium Explanation: Scandium is the element that has lowest density and is lightest.
What is the value of radius ratio of ionic crystal having coordination number six?
1.Greater than 0.732 2.in between 0.414 to 0.732 3.in between 0.225 to 0.414 4.Less than 0.225 Correct Answer: 1.in between 0.414 to 0.732 Explanation: The value of radius ratio of ionic...
What is the molar conductivity of 0.1 M NaCl its conductivity is 1.06 x 10-2Ω-1cm-1
1.1.06 x 102 Ω-1cm2 mol-1 2.1.06 x 10-2 Ω-1cm2 mol-1 3.9.4 x 10-2 Ω-1cm2 mol-1 4.5.3 x 103 Ω-1cm2 mol-1 Correct Answer: 1.1.06 x 102 Ω-1cm2 mol-1 Explanation: The molar conductivity of 0.1 M...
If a mixture of iodomethane and iodoethane is treated is treated with sodium metal in presence of dry ether it forms
1.popane and ethane 2.ethane and butane 3.propane and butane 4.ethane, propane and butane Correct Answer: 4. ethane, propane and butane Explanation: If a mixture of iodomethane and iodoethane...
Which of the following carbonyl compounds does NOT undergoes aldol condensation?
1.Acetone 2.Benzophenone 3.Acetaldehyde 4.Accetophenone Correct Answer: 2. Benzophenone Explanation: Benzophenone is the carbonyl compound which does not undergoes aldol...
Calculate the number of units cell in the 38.6g of noble having density 19.3gcm-1 and volume of one unit cell is 6.18 x 10-23cm3?
1.3.236 x 1022 2.6.180 x 1023 3.6.236 x 1020 4.3.236 x 1023 Correct Answer: 1.3.236 x 1022 Explanation: 3.236 x 1022 is the number of units cell in the 38.6g of noble having density 19.3gcm-1...
What is the percentage of formaldehyde in formalin?
1.60% 2.40% 3.10% 4.20% Correct Answer: 2.40% Explanation: The percentage of formaldehyde in formalin is 40%.
Which of the following antihistamine contain –CN group?
1.Dimetapp 2.Cimetidine 3.Terfenadine 4.Ranitidine Correct Answer: 2.Cimetidine Explanation: Cimetidine contain –CN group.
Which among the following coordination compounds does not have coordination number equal to number of ligands?
1.[pt(NH3)6]4+ 2.[Co(en)3]3+ 3.[Cu(NH3)4]2+ 4.[Co (NH3)6]3+ Correct Answer: 2.[Co(en)3]3+ Explanation: [Co(en)3]3+does not have coordination number equal to number of...
According to Andrews isothermals at What temperature the carbon dioxide gas starts to condense at 73 atmosphere?
1.21.5oC 2.30.98oC 3.13.1oC 4.48.1oC Correct Answer: 2.30.98o C Explanation: According to Andrews isothermals at 30.98oC the carbon dioxide gas starts to condense at 73...
Sodium crystallizes in bcc structure with radius 1.86 x 10-8cm.What is the edge length of unit cell of sodium?
1.4.3x 10-8 cm 2.3.72 x 10-8 cm 3.7.44 x 10-8 cm 4.5.26 x 10-8 cm Correct Answer: 1.4.3x 10-8 cm Explanation: Sodium crystallizes in bcc structure with radius 1.86 x 10-8cm. The edge length...
Which among the following reactions occurs at the zone of slag formation in extraction of iron by blast furnace?
1.C + ½ O2 → CO 2.CaO + SiO2 → CaSiO3 3.Fe2O3 + 3 CO → 2 Fe + 3CO2 4.Fe2O3 + 3C → 2 Fe + 3 CO Correct Answer: 2. CaO + SiO2 → CaSiO3 Explanation: Reaction Involved - CaO + SiO2...
In the reaction, N2 +3H2 → 2NH3, the rate of disappearance of H2 is 0.002 M/s. The rate of appearance of NH3 is
1.0.0133 M/s 2.0.023 M/s 3.0.004 M/s 4.0.032 M/s Correct Answer: 1.0.0133 M/s Explanation: In the given reaction , the rate of appearance of NH3 is 0.0133 M/s.
Identity the polymer obtained by heating n moles of isobutylene with n moles of isoprene at 1000C in presence of anhydrous AICl3
1.Butyl rubber 2.Buna-N 3.Buna-S 4.Neoprene rubber Correct Answer: 1.Butyl rubber Explanation: Butyl rubber is the polymer which is obtained by heating n moles of isobutylene with n moles of...
Which among the following gas is bubbled through the brine solution during the preparation of sodium carbonate in Solvay’s process?
1.CO2(g) 2.N2(g) 3.NO2(g) 4.O2(g) Correct Answer: 4.O2(g) Explanation: Oxygen gas is bubbled through the brine solution during the preparation of sodium carbonate in Solvay's...
Which among the following elements is a soft element as compared to others
1.CO 2.Zn 3.W 4.Mo Correct Answer: 2.Zn Explanation: Zinc is a soft element when compared to the rest of the elements.
Which of the following changes will changes will cause increase in vapour in vapour pressure of 1 molal aqueous KI solution at same temperature?
1.addition of 0.1 molal solution of NaCl 2.addition of 0.5 molal solution of Na2So4 3.addition of water 4.addition of 1 molal KI solution Correct Answer: 3. addition of water Explanation: By...
In the resonance hybrid of ozone molecule, O-O bond length is
1.128 pm 2.134.5 pm 3.121pm 4.148pm Correct Answer: 1.128 pm Explanation: In the resonance hybrid of ozone molecule, O-O bond length is 128 pm.
In the following reaction, What is the mass of KCL(s) produced ?
2KCLO3(s) 2KCL(s) + 3 O(g) H0 = -78 KJ. if 33.6L of oxygen gas is liberated at S.T.P. (at mass K=39, CL= 35.5 g mol-1) 1.48.0 g 2.7.45 g 3.24.0 g 4.74.5 g Correct Answer: 4.74.5 g...
Enthalpy of fusion of vaporization for water respectively are 6.01 Kj mol-1 and 45.07 kJ mol-1at 0degree C What is enthalpy of sublimation at 0degree C?
1.27.50 KJ mol-1 2.48.07 KJ mol-1 3.51.08 KJ mol-1 4.39.06 KJ mol-1 Correct Answer: 3.51.08 KJ mol-1 Explanation: Enthalpy of fusion of vaporization for water respectively are 6.01 Kj mol-1...
Which of the following statement is NOT correct about solution?
1.The three states of matter solid, liquid and gas may play role of either solute or solvent 2.the component of solution which constitute smaller part is called solute. 3.When water is solvent, the...
The P-P-P bond angle in white phosphorus is
1.90o 2.109o281 3.120o 4.600 Correct Answer: 4.600 Explanation: The P-P-P bond angle in white phosphorus is 4.600
Which of the following pairs of solution is isotonic? (Molar mass urea=60, sucrose=342 g mol-1)
1.3.0 gL-1 urea and 17.19 gL-1 sucrose 2.0.3 gL-1 urea and 1.719 gL-1 sucrose 3.0.gL-1 urea and 1.719 gL-1 sucrose 4.0.3 gL-1 urea and 17.19 gL-1 sucrose Correct Answer: 1.3.0 gL-1 urea and...
When alkyl halide is boiled with large excess of alcoholic ammonia it forms
1.Primary amine 2.tertiary amine 3.secondary amine 4.quatrnary ammonium salt Correct Answer: 1.Primary amine Explanation: When alkyl halide is boiled with large excess of alcoholic ammonia it...
Methoxy ethane on reaction with hot concentrated HI gives
1.iodomethane and ethanol 2.iodomethane and iodoethane 3.methanol and ethanol 4.methanol and iodoethane Correct Answer: 2.iodomethane and iodoethane Explanation: Methoxy ethane on reaction...
Which of the following oxyacid of Sulphur contains S=S linkage?
1.H2S2O4 2.H2SO3 3.H2S2O5 4.H2S2O2 Correct Answer: 4.H2S2O2 Explanation: H2S2O2 contains S=S linkage.
Identity the decreasing order of boiling point of alkanes (1)n-pentane (2) isopentane (3) Neopentane
1.Isopentene ˃ n-pentane ˃ Neopentane 2.Neopentane ˃Isopentane ˃ n-pentane 3.n-pentane ˃ Isopentane ˃ Neopentane 4.Isopentane ˃ Neopentane ˃ n-pentane Correct Answer: 3.n-pentane ˃ Isopentane...
Which of the following is a character of catalyst?
1.it changes the position of equilibrium. 2.it increases the rates of both forward and backward reaction equally in reversible reaction 3.it affects the energies of reactants and products of the...
An increase in the concentration of the reactants of a reaction leads to change in (1) heat of reaction (2) threshold energy (3) collision frequency (4) activation energy
Correct option (1) As the number of molecules per unit volume grows, so does the frequency of collisions.
Match the following and identify the correct option.
Correct option (4) CO(g) + H2(g) = Synthetic gas or water gas Bicarbonates of Ca2+ and Mg2+...
Anisole on cleavage with HI gives
Correct option (4) The methyl(phenyl) oxonium ion is formed when anisole reacts with protons from hydroiodic acid. The reaction is an SN2...
Which of the following oxoacid of sulphur has -O-O- linkage?
(1) 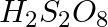 , peroxodisulphuric acid
, peroxodisulphuric acid
(2) 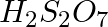 , pyrosulphuric acid
, pyrosulphuric acid
(3) 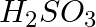 , sulphurous acid
, sulphurous acid
(4)  , sulphuric acid
, sulphuric acid
The correct option is (1) Diagrammatic representation of each compound is as follows,
What is the change in oxidation number of carbon in the following reaction? ![Rendered by QuickLaTeX.com \[CH _{4}( g )+4 Cl _{2}( g ) \rightarrow CCl _{4}( l )+4 HCl ( g )\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-80c7d3f47e40e2ab7490771786cbcebe_l3.png)
(1) 0 to 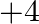
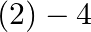 to
to 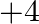
(3) 0 to 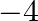
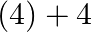 to
to 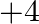
Correct option: (2) The oxidation state carbon of methane is -4 which is the reactant and that on product side is carbon tetrachloride is +4.
The mixture which shows positive deviation from Raoult’s law is
(1) Benzene + Toluene
(2) Acetone + Chloroform
(3) Chloroethane + Bromoethane
(4) Ethanol + Acetone
Correct option (4) Acetone + ethanol deviates from Raoult's law in a positive way. H-bonding exists in pure ethanol, and adding acetone to it causes some H-bonds to break. The observed vapour...
The freezing point depression constant 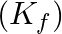 of benzene is
of benzene is 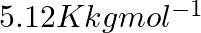 . The freezing point depression for the solution of molality
. The freezing point depression for the solution of molality 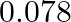 m containing a non-electrolyte solute in benzene is (rounded off upto two decimal places):
m containing a non-electrolyte solute in benzene is (rounded off upto two decimal places):
(1) 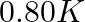 (2)
(2) 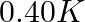 (3)
(3) 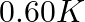 (4)
(4) 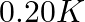
Correct option: (2) As we know that, we evaluate the value using the formula directly, $\begin{aligned} \Delta T _{ f }= i k _{ f } m \\ \Rightarrow \quad \Delta T _{ f } &=1 \times 5.12 \times...
Identify the incorrect statement.
(1) The transition metals and their compounds are known for their catalytic activity due to their ability to adopt multiple oxidation states and to form complexes.
(2) Interstitial compounds are those that are formed when small atoms like 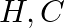 or
or 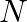 are trapped inside the crystal lattices of metals.
are trapped inside the crystal lattices of metals.
(3) The oxidation states of chromium in 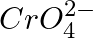 and
and 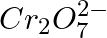 are not the same.
are not the same.
(4) 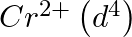 is a stronger reducing agent than
is a stronger reducing agent than 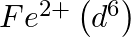 in water.
in water.
Correct option (3) Explanation: Interstitial compounds are those formed when small atoms such as H, B, C, or N are trapped inside metal crystal lattices. Fact: On the basis of standard reduction...
When a ray of light is incident normally on one refracting surface of an equilateral prism of refractive index 1.5, the emerging ray ![Rendered by QuickLaTeX.com \left[\sin ^{-1}\left(\frac{1}{1.5}\right)=41.8^{\circ}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-0caec26c6644af9c6eadbc41ca73eb61_l3.png)
A) just grazes the second refracting surface.
B) is deviated by 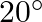 .
.
C) is deviated by 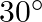 .
.
D) undergoes total internal reflection at second refracting surface.
Correct answer is D. Critical angle for the material of prism $C=\sin ^{-1}\left(\frac{1}{\mu}\right)$ $=\sin ^{-1}=42^{\circ}$ since angle of incidence at surface $A B\left(60^{\circ}\right)$ is...
For the following combination of logic gates, when all the three inputs are first high and then low, the output ‘ 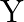 ‘ will respectively be
‘ will respectively be
A) 1,0
B) 0,0
C) 1,1
D) 0,1
Solution: Correct option is A. Here the gate $P$ is a logic symbol of AND gate and $Q$ is a NAND gate. So, the output of $P$ is $Y_{p}=A . B$ and the output of $Q$ is...
Which of the following equations shows the relationship between heat of reaction at constant pressure and heat of reaction at constant volume if the temperature is not constant?
$ 1.\,\,\Delta H-\Delta n=\Delta URT $ $ 2.\,\,\Delta H-\Delta U=\Delta nRT $ $ 3.\,\,\Delta H=\Delta nRT $ $ 4.\,\,\Delta H=\Delta U-RT $ Solution: $ 2.\,\,\Delta H-\Delta U=\Delta nRT $...
Which of the following elements is refined by zone refining?
A. Gallium B. Bismuth C. Copper D. Zinc Solution: gallium The zone refining method is generally used to refine metalloids and ultra-pure metal is obtained. This is based on the idea that impurities...
Slope of the straight line obtained by plotting 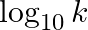 against
against 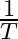 represents what term ? (A)
represents what term ? (A) 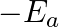 (B)
(B) 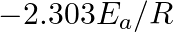 (C)
(C) 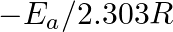 (D)
(D) 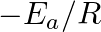
Correct option is C ( $-\mathrm{E}_{\mathrm{a}} / 2.303 \mathrm{R}$) Explanation: The Arrhenius equation is: $ \log _{10} \mathrm{k}=\log _{10} \mathrm{~A}-\frac{\mathrm{E}_{\mathrm{a}}}{2.303...
Calculate the work done during combustion of 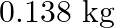 of ethanol,
of ethanol,  at at
at at 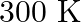 . Given :
. Given : 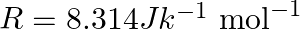 , molar mass of ethanol
, molar mass of ethanol 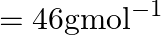 (A)
(A) 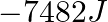 (B)
(B) 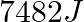 (C)
(C) 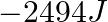 (D)
(D) 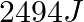
The correct option is B Explanation: The combustion of ethanol, involves the following reaction. $ C_{2} H_{5} O H(l)+3 O_{2}(g) \rightarrow 2 C O_{2}+3 H_{2} O $ Given, Mass of ethanol $=0.138...
With which halogen the reactions of alkanes are explosive ? (A) Fluorine (B) Chlorine (C) Bromine (D) Iodine
Correct answer is A (Fluorine) Explanation: The reactions of alkanes with fluorine are explosive. Fluorine is a highly reactive element exhibiting an exothermic reaction when it reacts with...
What is the geometry of water molecule ? (A) distorted tetrahedral (B) tetrahedral (C) trigonal planer (D) diagonal
Correct option is A Explanation: The tetrahedral geometry of the water molecule is deformed. The electrons in the O atom are divided into two lone pairs and two bond pairs. The sp 3 hybridisation of...
Lactic acid and glycollic acid are the monomers used for preparation of Polymer (A) Nylon2nylon6 (B) Dextron (C) PHBV (D) BunaN
Correct option is B Explanation: Dextron is a biodegradable copolymer of glycolic acid and lactic acid that contains ester linkages.
In case of R, S configuration the group having highest priority is (A) 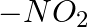 (B)
(B) 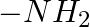 (C)
(C) 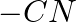
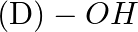
Correct option is D Explanation: The group with the highest priority in the R, S configuration is -OH. The atomic number of O is larger than that of N and C. As a result, oxygen takes priority.
Arenes on treatment with chlorine in presence of ferric chloride as a catalyst undergo what type of reaction ? (A) Electrophilic substitution (B) Nucleophilic substitution (C) Electrophilic addition (D) Nucleophilic addition
Correct option is A Explanation: Arenes undergo halogenation when exposed to chlorine in the presence of a Lewis acid catalyst, ferric chloride, or aluminium chloride, and in the absence of light....
Identify the oxidation states of titanium 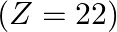 and copper
and copper  in their colourless compounds. (A)
in their colourless compounds. (A) 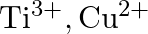 (B)
(B) 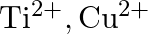 (C)
(C) 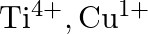 (D)
(D) 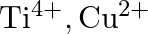
Correct option is C $\mathrm{T} \mathrm{i}^{4+}, \mathrm{Cu}^{+}$ Explanation: The oxidation states of titanium $(\mathrm{Z}=22)$ and copper $(\mathrm{Z}=29)$ in their colourless compounds are...
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule. (A) Ribulose (B) Ribose (C) Erythrose (D) Glyceraldehyde
D is the correct answer Explanation: The monosaccharide glyceraldehyde has only one asymmetric carbon atom in its molecule. An asterisk denotes an asymmetric carbon atom.
Which among the group -15 elements does NOT exists as tetra atomic molecule ? (A) Nitrogen (B) Phosphorus (C) Arsenic (D) Antimony
Correct option is A (Nitrogen) Explanation: Nitrogen does not exist as tetra atomic molecule. It exists as diatomic molecule $N_2$ that can be represented as N≡N.
The correct relation between elevation of boiling point and molar mass of solute is (A) 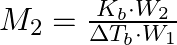 (B)
(B) 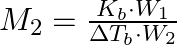 (C)
(C) 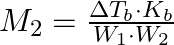 (D)
(D) 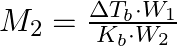
Correct option is (A) $M_{2}=\frac{K_{b} \cdot W_{2}}{\Delta T_{b} \cdot W_{1}}$ EXPLANATION: The correct relation between elevation of boiling point (ΔTb) and molar mass of solute (M2) is...
Which of the following reactions is used to prepare aryl fluorides from diazonium salts and fluoroboric acid ? (A) Sandmeyer reaction (B) BalzSchiemann reaction (C) Gattermann reaction (D) Swarts reaction
Correct option is B Balz-Schiemann reaction EXPLANATION: Balz-Schiemann reaction is used to prepare aryl fluorides from diazonium salts and fluoroboric acid. $ \mathrm{Ar}-\mathrm{N}_{2} \mathrm{X}...
The element that does NOT exhibit allotropy is (A) Phosphorus (B) Arsenic (C) Antimony (D) Bismuth
Correct option is D Explanation: Bismuth is the only element that does not exhibit allotropy. There are two allotropes of nitrogen (solid). There are three types of antimony allotropes. There are...
Conservation of hexane into benzene involves the reaction of (A) Hydration (B) Hydrolysis (C) Hydrogenation (D) Dehydrogenation
Correct option is D Explanation: Conversion of hexane into benzene involves the reaction of dehydrogenation.
Which of the following is NOT a tranquilizer ? (A) Meprobamate (B) Equanil (C) Chlordiazepoxide (D) Bromopheniiramine
Correct option is D Explanation: A tranquillizer is a medicine that is used to treat anxiety, fear, tension, agitation, and other mental problems, with the goal of reducing anxiety and tension....
Which element is obtained in the pure form by van Arkel method ? (A) Aluminium (B) Titanium (C) Silicon (D) Nickel
Correct option is B (Titanium) Explanation: The van Arkel process is used to obtain pure titanium. This procedure can also be used to obtain zirconium. Hall's procedure, Baeyer's process, or...
Which of the following polymers is used to manufacture clothes for firefighters ? (A) Thiokol (B) Kevlar (C) Nomex (D) Dynel
Correct option is C (Nomex) Explanation: Nomex is a synthetic fibre that is used to make clothing for firefighters. It's also utilised to make race car drivers' protective gear. Dynel is a synthetic...
What is the density of solution of sulphuric acid used as an electrolyte in lead accumulator? (A) 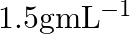 (B)
(B) 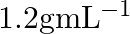 (C)
(C) 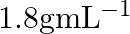 (D)
(D) 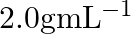
B $1.2 gmL^{-1}$ is the correct answer. Explanation: Sulphuric acid solution used as an electrolyte in a lead accumulator has a density of 1.2 gmL^-1. It is equivalent to 38 percent sulphuric acid...
Excess of ammonia with sodium hypochloride solution in the presence of glue or gelatine gives (A) 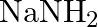 (B)
(B) 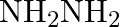 (C)
(C) 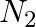 (D)
(D) 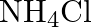
Correct option is B $\mathrm{NH}_{2} \mathrm{NH}_{2}$ Explanation: Excess of ammonia with sodium hypo chloride solution in the presence of glue or gelatine gives hydrazine $\left(\mathrm{NH}_{2}...
Which among the following elements of group-2 exhibits anomalous properties ? (A) Be (B) Mg (C) Ca (D) Ba
A is the correct answer. Explanation: Be has anomalous characteristics. This is due to beryllium's smaller atomic and ionic radii when compared to the other members of the group. It's also because...
Phenol in presence of sodium hydroxide reacts with chloroform to form salicyladehyde. The reaction is known as (A) Kolbe’s reaction (B) ReimerTiermann reaction (C) Stephen reaction (D) Etard reaction
Correct option is B Explanation: Salicyladehyde is formed when phenol interacts with chloroform in the presence of sodium hydroxide. The Reimer-Tiemann reaction is the name of the reaction.
Bauxite, the ore of aluminium, is purified by which process ? (A) Hoope’s process (B) Hall’s process (C) Mond’s process (D) Liquation process
Correct option is B Explanation: (i)The Hall's process is the most widely used industrial method for smelting aluminium. The process entails dissolving aluminium oxide in molten cryolite and...
What are the products of auto-photolysis of water? (A) 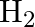 and
and 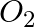 (B) Stream (C)
(B) Stream (C) 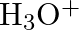 and
and 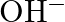 (D) Hydrogen peroxide
(D) Hydrogen peroxide
Correct Option is A Explanation: Water undergoes auto photolysis, which entails the breaking of chemical bonds as a result of the transmission of light energy to these bonds. It results in the...
In which among the following solids, Schottky defect is NOT observed ? (A) ZnS (B) NaCl (C) KCl (D) CsCl
Correct option is A (ZnS) Explanation: In ZnS, the Schottky defect does not exist. NaCl, KCl, and CsCl all have the Schottky defect. The Schottky defect occurs in solids where the cations and anions...
Two moles of an ideal gas are allowed to expand from a volume of 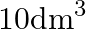 to
to 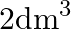 at
at 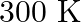 against a pressure of 101.325 KPa. Calculate the work done (A)
against a pressure of 101.325 KPa. Calculate the work done (A) 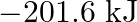 (B)
(B) 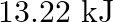 (C) -810.6kJ (D)
(C) -810.6kJ (D) 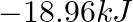
Correct option is A $-201.6 \mathrm{~kJ}$ Explanation: $ \begin{array}{l} \Delta \mathrm{V}=\mathrm{V}_{2}-\mathrm{V}_{1}=2 \mathrm{~m}^{3}-\left(10 \mathrm{dm}^{3} \times 10^{-3} \mathrm{~m}^{3} /...
Which of the following compounds does NOT undergo haloform reaction?
Correct option is C Explanation: Compounds containing methyl keto group or compounds (R−OH∣CH−CH3) that can be oxidised to methyl keto group can undergo haloform...
What is the 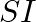 unit of density? (A)
unit of density? (A) 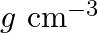 (B)
(B) 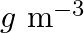 (C)
(C) 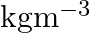 (D)
(D) 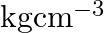
Correct option is C
The most basic hydroxide from following is (A) 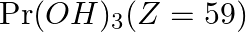 (B)
(B) 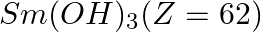 (C)
(C) 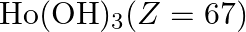 (D)
(D) 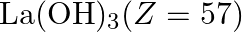
Correct option is D Explanation: The basic nature of hydroxides changes from La3+ to Lu3+ as a result of lanthanoid contraction. As a result, the covalent character of the Ln—OH connection becomes...
Which of the followings is most reactive towards addition reaction of hydrogen cynide to form corresponding cynohydrin? (A) Acetone (B) Formaldehyde (C) Acetaldehyde (D) Diethylketone
Correct option is B Explanation: Formaldehyde (HCHO) is most reactive towards addition reaction of hydrogen cyanide because it is least sterically hindered
In which substance does nitrogen exhibit the lowest oxidation state? (A) Nitrogen gas (B) Ammonia (C) Nitrous oxide (D) Nitric oxide
Correct option is B Explanation: Nitrogen exhibits lowest oxidation state in ammonia. so, option (B) is correct.
What is the number of donor atoms in dimethylglyoximato ligand? (A) 1 (B) 2 (C) 3 (D) 4
Correct option is (B) Explanation: The dimethylglyoximato ligand is a bidentate ligand. Bidentate ligands are those that have two atoms that directly coordinate with the metal atom or that donate...
Which of the followings is a tricarboxylic acid? (A) Citric acid (B) Malonic acid (C) Succinic acid (D) Malic acid
Correct option is A Explanation: Malonic acid, succinic acid and malic acid are dicarboxylic acids while citric acid is a tricarboxylic acid.
Molarity is (A) The number of moles of solute present in 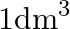 volueme of solution (B) The number of moles of solute dissolved in
volueme of solution (B) The number of moles of solute dissolved in 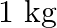 of solvent (C) The number of moles of solute dissolved in
of solvent (C) The number of moles of solute dissolved in 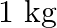 of solution (D) The number of moles of solute dissolved in
of solution (D) The number of moles of solute dissolved in 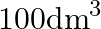 volume of solution
volume of solution
Correct option is A. (the number of moles of solute present in $1 \mathrm{dm}^{3}$ volume of solution) Explanation: Since the molarity is moles per litre and $1 \mathrm{dm}^{3}$ is equal to $1...
Which of the following carboxylic acids is most reactive towards esterification? (A) 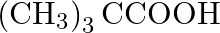 (B)
(B) 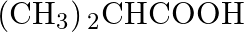 (C)
(C) 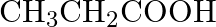 (D)
(D) 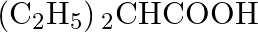
Correct option is C Explanation: Because the C atom of the carboxylic group is the least sterically hindered, $\mathrm{CH}_{3} \mathrm{CH}_{2} \mathrm{COOH}$ is the most reactive towards...
Which carbon atom of deoxy Ribose sugar in 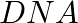 does NOT contain C-OH bond (A)
does NOT contain C-OH bond (A) 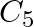 (B)
(B) 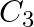 (C)
(C) 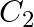 (D)
(D) 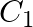
Correct option is C Deoxyribose is a five-carbon-atom pentose sugar. The ring's four carbon atoms are numbered 1', 2', 3', and 4'. - CH2OH has a carbon atom with the number 5'. At positions 1', 3',...
The molarity of urea (molar mass 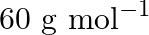 ) solution by dissolving
) solution by dissolving 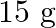 of urea in
of urea in 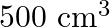 of water is (A) 2 mol
of water is (A) 2 mol 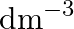 (B)
(B) 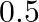 mol
mol 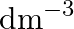 (C)
(C) 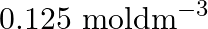 (D)
(D) 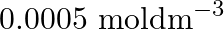
Correct option is B ($0.5 \mathrm{~mol} \mathrm{dm}^{-3}$) Explanation: The number of moles of urea $=\frac{\text { Mass of urea }}{\text { Molar mass of urea }}=\frac{15 \mathrm{~g}}{60 \mathrm{~g}...
The number of moles of electrons passed when current of 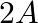 is passed through an solution of electrolyte for 20 minutes is (A)
is passed through an solution of electrolyte for 20 minutes is (A) 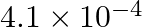 mol
mol 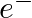 (B)
(B)  mol
mol 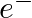 (C)
(C)  mol
mol 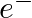 (D)
(D) 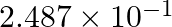 mol
mol 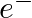
Correct option is C ($2.487 \times 10^{-2}$) mole Explanation: Current passed, $\mathrm{I}=2 \mathrm{~A}$ Time for which current is passed $\mathrm{t}=20 \mathrm{~min}=20 \mathrm{~min} \times 60...
What is the catalyst used for oxidation of 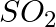 to
to 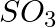 in lead chamber process for manufacture of sulphuric acid? (A) Nitric oxide (B) Nitrous oxide (C) Potassium iodide (D) Dilute
in lead chamber process for manufacture of sulphuric acid? (A) Nitric oxide (B) Nitrous oxide (C) Potassium iodide (D) Dilute 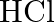
Correct option is A (Nitric oxide) Explanation: Nitric oxide (NO) is the catalyst used for oxidation of $\mathrm{SO}_{2}$ to $\mathrm{SO}_{3}$ in lead chamber process for manufacture of sulphuric...
Which of the following compounds reacts immediately with Lucas reagent? (A)  (B)
(B) 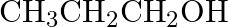 (C)
(C) 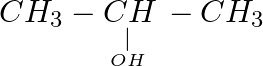 (D)
(D)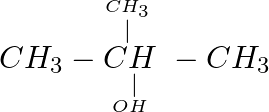
Correct option is D Explanation: A solution of anhydrous zinc chloride in strong hydrochloric acid is known as Lucas reagent. This solution is used to classify low-molecular-weight alcohols. The...
Which symbol replaces the unit of atomic mass, amu?
A)u
B)A
C)M
D)n
Correct option is A (u) Explanation: The atomic mass unit, amu, is represented by the symbol 'u.' The atomic mass of carbon-12, for example, is expressed as 12 u. As a result, option A is...
What is the oxidation number of gold in the complex ![Rendered by QuickLaTeX.com \left[A u C l_{4}\right]^{1-} ?](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-937d18e590456b146365a433cd94f370_l3.png) (A)
(A) 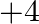 (B)
(B) 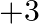 (C)
(C) 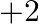 (D)
(D) 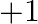
Correct option is B $+3$ Oxidation state of chlorine $\mathrm{Cl}$ is $-1$. Let the oxidation number of gold Au be $x$. $ \begin{array}{l} \therefore \mathrm{x}+4 \times(-1)=-1 \\ \Longrightarrow...
Which among the following oxoacids of phosphorus shows a tendency of disproportionation? (A) Phosphinic acid 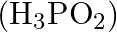 (B) Orthophosphoric acid
(B) Orthophosphoric acid 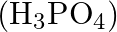 (C) Phosphinic acid
(C) Phosphinic acid 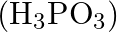 (D) Pyrophosphoric acid
(D) Pyrophosphoric acid 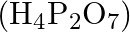
Correct option is C Phosphonic acid $\left(\mathrm{H}_{3} \mathrm{P} \mathrm{O}_{3}\right)$ Explanation: Phosphonic acid $\left(\mathrm{H}_{3} \mathrm{P} \mathrm{O}_{3}\right)$ shows a tendency of...
Which among the following metal crystallise as a simple cube? (A) Polonium (B) Iron (C) Copper (D) Gold
Correct option is A (Polonium) Explanation: Polonium forms a simple cube when crystallised. The BCC unit cell is formed when iron crystallises. The FCC unit cell is formed by the crystallisation of...
Identify the functional group that has electron donating inductive effect. (A) 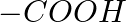 (B)
(B) 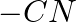 (C)
(C) 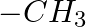 (D)
(D) 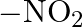
The correct answer is C. The inductive effect of $-CH 3$ is electron donating (+I). The remaining three, on the other hand, indicate an electron withdraing inductive action. As a result, the correct...
Identify the product 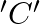 in the following reaction. Aniline
in the following reaction. Aniline 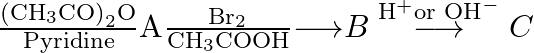 (A) Acetanilide (B)
(A) Acetanilide (B)  -Bromoacetanilide (C)
-Bromoacetanilide (C)  -Bromoaniline (D)
-Bromoaniline (D)  -Bromoaniline
-Bromoaniline
The correct answer is C. (p-Bromoaniline) Explanation: 1) The aniline's amino group is acetylated with acetic anhydride/pyridine. 2) It is then brominated with acetic acid and bromine. Because the...
Identify the element that forms amphoteric oxide. (A) Carbon (B) Zinc (C) Calcium (D) Sulphur
Correct option is B (Zinc) Explanation: Zinc form amphoteric metal oxide $\mathrm{ZnO}$. It reacts with acids as well as bases. $ \mathrm{ZnO}+2 \mathrm{HCl} \rightarrow...
Formation of 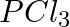 is explained on the basic of what hybridisation of phosphorus atom? (A)
is explained on the basic of what hybridisation of phosphorus atom? (A) 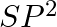 (B)
(B) 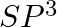 (C)
(C) 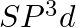 (D)
(D) 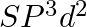
Correct option is B Explanation: It is possible to establish the type of hybridisation that occurs in an atom. Making use of the formula $ H=\frac{1}{2}[V+Y-C+A] $ Here, $V=$ valence shell electrons...
During galvanization of iron, which metal is used for coating iron surface? (A) Copper (B) Zinc (C) Nickel (D) Tin
Correct option is B (Zinc) Explanation: The process of adding a protective zinc coating to steel or iron to prevent rusting is known as galvanization or galvanising.
Which among the following equations represents the first law of thermodynamics under isobaric conditions? (A) 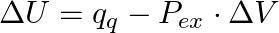 (B)
(B) 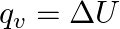 (C)
(C) 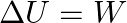 (D)
(D) 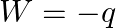 .
.
Correct option is A Explanation: According to first law of thermodynamics, $\Delta U=q+W$ In case of isobaric conditions, change of state is brought about at constant pressure, i.e. $d p=Q$...
A certain reaction occurs in two steps as (i) 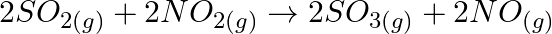 (ii)
(ii) 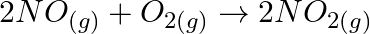 In the reaction, (A)
In the reaction, (A) 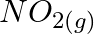 is intermediate (B)
is intermediate (B) 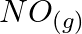 is intermediate (C)
is intermediate (C) 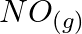 is catalyst (D)
is catalyst (D) 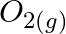 is intermediate
is intermediate
Correct option is B (NO (g) is intermediate) Explanation: Because NO (g) is created in the first stage and consumed in the second, it is considered intermediate. In the first process, a catalyst may...
Name the metal that is purified by placing the impure metal on sloping hearth of a reverberatory furnace and heating that above its melting point in absence of air. A) Mercury B) Galium C) Zirconium D) Copper
Correct option is D (Copper) Explanation: Copper is a purified metal that is purified by heating it over its melting point in the absence of air on a sloping hearth of a reverberatory surface.
Which halogen forms an oxyacid that contains the halogen atom in tripositive oxidation state ? A) Fluorine B) Chlorine C) Bromine D) Iodine
B is the correct answer (Chlorine) The halogen atom in the chlorine halogen creates an oxyacid, $HClO_2$, which has a tripositive oxidation state. The oxidation number of chlorine in $HClO_2$ is +3.
Which among the following solutions is NOT used in determination of the cell constant ? A) 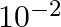 M KCl B)
M KCl B) 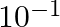 M KCl C) 1 M KCl D) Saturated KCl
M KCl C) 1 M KCl D) Saturated KCl
Correct Answer: D. Saturated $\mathrm{KCl}$ Explanation: Only the conventional solution of $\mathrm{KCl}$ is used to get the cell constant. Options (a), (b), and (c) are standard solutions, however...
A mixture of benzaldehyde and formaldehyde when treated with 50% NaOH yields A) Sodium benzoate and sodium formate B) Sodium formate and benzyl alcohol C) Sodium benzoate and methyl alcohol D) Benzyl alcohol and methyl alcohol
Correct option is B (sodium formate and benzyl alcohol) Explanation: When benzaldehyde and formaldehyde are treated with 50% NaOH, sodium formate and benzyl alcohol are formed. This is referred to...
Which of the following proteins is globular ? A) Collagen B) Albumin C) Myosin D) Fibroin
Correct option is B (Albumin) Explanation: Unlike fibrous or membrane proteins, globular proteins are relatively water soluble (forming colloids in water). Albumin is a protein that is globular in...
Given 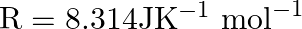 , the work done during combustion of
, the work done during combustion of 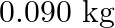 of ethane (molar mass
of ethane (molar mass 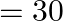 ) at
) at 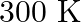 is A)
is A) 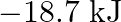 B)
B) 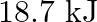 C)
C) 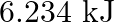 D)
D) 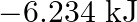
Correct option is B $18.7 \mathrm{~kJ}$ $ \begin{array}{l} \mathrm{C}_{2} \mathrm{H}_{6(\mathrm{~g})}+\frac{7}{2} \mathrm{O}_{2(\mathrm{~g})} \longrightarrow...
Which among the following functional groups has been given the highest priority while assigning R-S configuration? A) 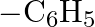 B)
B) 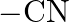 C)
C) 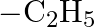 D)
D) 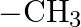
Correct option is B Explanation: Because -CN has the highest atomic number, the atom with the highest atomic number that is immediately linked to carbon will be seen first, followed by the remaining...
Which among the following group 15 element forms most stable pentavalent compound? A) Phosphorus B) Antimony C) Bismuth D) Arsenic
Correct option is A Explanation: Because of the availability of d-electrons, phosphorus is a 15th group element that forms the most stable pentavalent compound. The stability of the +5 oxidation...
The process in which metal surface is made inactive is called A) Passivation B) Galvanizing C) Corrosion D) Pickling
Correct option is A Explanation: Passivation is the process of rendering a metal surface inert. Metal is treated with high oxidising chemicals, such as concentrated nitric acid, in this procedure....
What is the volume of water consumed during acid hydrolysis of 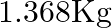 of sucrose? (Given
of sucrose? (Given  molar masses of sucrose
molar masses of sucrose 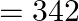 , water
, water 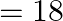 , density of water
, density of water 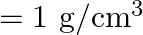 ) A)
) A) 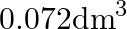 B)
B) 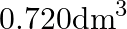 C)
C) 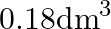 D)
D) 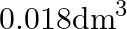
Correct option is A ($0.072 \mathrm{dm}^{3}$) $ \mathrm{C}_{12} \mathrm{H}_{22} \mathrm{O}_{11}+\mathrm{H}_{2} \mathrm{O} \stackrel{\mathrm{H}^{+}}{\longrightarrow} \mathrm{C}_{6} \mathrm{H}_{12}...
The correct order of reactivity of aldehydes and ketones towards hydrogen cyanide is A) 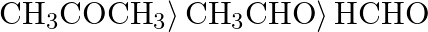 B)
B) 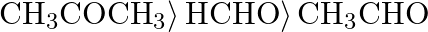 C)
C) 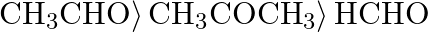 D)
D) 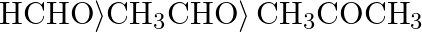
Correct option is D) $\left.\mathrm{HCHO}\rangle \mathrm{CH}_{3} \mathrm{CHO}\right\rangle \mathrm{CH}_{3} \mathrm{COCH}_{3}$ The following is the right order of reactivity of aldehydes and ketones...
What is the basicity of orthophosphorus acid ? A) One B) Two C) Three D) Four
B is the correct answer The basicity of orthophosphorous acid is two due to the presence of two - OH groups in its structure.
Identify the heteropolymer from the list given below. A) Polythene B) Nylon-6 C) Teflon D) Nylon-6, 6
D is the correct answer (Nylon-6, 6) A heteropolymer is one that is made up of two or more distinct (though often related) monomers. Hexamethylenediamine and adipic acid, which give nylon 66 its...
What is the chemical composition of Nicol’s prism? A) 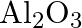 B)
B) 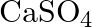 C)
C) 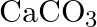 D)
D) 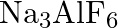
Correct option is C Explanation: A Nicol prism is a type of polarizer, which is an optical device that produces a polarised light beam. It is constructed in such a way that total internal reflection...
Identify a metalloid from the following list of elements. A) Carbon B) Neon C) Sodium D) Tellurium
Correct option is D Explanation: Tellurium is a silvery-white metalloid that possesses both metal and non-metal characteristics. Sodium is a metal, while carbon and neon are non-metals. As a result,...
Select a ferromagnetic material from the followings. A) Dioxygen B) Chromium (IV) oxide C) Benzene D) Dihydrogen monoxide
Correct option is B Explanation: Chromium (IV) dioxide is a ferromagnetic substance. These compounds' ferromagnetic properties allow them to be employed in data tapes, which are used to store data....
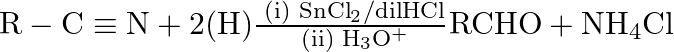 this reaction is known as A) Etard reaction B) Stephen reaction C) Hell-Vohlard-Zelinsky reaction D) Balz-Schiemann reaction
this reaction is known as A) Etard reaction B) Stephen reaction C) Hell-Vohlard-Zelinsky reaction D) Balz-Schiemann reaction
B is the correct answer. Explanation: Using tin chloride and dilution, aldehydes (R-CHO) are synthesised from nitriles (R-CN). Stephen's reaction is the reaction of HCl in an acidic media. As an...
For the reaction 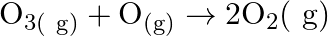 if the rate law expression is, rate
if the rate law expression is, rate ![Rendered by QuickLaTeX.com =\mathrm{K}\left[\mathrm{O}_{3}\right][\mathrm{O}]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-e69a8d7ed909b9b87bef5e92873c3d1c_l3.png) the molecularity and order of the reaction are respectively A) 2 and 2 B) 2 and
the molecularity and order of the reaction are respectively A) 2 and 2 B) 2 and 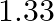 C) 2 and 1 D) 1 and 2
C) 2 and 1 D) 1 and 2
Correct option is A (2 and 2) Rate $=\mathrm{K}\left[\mathrm{O}_{3}\right][\mathrm{O}]$ Order $=1+1=2$ Hence, the overall order of the reaction is 2 . Now, $ \mathrm{O}_{3(9)}+\mathrm{O}_{(9)}...
![Rendered by QuickLaTeX.com \left[\mathrm{Cr}\left(\mathrm{NH}_{3}\right)_{6}\right]\left[\mathrm{Cr}(\mathrm{SCN})_{6}\right] \text { and }\left[\mathrm{Cr}\left(\mathrm{NH}_{3}\right)_{2}(\mathrm{SCN})_{4}\right]\left[\mathrm{Cr}\left(\mathrm{NH}_{3}\right)_{4}(\mathrm{SCN})_{2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-1c51c7d9e0fc5c8f37f7a46ee940d288_l3.png) are the examples of what type of isomerism ? A) Ionisation isomerism B) Linkage isomerism C) Coordination isomerism D) Solvate isomerism
are the examples of what type of isomerism ? A) Ionisation isomerism B) Linkage isomerism C) Coordination isomerism D) Solvate isomerism
Correct option is C (Coordination isomerism) Explanation: The above examples are of coordination isomerism. It is because there are two coordination spheres in a compound.
For which among the following reactions, change in entropy is less than zero ? A) Sublimation of Iodine B) Dissociation of Hydrogen C) Formation of water D) Thermal decomposition of Calcium Carbonate
Correct option is B Explanation: The change in entropy during hydrogen dissociation is less than zero, which indicates it is negative. Hydrogen dissociation is a non-spontaneous process. As a...
The only radioactive element among the lanthanoids is A) Gadolinium B) Holmium C) Promethium D) Neodynium
Correct option is C Explanation: Promethium (Pm) is a lanthanoid element with the atomic number 61 and the symbol Pm. Its isotopes are all radioactive. Lanthanoids such as gadolinium, holmium, and...
The overall reaction taking place at anode during electrolysis of fused sodium chloride using suitable electrode is A) Oxidation of chloride B) Reduction of sodium ions C) Reduction of chlorine D) Oxidation of Sodium atoms
Correct option is A During the electrolysis of fused sodium chloride with a suitable electrode, the chloride ion is oxidised at the anode. Chloride ions are oxidised to chlorine gas by giving up...
What is the most abundant element on earth ? A) Hydrogen B) Nitrogen C) Oxygen D) Silicon
Correct option is C Explanation: Silicate minerals make up more than 90% of the earth's crust. Feldspars and alkali feldspar are the most common silicates. Other common silicate minerals include...
Diethyl amine when treated with nitrous acid yields A) Diethyl ammonium nitrite B) Ethyl alcohol C) N-nitroso diethyl amine D) Triethyl ammonium nitrite
Correct option is C) N-nitroso diethyl amine Explanation: N-nitroso diethylamine is formed by treating diethylamine with nitrous acid. It's a strong carcinogen.
Potassium dichromate is a good oxidizing agent, in acidic medium the oxidation state of chromium changes by A) 2 B) 3 C) 4 D) 5
Correct option is B Explanation: Dichromate oxidizes to $\mathrm{Cr}^{+3}$ from $\mathrm{Cr}_{2} \mathrm{O}_{7}^{-2}$ in acidic medium according to the reaction $ \mathrm{Cr}_{2}...
Which metal among the followings has the highest packing efficiency ? A) Iron B) Tungsten C) Aluminium D) Polonium
Correct option is C Explanation: The packing efficiency is the fraction of volume occupied by constituent particles in a crystal structure (volume of space occupied by the spheres/total volume). The...
Which of the following compounds has highest boiling point ? A) Propan-1-ol B) n-Butane C) Chloroethane D) Propanal
Correct option is A) Propan-1-ol Explanation: Hydrogen bonds have the potential to occur in propan-1-ol. Because of the strongly E.N oxygen atom, intermolecular hydrogen bonding develop in...
How is ore of aluminium concentrated ? A) roasting B) leaching C) froth floatation D) using Wilfley table
B is the correct answer. Explanation: The leaching method concentrates aluminium ore. Impurities in aluminium ore include SiO2, iron oxides, and titanium oxide. The powdered ore is digested with a...
Which of the following compounds is most acidic in nature ? A) 4-Chlorobutanoic acid B) 3-Chlorobutanoic acid C) 2-Chlorobutanoic acid D) Butanoic acid
The correct answer is C(2-chlorobutanoic acid) In nature, 2-chlorobutanoic acid is the most acidic. Because there is an electron withdrawing group (Cl) next to the carboxylic acid group (COOH). As a...
Van’t Hoff factor of centimolal solution of ![Rendered by QuickLaTeX.com \mathrm{K}_{3}\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a99adc5c7be224b196c9e2507b50069e_l3.png) is
is 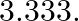 Calculate the percent dissociation of
Calculate the percent dissociation of ![Rendered by QuickLaTeX.com \mathrm{K}_{3}\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a99adc5c7be224b196c9e2507b50069e_l3.png) A)
A) 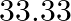 B)
B) 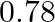 C) 78 D)
C) 78 D) 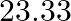
Correct option is C Explanation: $ \mathrm{K}_{3}\left[\mathrm{Fe}(\mathrm{CN})_{6}\right] \rightarrow 3 \mathrm{~K}^{+}+\left[\mathrm{Fe}(\mathrm{CN})_{6}\right]^{3-} $ $ \mathrm{i}=\alpha...
Replacement of diazonium group by fluorine is known as A) Gattermann reaction B) Sandmeyer reaction C) Balz-Schiemann reaction D) Etard reaction
Correct option is C Balz-Schiemann reaction Explanation: The Balz- Schiemann reaction is a chemical reaction in which anilines are transformed to aryl fluorides via diazonium fluoroborates....
If 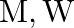 and
and 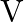 represent molar mass of solute, mass of solute and volume of solution in litres respectively, which among following equations is true? A)
represent molar mass of solute, mass of solute and volume of solution in litres respectively, which among following equations is true? A) 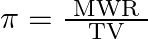 B)
B) 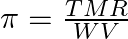 C)
C) 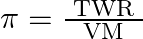 D)
D) 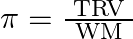
Correct option is C $\pi=\frac{\mathrm{TWR}}{\mathrm{VM}}$ $\pi=$ CRT where $\mathrm{C}$ is the concentration $\mathrm{C}=\frac{\text { Moles of solute }}{\text { volume }}$...
The correct IUPAC name of ![Rendered by QuickLaTeX.com \left[\mathrm{CO}\left(\mathrm{NH}_{3}\right)_{3}\left(\mathrm{NO}_{2}\right)_{3}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a87a614704284e544285de047b62719a_l3.png) A) Triammine trinitrito – N cobalt (III) B) Triammine trinitrito
A) Triammine trinitrito – N cobalt (III) B) Triammine trinitrito  cobalt (II) C) Triammine cobalt (III) nitrite D) Triammine trinitrito – N cobaltate (III)
cobalt (II) C) Triammine cobalt (III) nitrite D) Triammine trinitrito – N cobaltate (III)
Correct option is A) Triammine trinitrito - N cobalt Triammine trinitro-N-cobalt is the correct IUPAC name (III). In this scenario, the groups are ordered alphabetically, with the ammine group...
Electronic configuration of only one P block element is exceptional. One molecule of that element consists of how many atoms of it ? A) One B) Two C) Three D) Four
The best option is A Explanation: An only p-block element's electrical arrangement is uncommon. There is only one atom of that element in each molecule. Because helium has the electrical...
Which of the following is the most stable diazonium salt ? A) 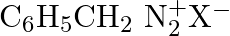 B)
B) 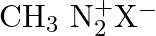 C)
C) 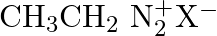 D)
D) 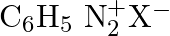
Correct option is B Due to resonance stabilisation between the benzene nucleus and the nitrogen atom, dizonium salts with an aryl group immediately connected to the nitrogen atom are the most...
Which polymer among the following polymers does NOT soften on heating ? A) Bakelite B) Polythene C) Polystyrene D) PVC
The correct option is A Explanation: Thermoplastic is a plastic material that can be melted and moulded repeatedly by just heating it. When heated, thermoplastics soften and become moldable. They...
The relationship between rate constant and half life period of zero order reaction is given by A) ![Rendered by QuickLaTeX.com \mathrm{t}_{\frac{1}{2}}=[\mathrm{A}]_{0} 2 \mathrm{k}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-4790aa54d3511e4a28ff21e7043d219a_l3.png) B)
B) 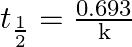 C)
C) ![Rendered by QuickLaTeX.com t_{\frac{1}{2}}=\frac{[\mathrm{A}]_{0}}{2 \mathrm{k}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-3dde05612d23f52ee0597c3f471cfe53_l3.png) D)
D) ![Rendered by QuickLaTeX.com \mathrm{t}_{\frac{1}{2}}=\frac{2[\mathrm{~A}]_{0}}{\mathrm{k}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a9a16937140e0a1a6dbc47fd5be5c9a5_l3.png)
Correct option is C $\mathrm{t}_{1 / 2}=\frac{\left[\mathrm{A}_{0}\right]}{2 \mathrm{k}}$ The relationship between rate constant and half-life period of zero-order reaction is given by: $...
Identify a ‘Chemical twin’ among the followings. A) Zr-Ta B) Nb-Tc C) Hf-Re D) Nb-Ta
D is the correct answer. Nb−Ta Chemical twins are those in which some elements' characteristics are identical. Zr and Hf have comparable atomic sizes, as do Nb and Ta. This is related to the...
Chlorination of ethane is carried out in presence of A) anhydrous AlBr3 B) mercuric chloride C) ultraviolet light D) zinc chloride
Correct option is C) ultraviolet light Chlorination of ethane is carried out in the presence of Ultraviolet light to form 1,2-hexachloro ethane.
Which among the following is a tranquilizer ? A) Aspirin B) Valium C) Penicillin D) Sulphanilamide
Correct option is B) Valium A tranquillizer is a medicine that causes people and animals to become calm. Anxiety disorders, alcohol withdrawal symptoms, and muscular spasms are all treated with...
Which among the following alloys is used in making instruments for electrical measurements ? A) Stainless steel B) Manganin C) Spiegeleisen D) Duralumin
Correct option is B) Manganin Manganin foil and wire are employed in the production of resistors, notably ammeter shunts, due to its low temperature coefficient of resistance and long-term...
What is the amount of work done when two moles of ideal gas is compressed from a volume of 1 m3 to 10 dm3 at 300 K against a pressure of 100 kPa ? A) 99 kJ B) – 99 kJ C) 114.9 kJ D) – 114.9 kJ
Correct option is A $99 \mathrm{~kJ}$ $ \mathrm{V}_{1}=1 \mathrm{~m}^{3} $ Pressure $=100 \mathrm{kPa}=100000 \mathrm{~Pa}$ $\mathrm{V}_{2}=10 \mathrm{dm}^{3}=(1 / 100) \mathrm{m}^{3}$ So, $W=P...
What is the possible number of monohydroxy derivatives of a hydrocarbon consisting of five carbon atoms with one methyl group as a branch ? A) 2 B) 3 C) 4 D) 5
Correct option is B Only three isomers of mono hydroxy derivatives of a hydrocarbon with five carbon atoms and one methyl group as a branch are feasible.
Molarity is defined as A) the number of moles of solute dissolved in one dm3 of the solution B) the number of moles of solute dissolved in 1 kg of solvent C) the number of moles of solute dissolved in 1 dm3 of the solvent D) the number of moles of solute dissolved in 100 ml of the solvent
Correct option is (A) The number of moles of solute dissolved in 1dm3 of the solution is called molarity. Molarity = (moles of solute dissoved) / (volume of solvent in dm3) So, answer is...
Which among the following is a feature of adiabatic expansion ? A) ΔV < 0 B) ΔU < 0 C) ΔU > 0 D) ΔT = 0
Correct option is D) ΔT = 0 T=0 for adiabatic expansion. An ideal gas is enclosed in an insulated container and then allowed to expand in a vacuum for adiabatic free expansion. The work done by or...
Half life period of a first order reaction, A→ product is 6.93 hour. What is the value of rate constant ? A) 1.596 h–1 B) 0.1 h–1 C) 4.802 h–1 D) 10 h–1
Correct option is B ($0.1 \mathrm{~h}^{-1}$) We know, $ \mathrm{t}_{\frac{1}{2}}=\frac{0.693}{\mathrm{k}} $ where $t_{\frac{1}{2}}$ is the half life and $\mathrm{k}$ is rate constant. $...
During conversion of glucose into glucose cyanohydrin, what functional group/atom of glucose is replaced ? A) hydrogen B) aldehydic group C) primary alcoholic group D) secondary alcoholic group
Correct option is B Aldehydic group Glucose reacts with hydrogen cyanide to form glucose cyanohydrin. In this reaction functional group is replaced that is −CHO group.
Reaction of which among the following ethers with HI in cold leads to formation of methyl alcohol ? A) ethyl methyl ether B) methyl propyl ether C) isopropyl methyl ether D) tert-butyl methyl ether
Correct option is D) tert-butyl methyl ether
Which among the following detergents is non-ionic in character ? A) Sodiumlauryl sulphate B) Pentaerythrityl stearate C) Cetyltrimethyl ammonium chloride D) Sodium n-dodecyl benzene sulphonate
B is the correct answer. Pentaerythrityl stearate is a kind of stearic acid. The uncharged, hydrophilic headgroups of non-ionic detergents distinguish them from ionic detergents. Polyoxyethylene or...
What oxoacid of sulphur contains S-S bond in its structure ? A) Disulphurous acid B) Disulphuric acid C) Perdisulphuric acid D) Hydrosulphurous acid
Correct option is (A)Disulphurous acid Disulphurous acid(H2S2O5) contain S-S bond in its structure.
In potentiometer experiment, the potential gradient can be increased by
A. increasing the cross-sectional area of the potentiometer wire.
B. using a wire of material of low specific resistance.
C. decreasing the cross-sectional area of the potentiometer wire.
D. decreasing the current through it.
Correct answer is C.
When a resistance ‘ 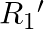 is connected across the terminal of a cell of e.m.f. ‘
is connected across the terminal of a cell of e.m.f. ‘ 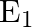 ‘ the current is ‘
‘ the current is ‘ 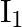 ‘. When the resistance is changed to
‘. When the resistance is changed to 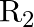 ‘ the current is ‘
‘ the current is ‘ 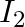 ‘. The internal resistance of the cell is
‘. The internal resistance of the cell is
A. 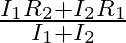
B. 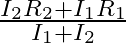
C. 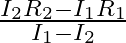
D. 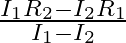
Correct answer is C. $\begin{array}{l} I_{1}=\frac{E}{r+R_{1}} \\ I_{2}=\frac{E}{r+R_{2}} \\ \frac{I_{1}}{I_{2}}=\frac{r+R_{2}}{r+R_{1}} \\ I_{1} r+I_{1} R_{1}=I_{2} r+I_{2} R_{2} \\...
The dimensions of angular momentum are
A. ![Rendered by QuickLaTeX.com \left[L^{2} M^{1} T^{-1}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-e3ae8151d351e21d072dc99a517680a5_l3.png)
B. ![Rendered by QuickLaTeX.com \left[L^{2} M^{2} T^{-2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-570bf7fd6b73030ec948ef99c8b75448_l3.png)
C. ![Rendered by QuickLaTeX.com \left[L^{2} M^{1} T^{1}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-8a2e9f8d186a5b9fceddc341575a2dfb_l3.png)
D. ![Rendered by QuickLaTeX.com \left[L^{2} M^{1} T^{-2}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-6d349a07b4098874e03d0238509c1b23_l3.png)
Correct answer is A.
Work done by the force on the body is the measure of the change in its
A. momentum.
B. mass.
C. potential energy.
D. kinetic energy.
Correct answer is D.
A wire suspended vertically is stretched by 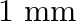 , when mass of
, when mass of 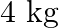 is attached to the lower end. Energy gained by the wire is (Take
is attached to the lower end. Energy gained by the wire is (Take 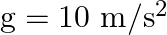 )
)
A. 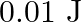
B. 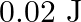
C. 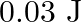
D. 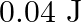
Correct answer is B
Three vessels A, B, C of different shapes have same base area and are filled with water up to same height ‘ 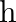 ‘. The respective forces exerted by water on the bases are ‘
‘. The respective forces exerted by water on the bases are ‘ 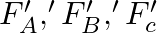 and respective weights are
and respective weights are 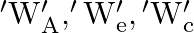 . Then
. Then
A. 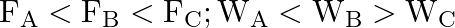
B. 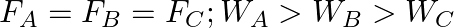
C. 
D. 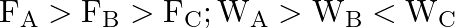
Correct answer is B.
In case of Light Emitting Diode (LED) light is emitted due to A. recombination of holes and electrons.
B. emission of holes and electrons.
C. diffusion of holes.
D. drifting of electrons
Correct answer is A.
The sequence of harmonics of a pipe open at one end and closed at the other end is 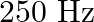 and
and 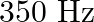 . The resonating length of the air column in its fundamental mode will be (velocity of sound in air
. The resonating length of the air column in its fundamental mode will be (velocity of sound in air 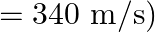
A. 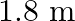
B. 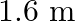
C. 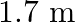
D. 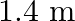
Correct answer is C. $\begin{array}{l} 2 n=n_{2}-n_{1} \\ =350-250=100 \\ n=\frac{100}{2}=50 H z \\ n=\frac{v}{4 l_{0}}=\frac{340}{4 \times l_{0}} \\ \therefore l=\frac{340}{4 \times...
The ratio of magnetization and magnetic intensity is called
A. permeability
B. magnetic induction
C. magnetic susceptibility
D. gyromagnetic ratio
Correct answer is C.
The ratio of magnetic induction along the axis to magnetic induction along the equator of a bar magnet for a point at the same distance 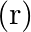 from the centre of magnet is
from the centre of magnet is
A. 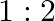
B. 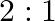
C. 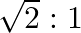
D. 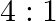
Correct answer is B. Magnetic induction at axial point $=\overrightarrow{\mathrm{B}}$ axial $=\frac{\mu \circ}{4 \pi} \frac{2 \overrightarrow{\mathrm{m}}}{\mathrm{r}^{3}}$ Magnetic induction at...
A metal surface ejects electrons when irradiated by blue light, but not when irradiated by green light. The electrons will also be ejected by the surface, when irradiated by
A. red light
B. voilet light
C. orange light
D. yellow light
Correct answer is B.
Two long, straight wires are set parallel to each other. Both the wires carry a current ‘I’ in opposite directions and separation between them is ‘ 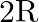 ‘. The magnetic induction at the midway between them is
‘. The magnetic induction at the midway between them is 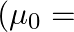 permeability of free space)
permeability of free space)
A. Zero
B. 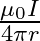
C. 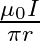
D. 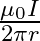
Correct answer is A. $\begin{array}{l} \mathrm{x}=\frac{\mathrm{r}}{2} \\ \mathrm{~B}_{1}=-\frac{\mu_{0} \mathrm{i}}{2 \pi \mathrm{x}} \\ \mathrm{B}_{2}=\frac{\mu_{0} \mathrm{i}}{2...
The component of the vector 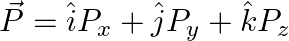 along the direction of
along the direction of 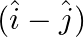 is
is
A. 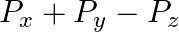
B. 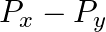
C. 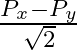
D. 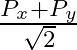
Correct answer is C.
A charged capacitor has charge ‘ 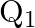 ‘ at potential
‘ at potential 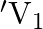 ‘. The charge on capacitor is increased to ‘
‘. The charge on capacitor is increased to ‘ 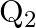 ‘ so that its potential increases to
‘ so that its potential increases to 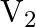 , then the difference between final and initial energy stored in capacitor is
, then the difference between final and initial energy stored in capacitor is
A. 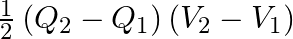
B. 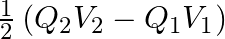
C. 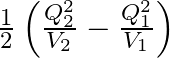
D. 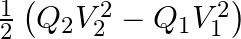
Correct answer is B.
In the formula for high resolving power microscope, the numerical aperture of the object of the microscope is given as 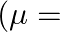 refractive index of oil and angle made by the object at the aperture =
refractive index of oil and angle made by the object at the aperture = 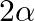 )
)
A. 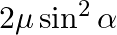
B. 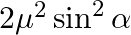
C. 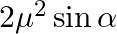
D. 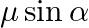
Correct answer is D.
Bending of beam can be reduced by using
A. a material with large value of Young’s modulus.
B. small value of the depth of the bar
C. large value of the length of the beam.
D. small value of the breadth of the bar
Correct answer is A.
‘M’  weight is suspended from a weightless spring and it has period ‘
weight is suspended from a weightless spring and it has period ‘ 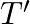 . If now ‘
. If now ‘ 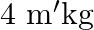 weight is suspended from the same spring, the new period will be
weight is suspended from the same spring, the new period will be
A. 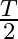
B. 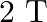
C. 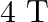
D. 
Correct answer is B. $\begin{array}{l} \mathrm{T}=2 \pi \sqrt{\frac{\mathrm{m}}{\mathrm{k}}} \\ \mathrm{T}_{1}=\mathrm{T} \\ \mathrm{m}_{1}=1 \mathrm{~kg} \\ \mathrm{~m}_{2}=4 \mathrm{~kg} \\...
A conical pendulum has bob of mass of 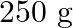 and a length of
and a length of 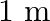 . If the horizontal circle traced by the bob has a diameter of
. If the horizontal circle traced by the bob has a diameter of  , then the tension in the string would be nearly
, then the tension in the string would be nearly 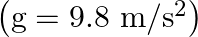
A. 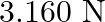
B. 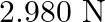
C. 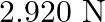
D. 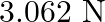
Correct answer is D.
In Biprism experiment the distance between the two virtual images of the slits in the magnified and diminished position are 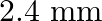 and
and 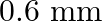 respectively. The distance between two coherent sources is
respectively. The distance between two coherent sources is
A. 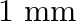
B. 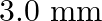
C. 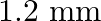
D. 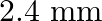
Correct answer is C. When, 1) magnified, distance between images, $\mathrm{d}_{1}=2.4$ 2) dimnished, distance between images, $\mathrm{d}_{2}=0.6$ $\therefore$ Distance between virtual sources...
Half-lives of two radioactive elements A and B are 20 minute and 40 minute respectively. Initially the samples have equal nuclei. After 80 minute, the ratio of decayed number of A and B nuclei will be
A. 
B. 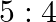
C. 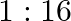
D. 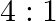
Correct answer is B. $\begin{array}{l} \mathrm{n}=\mathrm{n}_{0} \mathrm{e}^{-\alpha t} \\ \alpha_{\mathrm{A}}=\ln 2 / 20 \\ \alpha_{\mathrm{B}}=\ln 2 / 40 \end{array}$ After 80 min, remaining...
The S.I. unit of impulse is the same as that of
A. linear velocity.
B. angular velocity.
C. linear momentum.
D. angular momentum.
Correct answer is C.
Dimensions of magnetic intensity are
A. ![Rendered by QuickLaTeX.com \left[L^{-1} M^{0} T^{0} I^{1}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-61bc00b014420d451f210b1b6d369577_l3.png)
B. ![Rendered by QuickLaTeX.com \left[L^{-1} M^{0} T^{0} I^{-1}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c460b6b0a8d363b4b9c32706e423f306_l3.png)
C. ![Rendered by QuickLaTeX.com \left[L^{-1} M^{0} T^{0} I^{-1}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c460b6b0a8d363b4b9c32706e423f306_l3.png)
D. ![Rendered by QuickLaTeX.com \left[L^{-1} M^{1} T^{1} I^{-1}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-9ab8d27e17f1706b0fe994dd5197b794_l3.png)
Correct answer is A.
Two vibrating tuning forks produce waves which are represented by 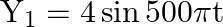 and
and 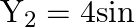
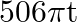 . The number of beats produced per minute by these two waves is
. The number of beats produced per minute by these two waves is
A. 60
B. 180
C. 360
D. 3
Correct answer is B. $\begin{array}{l} \mathrm{Y}_{1}=4 \sin (500 \pi \mathrm{t}) \\ \mathrm{Y}_{2}=2 \sin (506 \pi \mathrm{t}) \\ \mathrm{f}_{1}=\frac{\omega_{1}}{2 \pi}=\frac{500 \pi}{2 \pi}=250...
The innermost orbit of the hydrogen atom has a diameter of 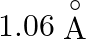 , the diameter of the tenth orbit is
, the diameter of the tenth orbit is
A. 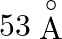
B. 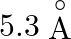
C. 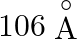
D. 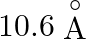
Correct answer is C. Radius of an atom is directly proportional to $n^{2}$ and radius of $n$th orbit which is equal to radius of first orbit multiplied by $n^{2}$ So that will give you. Diameter of...
A sonometer wire of length ‘ 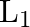 ‘ is in unison with a tuning fork of frequency’n’. When the vibrating length of the wire is reduced to ‘
‘ is in unison with a tuning fork of frequency’n’. When the vibrating length of the wire is reduced to ‘ 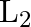 ‘, it produces ‘
‘, it produces ‘ 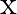 ‘ beats per second with the fork. The frequency of the fork is
‘ beats per second with the fork. The frequency of the fork is
A. 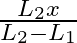
B. 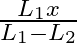
C. 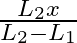
D. 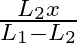
Correct answer is D. For the length $L_{1}, n_{1}=\frac{1}{2 L_{1}} \sqrt{\frac{T}{m}}=n \ldots(1)$ where $m=$ mass per unit length For the length $L_{2}, n_{2}=\frac{1}{2 L_{2}} \sqrt{\frac{T}{m}}...
Which of the following statements is NOT true for a particle moving in circle with a constant angular speed?
A. The radial acceleration vector is tangent to the circle.
B. The linear velocity vector is tangent to the circle.
C. The radial acceleration vector points towards the centre of the circle.
D. The linear velocity and radial acceleration vectors at a point are perpendicular to each other.
Correct answer is A.
A particle performs simple harmonic motion from mean position, with period 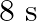 . The distance travelled by it between 1 st and 2 nd second of its motion is (A = amplitude of S. H. M.)
. The distance travelled by it between 1 st and 2 nd second of its motion is (A = amplitude of S. H. M.)
A. 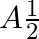
B. 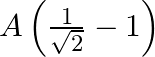
C. 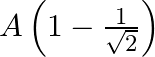
D. 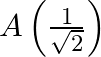
Correct answer is C. In $2 \mathrm{~s}$ (which is equal to $\frac{T}{4}$ ), one amplitude will be convered. In $1 \mathrm{st}$ second $x=a \sin \left(\frac{\pi}{4}\right)=\frac{a}{\sqrt{2}}$...
A capillary tube is dipped in water, the water rises in it to a height ‘h’. If ‘  ‘ is the radius of bore of tube then
‘ is the radius of bore of tube then
A. 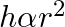
B. 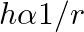
C. 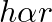
D. 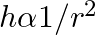
Correct answer is B. We have, $\mathrm{h}=\frac{2 \sigma \cos \theta}{\mathrm{r} \rho \mathrm{g}}$ $\Rightarrow \mathrm{h} \cdot \mathrm{r}=$ constant
When an npn transistor is used as an amplifier
A. electrons move from base to collector.
B. holes move from emitter to base.
C. electrons move from collector to base,
D. holes move from base to collector.
Correct answer is A.
A charge ‘ 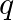 ‘ is enclosed by a Gaussian spherical surface of radius ‘R’. If, now, radius of the spherical surface is doubled the outward electric flux will
‘ is enclosed by a Gaussian spherical surface of radius ‘R’. If, now, radius of the spherical surface is doubled the outward electric flux will
A. doubled
B. remain same
C. increase four times
D. reduce to half
Correct answer is B. Method 1: Using Number of Field Lines - We know that, Electric flux is proportional to number of field lines. - Form figure, we see that the number of field lines crossing both...
The rate of cooling of a body is 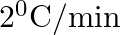 when the body is at
when the body is at 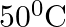 above the temperature of surroundings. When the body is at
above the temperature of surroundings. When the body is at 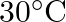 above the temperature of surroundings, its rate of cooling will be
above the temperature of surroundings, its rate of cooling will be
A. 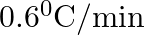
B. 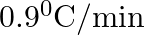
C. 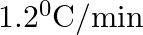
D. 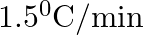
Correct answer is C. Rate of cooling $\frac{\mathrm{dT}}{\mathrm{dt}}=-\frac{\mathrm{k}}{\mathrm{ms}}\left(\mathrm{T}-\mathrm{T}_{\mathrm{s}}\right)$ Or $2=\frac{-\mathrm{k}}{\mathrm{ms}}(50)$ Or...
The mass and radius of earth is 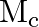 , and
, and 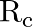 , respectively and that of moon is
, respectively and that of moon is 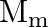 and
and 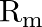 respectively. The distance between the centre of earth and that of moon is ‘D’ . The minimum speed required for a body (mass ‘m’) to project from a point midway between their centres to escape to infinity is
respectively. The distance between the centre of earth and that of moon is ‘D’ . The minimum speed required for a body (mass ‘m’) to project from a point midway between their centres to escape to infinity is
A. 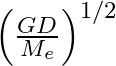
B. 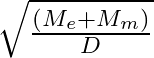
C. 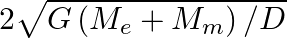
D. 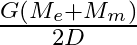
Correct answer is C. From conservation of energy, since the velocity will be o at infinity, $\frac{1}{2} \mathrm{mv}^{2}-\frac{\mathrm{GmM}_{e}}{\mathrm{~d} / 2}-\frac{\mathrm{GmM}_{d}}{\mathrm{~d}...
The magnitude of the induced e.m.f. produced in the coil when a magnet is inserted into it does ‘NOT’ depend upon the
A. speed of approach of the magnet.
B. magnetic moment of the magnet.
C. number of turns of the coil.
D. resistance of the coil.
Correct answer is D.
From the position of smaller angular displacement  , the bob of simple pendulum of length ‘L’ is released then its linear displacement at time ‘t’ is
, the bob of simple pendulum of length ‘L’ is released then its linear displacement at time ‘t’ is
A. 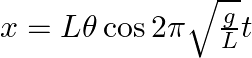
B. 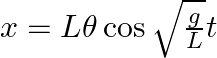
C. 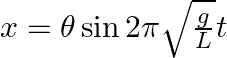
D. 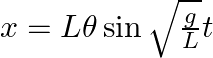
Correct answer is B $\begin{array}{l} x=1 \cos w t \\ =1 \sqrt{\frac{g}{L}} t \cos \theta \end{array}$
In a cyclotron the time required by positive ion to traverse semicircle does NOT depend upon
A. mass of the particle.
B. velocity of the particle.
C. strength of magnetic field.
D. charge of the particle.
Correct answer is C.
A point source is placed in air. The spherical wavefront has radius ‘ 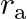 ‘ after time ‘t’. If the same point source is placed in the medium of refractive index ‘
‘ after time ‘t’. If the same point source is placed in the medium of refractive index ‘ 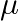 ‘, the radius of spherical wavefront in the medium in same time
‘, the radius of spherical wavefront in the medium in same time 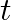 is
is
A. 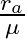
B. 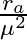
C. 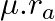
D. 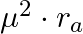
Correct answer is A. Here Velocity of wave front $=\frac{R_{9}}{t}$ $\begin{array}{l} u=-1 \\ \frac{e}{R a / t}=1 \\ e=R a / t \end{array}$ in the meduim $u$ $\text { velochy }=R \frac{a}{t}$ so,...
If angular momentum of a rotating body about a fixed axis is increased by 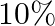 , its rotational kinetic energy will be increased by
, its rotational kinetic energy will be increased by
A. 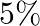
B. 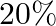
C. 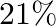
D. 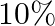
Correct answer is B. Given, $\frac{\Delta \mathrm{L}}{\mathrm{L}}=10 \%$ The expression for the rotational kinetic energy in terms of angular momentum, $\mathrm{K}=\frac{\mathrm{L}^{2}}{2...
When an a.c. source is connected across a pure inductor of self inductance (L) then e.m.f. induced in it
A. Leads the current by phase 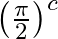
B. Lags behind the current by phase 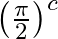
C. Lags behind the current by phase 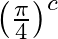
D. Leads the current by phase 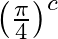
Correct answer is A.
Acceleration due to gravity at height ‘h’ above the surface of the earth is 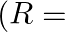 Radius of the earth)
Radius of the earth)
A. 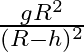
B. 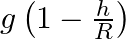
C. 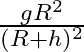
D. 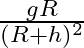
Correct option is C. let we consider, radius of earth $\mathrm{R}$ and mass is $\mathrm{M}$ and we know that acceleration due to gravity at surface G is universal gravitational constant. consider, a...
The excess of pressure inside a soap bubble is twice the excess of pressure inside another soap bubble. The volume of first bubble is ‘n’ times the volume of second bubble. The value of ‘ 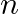 ‘ must be
‘ must be
A. 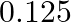
B. 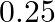
C. 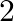
D. 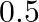
Correct answer is A. Given, $\frac{4 \mathrm{~T}}{\mathrm{r}_{1}}=2 \times \frac{4 \mathrm{~T}}{\mathrm{r}_{2}}$ or $\mathrm{r}_{2}=2 \mathrm{r}_{1}$ $\frac{4}{3} \pi \mathrm{r}_{1}^{3}=\mathrm{n}...
If the telecast is to cover a range of 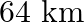 , the height of
, the height of 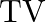 tower should be (Radius of earth,
tower should be (Radius of earth, 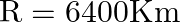 )
)
A. 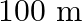
B. 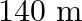
C. 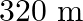
D. 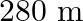
Correct answer is C. Here, Range $=d=64 \mathrm{~km}$, height $=\mathrm{h}$ and radius of earth $=$ $r=6400 \mathrm{~km}$. $\begin{array}{l} \text { Now }, d=\sqrt{2 h R} \\ \Rightarrow...
If two non-zero vectors 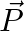 and
and 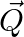 are perpendicular to each other then the magnitude of their vector product is
are perpendicular to each other then the magnitude of their vector product is  ,
,
A. 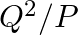
B. 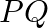
C. 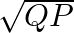
D. 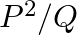
Correct answer is B. Vector product is calculated as, $\vec{P}\times\vec{Q}=PQsin90^{\circ}$ $\vec{P}\times\vec{Q}=PQ$
A wall is hit elastically and normally by ‘ 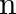 ‘ balls per second. If ‘
‘ balls per second. If ‘ 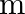 ‘ is the mass and ‘u’ is velocity for each molecule, the force exerted by the balls on the wall is
‘ is the mass and ‘u’ is velocity for each molecule, the force exerted by the balls on the wall is
A. 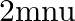
B. 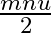
C. 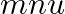
D. 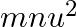
Correct answer is A. Since the ball rebounds with same speed but in opposite direction. So, change in linear momentum of one ball $\Delta...
A solid sphere is rolling down a frictionless surface with translational velocity 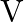 . It climbs the inclined plane from ‘A’ to ‘
. It climbs the inclined plane from ‘A’ to ‘ 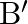 and then moves away from ‘
and then moves away from ‘ 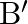 on the smooth horizontal surface. The value of
on the smooth horizontal surface. The value of 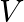 should be
should be
A. 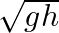
B. ![Rendered by QuickLaTeX.com \geq\left[\frac{10 g h}{7}\right]^{1 / 2}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-90f667f0759902947f521ac966f472fc_l3.png)
C. 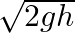
D. 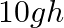
Correct answer is B. Applying law of conservation of energy for rotating body, $\begin{array}{l} \frac{1}{2} \mathrm{mv}^{2}+\frac{1}{2} \mathrm{I} \omega^{2}=\mathrm{mgh} \\ \frac{1}{2}...
If the errors involved in the measurements of a side and mass of a cube are 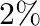 and
and  respectively, what is the maximum permissible error in the density of the material ?
respectively, what is the maximum permissible error in the density of the material ?
A. 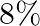
B. 
C. 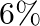
D. 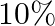
Correct answer is D. Density $\rho=\frac{M}{V}=\frac{M}{L^{3}}$ The percentage error in density is given by, $\begin{array}{l} \frac{\Delta \rho}{\rho} \times 100=\frac{\Delta M}{M} \times 100-3...
Glass has refractive index 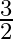 and water has refractive index
and water has refractive index 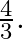 If the speed of light in glass is
If the speed of light in glass is 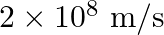 , then the speed of light in water will be
, then the speed of light in water will be
A. 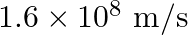
B. 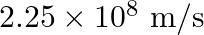
C. 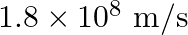
D. 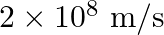
Correct answer is B. $\begin{array}{l} \quad \mathrm{v}_{\text {water }}=\frac{\mathrm{c}}{\mathrm{n}_{\text {water }}} \\ \therefore \quad \mathrm{v}_{\mathrm{water}}=\frac{3 \times 10^{8}}{(4 /...
A transverse wave is travelling on a string with velocity ‘ 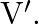 The extension in the string is ‘
The extension in the string is ‘ 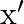 . If the string is extended by
. If the string is extended by 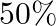 , then the speed of wave along the string will be nearly (Hooke’s law is obeyed)
, then the speed of wave along the string will be nearly (Hooke’s law is obeyed)
A. 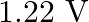
B. 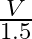
C. 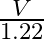 D.
D. 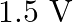
Correct option is A. $\begin{array}{l} v=\sqrt{\frac{T}{m}} \\ \therefore \frac{\mathrm{V}_{2}}{\mathrm{~V}_{1}}=\sqrt{\frac{\mathrm{T}_{2}}{\mathrm{~T}_{1}}} \\ =\sqrt{1.5} \approx 1.22...
A galvanometer of resistance 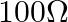 is connected to a battery of
is connected to a battery of 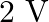 with a resistance of
with a resistance of 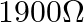 in series. The deflection obtained is 30 divisions. To reduce this deflection by 10 divisions the additional resistance required to be connected in series is
in series. The deflection obtained is 30 divisions. To reduce this deflection by 10 divisions the additional resistance required to be connected in series is
A. 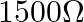
B. 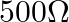
C. 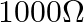
D. 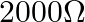
Correct option is C. Total resistance in the circuit is $R=1900+100=2000 \Omega$ $\therefore$ Current $I_{1}=\frac{2}{2000}=10^{-3} A$ The deflection is decreased from 30 to 20 divisions $\therefore...
Which one of the following is NOT correct?
A. A real, inverted and same size image can be obtained by using concave mirror
B. A real, inverted and same size image can be obtained by using convex mirror
C. the size of the image formed by convex mirror is always smaller than size of the object
D. A virtual, erect, magnified image can be obtained by using concave mirror.
The wrong option is (B).
