Correct option is (A) $\frac{3 \mathrm{v}}{2}$ $\begin{array}{l} \mathrm{hv}=\mathrm{W}+\frac{\mathrm{v}_{0}}{2} \mathrm{e} \\ \frac{\mathrm{h} v}{2}=\mathrm{W}+\mathrm{v}_{0} \mathrm{e}...
When a certain photosensitive surface is illuminated with monochromatic light of frequency v, the stopping potential for the photocurrent is
Identify the invalid equation.
(A) $\vartriangle $H = $\sum $H products - $\sum $Hreactants (B) $\vartriangle $H = $\vartriangle $U + P$\vartriangle $V (C) $\vartriangle $ HOreaction = $\sum $H0(product bonds) - $\sum...
Calculate the work done during compression of 2 mol of ideal gas from a volume of 1m3 to10dm3 at 300 K against a pressure of 100 Kpa.
(A)‑ 99 kJ (B) + 99 kJ (C)+ 22.98 kJ (D)‑ 22.98 kJ Correct Answer: (B) + 99 kJ Explanation: Formula Used - W=−2.303nRTlogV2/V1
Which element among the following does form Pπ−Pπ multiple bonds?
(A)Arsenic (B) Nitrogen (C)Phosphorus (D)Antimony Correct Answer: (B) Nitrogen Explanation: The element Nitrogen form Pπ−Pπ multiple bonds.
Tert‑butyl methyl ether on treatment with hydrogen iodide in cold gives
(A) tert‑butyl iodide and methyl iodide (B) tert‑butyl alcohol and methyl alcohol (C) tert‑butyl alcohol and methyl iodide (D) tert‑butyl iodide and methyl alcohol Correct Answer: (D)...
Name the process that is employed to refine aluminum.
(A) Hallʹs process (B) Mond process (C) Hoopeʹs process (D) Serperckʹs process Correct Answer: (C) Hoopeʹs process Explanation: Hoopeʹs process is the process that is employed to refine...
The colour and magnetic nature of manganate ion (MnO42-) is
(A) green, paramagnetic (B) purple, diamagnetic (C) green, diamagnetic (D) purple, paramagnetic Correct Answer: (A) green, paramagnetic Explanation:...
The osmotic pressure of solution containing 34.2 g of cane suger (molar mass =342 mol-1 ) in 1L of solution at is (Given, R = 0.082 L atm K-1 mol-1 )
(A)2.40 atm (B) 3.6 atm (C)24 atm (D)0.0024 atm Correct Answer: (A)2.40 atm Explanation: Calculation – $\begin{array}{l} \pi = CRT = \frac{W}{{M \times V}}RT\\ \pi =...
In assigning R‑S configuration which among the following group has highest priority?
(A)-SO3H (B) -COOH (C) -CHO (D)-C6H5 Correct Answer: (A) -SO3H Explanation: The decreasing order of priority is -SO3H > −COOH > −CHO > −C6H5
Which among the following equations represents the reduction taking place in lead accumulator at positive electrode, while it is being used as source of electrical energy?
(A) Pb Pb2+ (B) Pb4+ Pb (C) Pb2+ Pb (D) Pb4+ Pb2+ Correct Answer: (D) Pb4+ Pb2+ Explanation: Reaction Involved – PbO2(s) + 4H+(aq) + SO42-(aq) + 2e- → PbSO4(s) +...
For which among the following equimolar aqueous solution Vanʹt Hoff factor has the lowest value?
(A) Aluminum Chloride (B) Potassium Sulphate (C) Ammonium Chloride (D) Urea Correct Answer: (D) Urea Explanation: Urea is non-electrolyte and does not dissociate in aqueous solution and so...
The amino acid which is basic in nature is
(A) Histidine (B) Tyrosine (C) Proline (D) Valine Correct Answer: (A)Histidine Explanation: Histidine is the amino acid that is basic in nature.
Which element among the following does not form diatomic molecules?
(A) Argon (B) Oxygen (C) Nitrogen (D) Bromine Correct Answer: (A)Argon Explanation: The element Argon is a noble gas so it does not form diatomic molecules.
A molecule of Stachyose contains how many carbon atoms?
(A)6 (B) 12 (C) 18 (D) 24 Correct Answer: (D)24 Explanation:
What of the SI unit of conductivity?
(A) Sm (B) Sm-1 (C) Sm2 (D) Sm-2 Correct Answer: (B) Sm-1 Explanation: The SI unit of conductivity is Sm-1
Which of the following is Baeyerʹs reagent?
(A) alkaline KMnO4 (B) acidic K2Cr2O7 (C) alkaline Na2Cr2O7 (D) MnO2 Correct Answer: (A) alkaline KMnO4 Explanation: The alkaline KMnO4 is the Baeyerʹs reagent.
What is the chief constituent of Pyrex glass?
(A) B2O3 (B) SiO2 (C) Al2O3 (D) Na2O Correct Answer: (B) SiO2 Explanation: SiO2 is the chief constituent of Pyrex glass.
Which of the following compounds has lowest boiling point?
(A) n‑butyl alcohol (B) isobutyl alcohol (C) tert‑butyl alcohol (D) sec‑butyl alcohol Correct Answer: (C) Tert‑butyl alcohol Explanation: Tert‑butyl alcohol is the compound that has the...
The rate constant for a first order reaction is 7.0 X 10-4s-1. If initial concentration of reactant is 0.080M, what is the half-life of reaction?
(A) 990S (B) 79.2S (C) 12375S (D)10.10x10-4s Correct Answer: (A) 990S Explanation: Calculation – Half-life period, t1/2 = 0.693/k The half-life period t1/2 = 0.693/7.0 X 10-4s-1 t1/2...
The polymer used in making handles of cookers and frying pans is
(A) Bakelite (B) nylon‑2‑nylon‑6 (C) orlon (D) polyvinyl chloride Correct Answer: (A) Bakelite Explanation: Bakelite is the...
What is the actual volume occupied by water molecules present in 20 cm3 of water?
(A) 20 cm3 (B) 10 cm3 (C) 40 cm3 (D) 24.89 dm3 Correct Answer: (D) 24.89 dm3 Explanation: The actual volume occupied by water molecules present in 20 cm3 of water is...
Which of following coordinate complexes is an exception to EAN rule? (Given At. No. Pt = 78, Fe = 26, Zn = 30, Cu = 29)
(A)[Pt(NH3)6]4+ (B)[Fe(CN)6]4- (C)[Zn(NH3)4]2+ (D)[Cu(NH3)4]2+ Correct Answer: D)[Cu(NH3)4]2+ Explanation: Calculation – EAN = Z – X + Y EAN = 29 − 2 + 8 = 35 The atomic number of nearest...
Which of the following statements is incorrect in case of Hofmann bromamide degradation?
(A) Reaction is useful for decreasing length of carbon chain by one carbon atom (B) It gives tertiary amine (C) It gives primary amine (D) Aqueous or alcoholic KOH is used with bromine ...
Which of the following statements is INCORRECT for pair of elements Zr ‑ Hf?
(A) Both possess same number of valence electrons. (B) Both have identical atomic sizes (C) Both have almost identical ionic radii (D) Both of these belong to same period of periodic table ...
Aldehydes or ketones when treated with C6H5-NH- NH2 the product formed is
(A) semi carbazone (B) phenylhydrazone (C) hydrazone (D) oxime Correct Answer: (B) phenylhydrazone Explanation: Aldehydes or ketones when treated with C6H5-NH- NH2...
Solubility of which among the following solids in water changes slightly with temperature?
(A) KNO3 (B) NaNO3 (C) KBr (D) NaBr Correct Answer: (D) NaBr Explanation: The solubility of the solid, NaBr in water changes slightly with temperature.
What is the quantity of hydrogen gas liberated when 46 g sodium reacts with excess ethanol? (Given At. mass of Na = 23)
(A) 2.4 x 10-3kg (B) 2.0 x 10-3kg (C)4.0 x 10-3 kg (D)2.4 x 10-2 kg Correct Answer: (B) 2.0 x 10-3kg Explanation: 2.0 x 10-3kg is...
Identify the weakest oxidizing agent among the following.
(A) Li+ (B)Na+ (C)H2Se (D)H2Te Correct Answer: A) Li+ Explanation: Li+ is the weakest oxidizing agent.
The monomers used in preparation of dextron are
(A) lactic acid and glycolic acid (B) 3 ‑ Hydroxy butanoic acid and 3 ‑ Hydroxy pentanoic acid (C) styrene and 1, 3 ‑ Butadiene (D) hexamethylenediamine and adipic acid Correct Answer:...
Which among the following compounds does not act as a reducing agent?
(A) H2O (B) H2S (C) H2Se (D)H2Te Correct Answer: (A) H2O Explanation: The water H2O is the compound that do not act as a reducing agent.
Which of the following processes is not used to preserve the food?
(A) Irradiation (B) Addition of salts (C) Addition of heat (D) Hydration Correct Answer: (D) Hydration Explanation: Hydration is not used to preserve food.
In case of substituted aniline the group which decreases the basic strength is
(A)-OCH3 (B) -CH3 (C) –NH2 (D) –C6H5 Correct Answer: (D) –C6H5 Explanation:...
Which among the following equations represents Arrhenius equation?
(A) k =AeEa/RT (B)K= AeRT/Ea (C) k =A/eEa/RT (D)K=A/eRT/Ea Correct Answer: (C) k =A/eEa/RT Explanation: The equation that represents Arrhenius equation is k =A/eEa/RT
Which of the following compounds will give positive idoform test?
(A) Isopropyl alcohol (B) Propionaldehyde (C) Ethyl phenyl ketone (D) Benzyl alcohol Correct Answer: (B) Propionaldehyde Explanation: Propionaldehyde is the compound that give possible...
The first law of thermodynamics for isothermal process is
(A) q=-W (B) U=q (C) U=q (D) U=-qe Correct Answer: (A) q=-W Explanation: The first law of thermodynamics for isothermal process is q=-W
The conversion of ethyl bromide to ethyl iodide using sodium iodide and dry acetone, this reaction is known as
(A) Swarts reaction (B) Finkelstein reaction (C) Sandmeyer reaction (D) Stephen reaction Correct Answer: (B) Finkelstein reaction Explanation:...
What is the hybridization of carbon atoms in fullerene?
(A) sp3 (B) sp (C) sp3 (D) dsp3 Correct Answer: (C) sp3 Explanation: sp3 is the hybridization of carbon atoms in fullerene.
Which of the following is used as antiseptic?
(A) Chloramphenicol (B) Bithional (C) Cimetidine (D) Chlordiazepoxide Correct Answer: (B) Bithional Explanation: Bithional is used as an antiseptic.
In preparation of sulphuric acid from sulphur dioxide in lead chamber process. What substance is used as a catalyst?
(A) Mangenese dioxide (B) Vandadium pentoxide (C) Nitric oxide (D) Reney Nickel Correct Answer: (C) Nitric oxide Explanation: Nitric oxide is the catalyst that is used in the preparation...
The correct charge on and co‑ordination number of ‘Fe’ in K3[Fe(Cn)6] is
(A)+2, 4 (B) +3, 6 (C)+2, 6 (D)+3, 3 Correct Answer: (B) +3, 6 Explanation: The correct charge on and co‑ordination number of ‘Fe’ in K3[Fe(Cn)6]...
Which among the following reactions is an example of pseudo first order reaction?
(A) Inversion of cane sugar. (B) Decomposition of H2O2 (C) Conversion of cyclopropane to propane (D) Decomposition of N2O5 Correct Answer: (A) Inversion of cane sugar Explanation:...
The amine which reacts with p ‑ toluencsulphonyl chloride to give a clear solution which on acidification give insoluble compound is
(A)C2H5NH2 (B)(C2H5)2NH (C)(C2H5)3N (D)CH3NHC2H5 Correct Answer: (A)C2H5NH2 Explanation: The amine which reacts with p ‑ toluencsulphonyl chloride to give a clear solution which on...
The work done during combustion of 9 x 10-2 kg of ethane, C2H6(g) at 300 K is (Given R=8.314J deg-1 mol-1 atomic mass C=12, H= 1)
(A) 6.236 KJ (B) -6.236KJ (C) 18.71KJ (D) -18.71KJ Correct Answer: (A) 6.236 KJ Explanation: The work done during combustion of 9 x 10-2 kg of ethane, C2H6(g) at 300 K is 6.236...
What type of sugar molecule is present in DNA?
(A) D‑3‑deoxyribose (B) D‑ribose (C) D‑2‑deoxyribose (D) D‑Glucopyranose Correct Answer: (B) D‑ribose Explanation: D‑ribose is a sugar molecule that is present in the...
The molality of solution containing 15.20 g of urea, (molar mass = 60) dissolved in 150 g of water is
(A) 1.6889 mol kg-1 (B) 0.1689 mol kg-1 (C) 0.5922 mol kg-1 (D) 0.2533 mol kg-1 Correct Answer: (A) 1.6889 mol kg-1 Explanation:...
The acid which contains both ‑OH and ‑COOH groups is
(A) phthalic acid (B) adipic acid (C) glutaric acid (D) salicylic acid Correct Answer: (D) salicylic acid Explanation: The acid which contains both ‑OH and ‑COOH groups is...
Identify the compound in which phosphorus exists in the oxidation state of +1.
(A) Phosphonic acid (H3PO3) (B) Phosphinic acid (H3PO2) (C) Pyrophosphorus acid (H4P2O5) (D) Orthophosphoric acid (H3PO4) Correct Answer: (B) Phosphinic acid (H3PO2) Explanation:...
(+) 2‑ Methylbutan ‑1‑ol and (‑)2‑Methylbutan‑1‑ol have different values for which property?
(A) Boiling point (B) Relative density (C) Refractive index (D) Specific rotation Correct Answer: (D) Specific rotation Explanation:...
Which among the following is not a mineral of iron?
(A)Haematite (B) Magnesite (C)Magenetite (D)Siderite Correct Answer: (B) Magnesite Explanation: Magnesite is not a mineral of iron.
Nitration of which among the following compounds yields cyclonite?
(A) Formaldehyde (B) benzaldehyde (C) Urotropine (D) Acetaldehyde ammonia Correct Answer: (A) Formaldehyde Explanation: The nitration of the compound formaldehyde yields...
How is sodium chromate converted into sodium dichromate in the manufacture of potassium dichromate from chromite ore?
A) By the action of concentrated sulphuric acid B) By roasting with soda ash C) By the action of sodium hydroxide D) By the action of lime stone Correct Answer: A) By the action of...
In dry cell, what acts as negative electrode?
A) Zinc B) Graphite C) Ammonium chloride D) Manganese dioxide Correct Answer: A) Zinc Explanation: Zinc-carbon battery is a dry cell battery between a zinc metal electrode and a carbon fiber...
Select the compound which on treatment with nitrous acid liberates nitrogen.
A) Nitroethane B) Triethylamine C) Diethylamine D) Ethylamine Correct Answer: D) Ethylamine Explanation: Ethylamine is the compound which on treatment with nitrous acid liberates nitrogen....
5.0 g of sodium hydroxide (molar mass 40 g mol-1) is dissolved in little quantity of water and the solution is diluted up to 100 ml. What is the molarity of the resulting solution?
A) 0.1 mol dm -3 B) 1.0 mol dm-3 C) 0.125 mol dm-3 D) 1.25 mol dm-3 Correct Answer: D) 1.25 mol dm-3 Explanation: Calculation - Moles of solute is 5/40 => 0.125 moles Volume of the...
Which of the following compounds when treated with dibenzyl cadmium yields benzyl methyl ketone?
A) Acetone B) Acetaldehyde C) Acetic acid D) Acetyl chloride Correct Answer: D) Acetyl chloride Explanation: Acetyl chloride is the compound when treated with dibenzyl cadmium yields benzyl...
Which halide of magnesium has highest ionic character?
A) Chloride B) Bromide C) Iodide D) Fluoride Correct Answer: D) Fluoride Explanation: Fluoride is the halide of magnesium has highest ionic character.
The reaction takes place in two steps as
i) NO2Cl(g) ⎯k1→ NO2 (g) + Cl (g) ii) NO2Cl(g) + Cl (g) ⎯k2⎯→ NO2 (g) + Cl2 (g) Identify the reaction intermediate A) NO2Cl(g) B) NO2 (g) C) Cl2 (g) D) Cl (g) Correct Answer: D) Cl (g)...
Which of the following amino acids is basic in nature?
A) Valine B) Tyrosine C) Arginine D) Leucine Correct Answer: C) Arginine Explanation: Arginine is the amino acids that is basic in nature.
Which among the following phenolic compounds is most acidic in nature?
A) p-aminophenol B) phenol C) m-nitrophenol D) p-nitrophenol Correct Answer: D) p-nitrophenol Explanation: p-nitrophenol is the phenolic compounds is most acidic in nature.
The relation between solubility of a gas in liquid at constant temperature and external pressure is stated by which law?
A) Raoult’s law B) van’t Hoff Boyle’s law C) van’t Hoff Charles’ law D) Henry’s law Correct Answer: D) Henry’s law Explanation: Henry’s law is the law that states the relation between...
Which among the following solids is a nonpolar solid?
A) Hydrogen chloride B) Sulphur dioxide C) Water D) Carbon dioxide Correct Answer: D) Carbon dioxide Explanation: Carbon dioxide is a nonpolar solid. The bonds that are part of the ionic are...
Identify the metal that forms colourless compounds.
A) Iron (Z = 26) B) Chromium (Z = 24) C) Vanadium (Z = 23) D) Scandium (Z = 21) Correct Answer: D) Scandium (Z = 2) Explanation: Scandium (Z = 2) is the metal that forms colourless...
What is the highest oxidation state exhibited by group 17 elements?
A) + 1 B) + 3 C) + 5 D) + 7 Correct Answer: D) + 7 Explanation: The highest oxidation state exhibited by group 17 element is +7.
Mathematical equation of first law of thermodynamics for isochoric process is
A) ΔU=qv B) −ΔU =qv C) q =−W D) ΔU=W Correct Answer: A) ΔU=qv Explanation: The mathematical equation of first law of thermodynamics for isochoric process is ΔU=qv
Name the catalyst used in commercial method of preparation of phenol.
A) Silica B) Calcium phosphate C) Anhydrous aluminium chloride D) Cobalt naphthenate Correct Answer: D) Cobalt naphthenate Explanation: Cobalt naphthenate is the catalyst that is used in...
The rate constant and half-life of a first order reaction are related to each other as
A) t1/2=0.693/K B) t1/2=0.693K C) K=0.693 t1/2 D) K t1/2=1/0.693 Correct Answer: A) t1/2=0.693/K Explanation: The rate constant and half-life of a first order reaction are related to each...
What is the combining ratio of glycerol and fatty acids when they combine to form triglyceride?
A) 3 : 4 B) 3 : 2 C) 1 : 3 D) 1 : 2 Correct Answer: C) 1 : 3 Explanation: The combining ratio of glycerol and fatty acids when they combine to form triglyceride is 1 : 3.
The molecular formula of Wilkinson catalyst, used in hydrogenation of alkenes is
A) Co (CO)8 B) (Ph3P)3 RhCl C) [Pt (NH3)2 Cl2] D) K [Ag (CN)2] Correct Answer: B) (Ph3P)3 RhCl Explanation: The molecular formula of Wilkinson catalyst which is used in hydrogenation of...
The criterion for a spontaneous process is
A) ΔG>0 B) ΔG< 0 C) ΔG=0 D) ΔSTotal<0 Correct Answer: B) ΔG< 0 Explanation: The criterion for a spontaneous process is ΔG< 0
Brown ring test is used for detection of which radical?
A) Ferrous B) Nitrite C) Nitrate D) Ferric Correct Answer: C) Nitrate Explanation: Brown ring test is used for detection of Nitrate.
The reagent used in Wolff-Kishner reduction is
A) NH2 – NH2 and KOH in ethylene glycol B) Zn – Hg/conc.HCl C) NaBH4 D) Na – Hg/H2O Correct Answer: A) NH2 – NH2 and KOH in ethylene glycol Explanation: The reagent used in Wolff-Kishner...
Which of the following is a neutral complex?
A) [Pt (NH3)2 Cl2] B) [Co (NH3)6] Cl3 C) [Ni (NH3)6] Cl2 D) K4 [Fe (CN)6] Correct Answer: A) [Pt (NH3)2 Cl2] Explanation: [Pt (NH3)2 Cl2] is a neutral complex.
Identify the compound amongst the following of which 0.1 M aqueous solution has highest boiling point.
A) Glucose B) Sodium chloride C) Calcium chloride D) Ferric chloride Correct Answer: D) Ferric chloride Explanation: Ferric chloride is the compound in which 0.1M aqueous solution has the...
What is the reagent used in Etard reaction?
A) Chromyl chloride B) Ethanoyl chloride C) SnCl2 and HCl D) Cadmium chloride Correct Answer: A) Chromyl chloride Explanation: Chromyl chloride is the reagent used in Etard...
The most abundant noble gas in atmosphere is
A) Neon B) Argon C) Xenon D) Krypton Correct Answer: B) Argon Explanation: Argon is the most abundant noble gas in the atmosphere.
Identify an extensive property amongst the following
A) Viscosity B) Heat capacity C) Density D) Surface tension Correct Answer: B) Heat capacity Explanation: Heat capacity is an extensive property.
Which of the following carboxylic acids is a tricarboxylic acid?
A) Oxalic acid B) Citric acid C) Succinic acid D) Adipic acid Correct Answer: B) Citric acid Explanation: Citric acid is a weak tricarboxylic acid that occurs mostly in the citrus...
Average rate of reaction 2 SO2 (g) + O2 (g) ⎯→ 2 SO3 (g) is written as
A) Δ[SO2]/ Δt B) – Δ[O2]/ Δt C) ½ Δ[SO2]/ Δt D) Δ[SO3]/ Δt Correct Answer: B) – Δ[O2]/ Δt Explanation: Average rate of reaction 2 SO2 (g) + O2 (g) ⎯⎯ → 2 SO3 (g) is written as – Δ[O2]/...
What is the amount of work done when 0.5 mole of methane, CH4 (g), is subjected to combustion at 300 K? (given, R = 8.314 J K-1 mol-1)
A) – 2494 J B) – 4988 J C) + 4988 J D) + 2494 J Correct Answer: D) + 2494 J Explanation: Using the formula W = −PΔV, + 2494 J is the amount of work done when 0.5 mole of methane, CH4 (g), is...
Primary nitroalkanes are obtained in good yield by oxidising aldoximes with the help of
A) trifluoro peroxyacetic acid B) acidified potassium permanganate C) concentrated nitric acid D) potassium dichromate and dilute sulphuric acid Correct Answer: A) trifluoro peroxyacetic acid...
If ‘n’ represents total number of asymmetric carbon atoms in a compound, the possible number of optical isomers of the compound is
A) 2n B) n2 C) 2n D) 2n + 2 Correct Answer: C) 2n Explanation: General formula for calculating stereoisomer - 2n, where n is the number of chiral centers. The stereoisomers have same chemical...
Which is the most stable allotrope of Sulphur?
A) Octahedral sulphur B) Monoclinic Sulphur C) Plastic Sulphur D) Colloidal Sulphur Correct Answer: A) Octahedral sulphur Explanation: Sulfur mainly exists in two types of allotropes: Rhombic...
Correct statement for thermoplastic polymer is
A) It does not become soft on heating under pressure B) It cannot be remolded C) It is either linear or branched chain polymer D) It is cross-linked polymer Correct Answer: C) It is either...
How many Faradays of electricity are required to deposit 10 g of calcium from molten calcium chloride using inert electrodes? (molar mass of calcium = 40 g mol-1)
A) 0.5 F B) 1 F C) 0.25 F D) 2 F Correct Answer: A) 0.5 F Explanation: Reaction Involved – Ca2+ + 2e- Ca Calculation - Number of moles of Ca = $\frac{{10g}}{{40g/mol}}$ => 0.25mol...
Name the reagent that is used in leaching of gold
A) Carbon B) Sodium cyanide C) Carbon monoxide D) Iodine Correct Answer: B) Sodium cyanide Explanation: Sodium cyanide is the reagent that is used in leaching of gold.
Which of the following is an analgesic?
A) Ofloxacin B) Penicillin C) Aminoglycosides D) Paracetamol Correct Answer: D) Paracetamol Explanation: Paracetamol is an analgesic.
The compound which is not formed when a mixture of n-butyl bromide and ethyl bromide treated with sodium metal in presence of dry ether is
A) Butane B) Octane C) Hexane D) Ethane Correct Answer: D) Ethane Explanation: The compound which is not formed when a mixture of n-butyl bromide and ethyl bromide treated with sodium metal...
What is the general molecular formula of the products obtained on heating lanthanoids (Ln) with Sulphur?
A) LnS B) LnS3 C) Ln3S2 D) Ln2S3 Correct Answer: D) Ln2S3 Explanation: Reaction Involved – 2Ln + 3S Ln2S3
Butylated hydroxy anisole is
A) an anti-oxidant B) cleansing agent C) disinfectant D) an antihistamine Correct Answer: A) an anti-oxidant Explanation: Butylated hydroxy anisole is an anti-oxidant.
In the cell represented by Pb(1) |Pb2+ (1M)||Ag+(1), the reducing agent is
A) Pb B) Pb2+ C) Ag D) Ag+ Correct Answer: A) Pb Explanation: Reaction Involved – Pb Pb2+ + 2e-
Which metal crystallizes in a simple cubic structure?
A) Polonium B) Copper C) Nickel D) Iron Correct Answer: A) Polonium Explanation: Polonium is the metal that crystallizes in a simple cubic structure.
The amine ‘A’ when treated with nitrous acid gives yellow oily substance. The amine A is
A) trimethylamine B) trimethylamine C) aniline D) methyl phenylamine Correct Answer: D) methyl phenylamine Explanation: Reaction Involved: Ph – NH – CH3 + OH – N = O Ph – N(CH3) – N = O +...
The element that does not form acidic oxide is
A) Carbon B) Phosphorus C) Chlorine D) Barium Correct Answer: D) Barium Explanation: Barium is the element that does not form acidic oxide.
While assigning R, S configuration the correct order of priority of groups attached to chiral carbon atom is
A) CONH2 > COCH3 > CH2OH > CHO B) CONH2 > COCH3 > CHO > CH2OH C) COCH3 > CONH2 > CHO > CH2OH D) CHO > CH2OH > COCH3 > CONH2 Correct Answer: B) CONH2 >...
Bulletproof helmets are made from
A) Lexan B) Saran C) Glyptal D) Thiokol Correct Answer: A) Lexan Explanation: Bulletproof helmets are made from Lexan.
Which metal is refined by Mond Process?
A) Titanium B) Copper C) Nickel D) Zinc Correct Answer: C) Nickel Explanation: Nickel is the metal which is refined by Mond process. Steps Involved: NiO(s) + H2(g) Ni(s) + H2O Ni(s) +...
Isopropyl methyl ether when treated with cold hydrogen iodide gives
A) isopropyl iodide and methyl iodide B) isopropyl alcohol and methyl iodide C) isopropyl alcohol and methyl alcohol D) isopropyl iodide and methyl alcohol Correct Answer: B) isopropyl...
In face centered cubic unit cell, what is the volume occupied?
A) 4/3 πr3 B) 8/3 πr3 C) 16/3 πr3 D) 64 πr3/3 Correct Answer: C) 16/3 πr3 Explanation: Calculation - In fcc unit cell, no. of atoms are = [6×1/2]+[8×1/8] => 4 Volume of 1 spherical atom...
Glucose on oxidation with bromine water yields gluconic acid. This reaction confirms presence of
A) six carbon atoms linked in straight chain B) secondary alcoholic group in glucose C) aldehyde group in glucose D) primary alcoholic group in glucose Correct Answer: C) aldehyde group in...
The schematic symbol of light emitting diode is (LED)
Solution: The correct Answer is B.
In balanced metre bridge, the resistance of bridge wire is 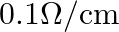 . Unknown resistance ‘
. Unknown resistance ‘ 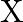 ‘ is connected in left gap and
‘ is connected in left gap and 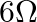 in right gap, null point divides the wire in the ratio
in right gap, null point divides the wire in the ratio 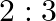 . Find the current drawn from the battery of
. Find the current drawn from the battery of 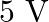 having negligible resistance.
having negligible resistance.
A) 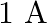
B) 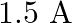
C) 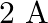
D) 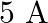
Answer is (A) $\frac{\ell_{1}}{\ell_{2}}=\frac{2}{3}=\frac{\mathrm{v}_{1}}{\mathrm{v}_{2}}$ Now $\quad \mathrm{v}_{1}+\mathrm{v}_{2}=\mathrm{v}=5 \mathrm{~V} \quad \mathrm{QV}=\mathrm{IR}$ For...
The magnetic field (B) inside a long solenoid having ‘n’, turns per unit length and carrying current ‘I’ when iron core is kept in it is 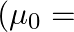 permeability of vacuum,
permeability of vacuum, 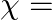 magnetic susceptibility)
magnetic susceptibility)
A) 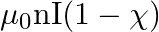
B) 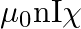
C) 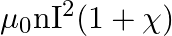
D) 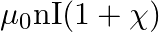
Answer is (D) $\mathrm{B}=\mu_{0} \mathrm{ni}(1+\chi)$
When an electron in Hydrogen atom revolves in stationary orbit, it
A) does not radiate light though its velocity changes
B) does not radiate light and velocity remains unchanged
C) radiates light but its velocity is unchanged
D) radiates light with the change of energy
Answer is (A) For an electron revolving in stationary orbits it doesn't radiate light through is velocity change.
Two particles  and
and 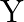 having equal charges after being accelerated through same potential difference enter a region of uniform magnetic field and describe a circular paths of radii ‘
having equal charges after being accelerated through same potential difference enter a region of uniform magnetic field and describe a circular paths of radii ‘ 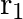 ‘ and ‘
‘ and ‘ 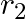 ‘ respectively. The ratio of the mass of
‘ respectively. The ratio of the mass of 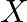 to that of
to that of 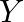 is
is
A) 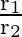
B) 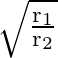
C) ![Rendered by QuickLaTeX.com \left[\frac{\mathrm{r}_{2}}{\mathrm{r}_{1}}\right]^{2}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-8576e94466b638e76d49f6a15d3e2940_l3.png)
D) ![Rendered by QuickLaTeX.com \left[\frac{\mathrm{r}_{1}}{\mathrm{r}_{2}}\right]^{2}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-508e81114cccf8287500c32565603618_l3.png)
Answer is (A) The force acting on the particle inside magnetic field is $F_{B}=q v B \sin \theta$ This provides the necessary centripetal force $F_{c}=\frac{m v^{2}}{r}$ $\begin{array}{l} \therefore...
From Brewster’s law, except for polished metallic surfaces, the polarising angle
A) depends on wavelength and is different for different colours
B) independent of wavelength and is different for different colours
C) independent of wavelength and is same for different colours
D) depends on wavelength and is same for different colours
Answer is (A) According to Brewster's law, polarizing angle depends on wavelength and is different for different colors.
When light of wavelength ‘ 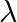 ‘ is incident on photosensitive surface, the stopping potential is ‘
‘ is incident on photosensitive surface, the stopping potential is ‘ 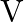 ‘. When light of wavelength ‘
‘. When light of wavelength ‘ 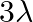 ‘ is incident on same surface, the stopping potential is
‘ is incident on same surface, the stopping potential is 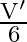 . Threshold wavelength for the surface is
. Threshold wavelength for the surface is
A) 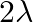
B) 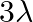
C) 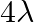
D) 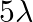
Answer is (D) Using $\frac{\mathrm{hc}}{\lambda}-\phi=\mathrm{eV}$ When, wavelength $\lambda$ is incident on metallic surface, the stopping potential required to stop most energetic electron is...
Resolving power of telescope increases when
A) wavelength of light decreases
B) wavelength of light increases
C) focal length of eye-piece increases
D) focal length of eye-piece decreases
Answer is (A) Resolving power of telescope is given by $\mathrm{P}=\frac{\mathrm{d}}{1.22 \lambda}$ When $\lambda$ decreases, resolving power decreases and vice versa
Magnetic flux passing through a coil is initially 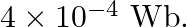 It reduces to
It reduces to 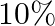 of its Loriginal value in ‘t’ second. If the e.m.f. induced is
of its Loriginal value in ‘t’ second. If the e.m.f. induced is 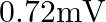 then ‘t’ in second is
then ‘t’ in second is
A) 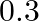
B) 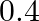
C) 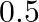
D) 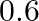
Answer is (C) $\begin{array}{l} \phi_{1}=4 \times 10^{-4} \mathrm{wb} \text { and } \phi_{2}=0.1 \times \phi_{1}=4 \times 10^{-5} \mathrm{wb} \\ \omega=0.72 \mathrm{~Hz} \\ \therefore \quad...
The amount of work done in increasing the voltage across the plates of capacitor from 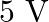 to
to 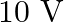 is ‘
is ‘ 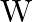 ‘. The work done in increasing it from
‘. The work done in increasing it from 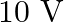 to
to 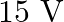 will be
will be
A) 
B) 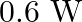
C) 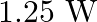
D) 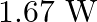
Answer is (D) The work done in increasing the potential is given by $\mathrm{W}=\mathrm{d} \mathrm{U} \propto\left(\mathrm{V}_{2}^{2}-\mathrm{V}_{1}^{2}\right)$ $\therefore...
Two coherent sources ‘P’ and ‘Q’ produce interference at point ‘A’ on the screen where there is a dark band which is formed between 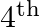 bright band and
bright band and 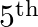 bright band. Wavelength of light used is
bright band. Wavelength of light used is 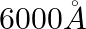 . The path difference between PA and QA is
. The path difference between PA and QA is
A) 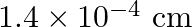
B) 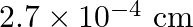
C) 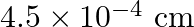
D) 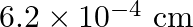
Answer is (B) Here $\Delta \mathrm{x}=\frac{\lambda \delta}{2 \pi}$ For $n=4^{\text {th }}$ dark bone $\delta=(2 n+1) \pi \Rightarrow \delta=(8+1) \pi=9 \pi$ $\begin{array}{l} \Delta x=\frac{\lambda...
A galvanometer of resistance 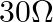 is connected to a battery of emf
is connected to a battery of emf 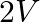 with
with 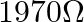 resistance in series. A full scale deflection of 20 divisions is obtained in the galvanometer. To reduce the deflection to 10 divisions, the resistance in series required is
resistance in series. A full scale deflection of 20 divisions is obtained in the galvanometer. To reduce the deflection to 10 divisions, the resistance in series required is
A) 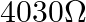
B) 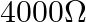
C) 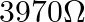
D) 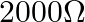
Answer is (C) $\mathrm{i}=\frac{\mathrm{V}}{\mathrm{R}_{\mathrm{eff}}}=\frac{\mathrm{V}}{1790+30}=\frac{2}{2000}=1 \times 10^{-3} \mathrm{~A}=1 \mathrm{~mA}$ This current provides full scale...
The LC parallel resonant circuit
A) has a very high impedance B) has a very high current
C) acts as resistance of very low value
D) has zero impedance
Answer is (A) LC parallel resonant circuit has very high impedance.
Two identical parallel plate air capacitors are connected in series to a battery of e.m.f. ‘V’. If one of the capacitor is completely filled with dielectric material of constant ‘ 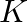 ‘, then potential difference of the other capacitor will become
‘, then potential difference of the other capacitor will become
A) 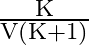
B) 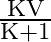
C) 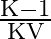
D) 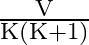
Answer is (B) When capacitors are connected is series $C_{e q}=\frac{C_{1} C_{2}}{C_{1}+C_{2}}$ and the potential areas plates of capacitors is given by $\mathrm{V}=\frac{\mathrm{Q}}{\mathrm{C}}$ If...
In Bohr’s theory of Hydrogen atom, the electron jumps from higher orbit ‘ 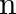 ‘ to lower orbit, ‘p’. The wavelength will be minimum for the transition
‘ to lower orbit, ‘p’. The wavelength will be minimum for the transition
A)  to
to 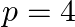
B) 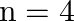 to
to 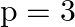
C) 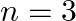 to
to 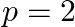
D) 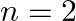 to
to 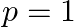
Answer is (A) According to Bohr's theory the wavelength of radiation emitted is given by $\frac{1}{\lambda}=R\left(\frac{1}{\mathrm{ne}^{2}}-\frac{1}{\mathrm{ne}^{2}}\right)$ For $n_{i}=5$ and...
The maximum frequency of transmitted radio waves above which the radio waves are no longer reflected back by ionosphere is – 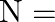 maximum electron density of ionosphere,
maximum electron density of ionosphere, 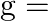 acceleration due to gravity)
acceleration due to gravity)
A) 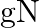
B) 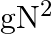
C) 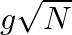
D) 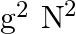
Answer is (C) For ionosphere, the maximum frequency of radio waves that can be reflected back is given by $g \sqrt{N}$. Where $N$ is maximum electron density of ionosphere and $g$ is acceleration...
A ray of light travelling through rarer medium is incident at very small angle ‘i’ on a glass slab and after refraction its velocity is reduced by 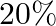 . The angle of deviation is
. The angle of deviation is
A) 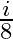
B) 
C) 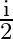
D) 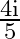
Answer is (B) As at interface, velocity of light reduces by $20 \%$ of $\mathrm{C}$ $\therefore \quad \delta=20 \% \mathrm{i}=\frac{20}{100} \times \mathrm{i}=\frac{\mathrm{i}}{5}$
Light of wavelength ‘ 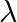 ‘ which is less than threshold wavelength is incident on a photosensitive material. If incident wavelength is decreased so that emitted photoelectrons are moving with same velocity then stopping potential will
‘ which is less than threshold wavelength is incident on a photosensitive material. If incident wavelength is decreased so that emitted photoelectrons are moving with same velocity then stopping potential will
A) increase
B) decrease
C) be zero
D) become exactly half
Answer is (A) According to photoelectric equation, $\frac{\mathrm{hc}}{\lambda}-\phi=\mathrm{E} \quad$ where $\mathrm{E}=\frac{\mathrm{p}^{2}}{2 \mathrm{~m}}(\mathrm{~K} . \mathrm{E})$ If $E$ is...
In an oscillator, for sustained oscillations, Barkhausen criterion is 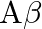 equal to
equal to 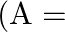 voltage gain without feedback,
voltage gain without feedback, 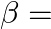 feedback factor )
feedback factor )
A) zero
B) 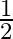
C) 1
D) 2
Answer is (A) For sustained oscillations according to Barkhausen criteria. $A \cdot \beta=1$
Three parallel plate air capacitors are connected in parallel. Each capacitor has plate area 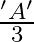 and the separation between the plates is ‘
and the separation between the plates is ‘ 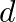 ‘, ‘
‘, ‘ 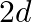 ‘ and ‘ 3 d’ respectively. The equivalent capacity of combination is
‘ and ‘ 3 d’ respectively. The equivalent capacity of combination is 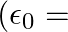 absolute permittivity of free space)
absolute permittivity of free space)
A) 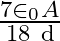
B) 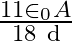
C) 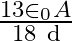
D) 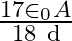
Answer is (B) $\mathrm{C}=\mathrm{QV} \quad$ and $\mathrm{C}=\frac{\varepsilon_{0} \mathrm{~A}}{\mathrm{~d}}$ As given $\mathrm{C}_{1}=\frac{\varepsilon_{0} \cdot \mathrm{A}}{3 \mathrm{~d}} ; \quad...
Interference fringes are produced on a screen by using two light sources of intensities ‘I’ and ‘9I’. The phase difference between the beams is 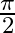 at point
at point 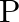 and
and 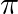 at point
at point 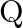 on the screen. The difference between the resultant intensities at point
on the screen. The difference between the resultant intensities at point 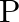 and
and  is
is
A) 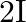
B) 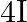
C) 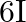
D) 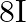
Answer is (C) At point $\mathrm{P}$, resultant intensity of interfering wave is $\left(\mathrm{I}_{\mathrm{p}}\right)_{\mathrm{res}}=\mathrm{I}_{1}+\mathrm{I}_{2}+2 \sqrt{\mathrm{I}_{1}...
An electron of mass ‘ 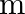 ‘ has de-Broglie wavelength ‘
‘ has de-Broglie wavelength ‘ 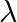 ‘ when accelerated through potential difference ‘
‘ when accelerated through potential difference ‘ 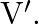 When proton of mass ‘
When proton of mass ‘ 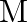 ‘, is accelerated through potential difference ‘
‘, is accelerated through potential difference ‘ 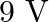 ‘, the de-Broglie wavelength associated with it will be (Assume that wavelength is determined at low voltage)
‘, the de-Broglie wavelength associated with it will be (Assume that wavelength is determined at low voltage)
A) 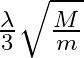 B)
B) 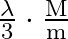
C) 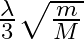
D) 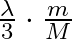
Answer is (C) When electron or any charged particle is accelerated through potential difference $v$, then kinetic energy gained is given by $E=e V$ $\begin{array}{l} E=\frac{1}{2} m...
Alternating current of peak value 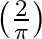 ampere flows through the primary coil of the transformer. The coefficient of mutual inductance between primary and secondary coil is 1 henry. The peak e.m.f. induced in secondary coil is (Frequency of a.c.
ampere flows through the primary coil of the transformer. The coefficient of mutual inductance between primary and secondary coil is 1 henry. The peak e.m.f. induced in secondary coil is (Frequency of a.c. 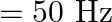 )
)
A) 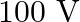
B) 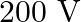
C) 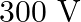
D) 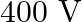
Answer is (B) Peak value of current $I_{0}=1_{\operatorname{nms}} \times\sqrt{2}=\left(\frac{2}{\pi}\right)$ Amp Co-efficient of mutual inductance is $M=1$ horn Induced emf in secondary is given by...
An iron rod is placed parallel to magnetic field of intensity 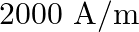 . The magnetic flux through the rod is
. The magnetic flux through the rod is 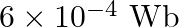 and its cross-sectional area is
and its cross-sectional area is 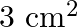 . The magnetic permeability of the rod in
. The magnetic permeability of the rod in 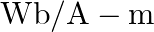 is
is
A) 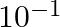
B) 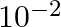
C) 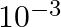
D) 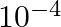
Answer is (C) $\mu=\mathrm{B} \cdot \mathrm{H}=\frac{\phi}{\mathrm{A}} \times \mathrm{H}=\frac{6 \times 10^{-4}}{3 \times 10^{-4}} \times \frac{1}{2 \times 10^{3}}=10^{-3}$
In potentiometer experiment, null point is obtained at a particular point for a cell on potentiometer wire  long. If the length of the potentiometer wire is increased without changing the cell, the balancing length will (Driving source is not changed)
long. If the length of the potentiometer wire is increased without changing the cell, the balancing length will (Driving source is not changed)
A) increase
B) decrease
C) not change
D) becomes zero
Answer is (A) For potentiometer, when null point is obtained for a particular cell (EV) at L $\mathrm{cm}$, (say). Whose length is $\mathrm{x} \mathrm{cm} \quad \therefore \mathrm{E}=\mathrm{L}...
The value of gravitational acceleration ‘ 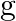 ‘ at a height ‘h’ above the earth’s surface is
‘ at a height ‘h’ above the earth’s surface is 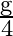 then
then 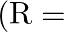 radius of earth)
radius of earth)
A) 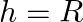
B) 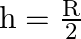
C) 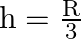
D) 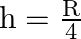
Answer is (A) $g^{\prime}=g\left(\frac{R}{R+h}\right)^{2}$ When $g^{\prime}=\frac{g}{4}$ then. $\begin{array}{l} \frac{g}{4}=g \times\left(\frac{R}{R+h}\right)^{2} \Rightarrow...
Angular speed of hour hand of a clock in degree per second is
A) 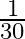
B) 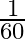
C) 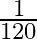
D) 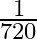
Answer is (C) $\omega=\frac{\theta}{t}=\frac{2 \pi^{c}}{12 \times 3600}=\frac{360^{\circ}}{12 \times 3600}=\frac{1}{12 \times 10}=\frac{1}{120} \mathrm{deg} / \mathrm{sec}$
A simple pendulum of length ‘ 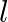 ‘ has maximum angular displacement ‘
‘ has maximum angular displacement ‘ 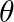 ‘. The maximum kinetic energy of the bob of mass ‘
‘. The maximum kinetic energy of the bob of mass ‘ 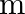 ‘ is (g = acceleration due to gravity)
‘ is (g = acceleration due to gravity)
A) 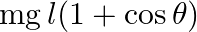
B) 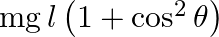
C) 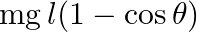
D) 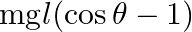
Answer is (C) When bob is at rest, the pendulum has only potential energy which is given as $\mathrm{P.E}=\mathrm{mg} \ell$. When bob displaces by small angler displacement $\theta$, the pendulum...
Let ‘ 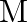 ‘ be the mass and ‘
‘ be the mass and ‘ 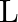 ‘ be the length of a thin uniform rod. In first case, axis of rotation is passing through centre and perpendicular to the length of the rod. In second case axis of rotation is passing through one end and perpendicular to the length of the rod. The ratio of radius of gyration in first case to second case is
‘ be the length of a thin uniform rod. In first case, axis of rotation is passing through centre and perpendicular to the length of the rod. In second case axis of rotation is passing through one end and perpendicular to the length of the rod. The ratio of radius of gyration in first case to second case is
A) 1
B) 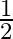
C) 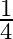
D) 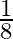
Answer is (B) M.I of rod whose axis of rotation is passing through center and perpendicular to the plane of rod is $\mathrm{I}=\frac{\mathrm{ML}^{2}}{12} \quad$ and $\quad...
A liquid drop having surface energy ‘ 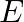 ‘ is spread into 512 droplets of same size. The final surface energy of the droplets is
‘ is spread into 512 droplets of same size. The final surface energy of the droplets is
A) 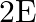
B) 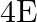
C) 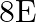
D) 
Answer is (C) The surface area of the liquid drop is $A=4 \pi R^{2}$ Its surface energy is $\mathrm{E}$ When the drop splits in 512 droplets, the surface area of each droplets is $A_{2}=512 \times...
A black rectangular surface of area ‘A’ emits energy ‘E’ per second at 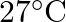 . If length and breadth are reduced to
. If length and breadth are reduced to 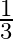 rd of initial value and temperature is raised to
rd of initial value and temperature is raised to 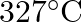 then energy emitted per second becomes
then energy emitted per second becomes
A) 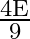
B) 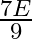
C) 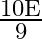
D) 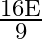
Answer is (D) $\mathrm{E}=\mathrm{e} \sigma \cdot \mathrm{A}\left(\mathrm{T}^{4}-\mathrm{T}_{0}^{4}\right) \text { and } \mathrm{A}=\ell \mathrm{b}$ When $\ell$ and $b$ changes to $\frac{\ell}{3}$...
Two particles of masses ‘ 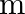 ‘ and ‘
‘ and ‘ 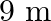 ‘ are separated by a distance ‘
‘ are separated by a distance ‘ 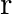 ‘. At a point on the line joining them the gravitational field is zero. The gravitational potential at that point is (G = Universal constant of gravitation)
‘. At a point on the line joining them the gravitational field is zero. The gravitational potential at that point is (G = Universal constant of gravitation)
A) 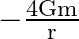
B) 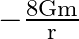
C) 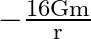
D) 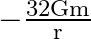
Answer is (C) For the system described above $\mathrm{U}=\mathrm{m} \cdot \mathrm{V} \quad \therefore \mathrm{V}=\frac{\mathrm{U}}{\mathrm{m}}=\frac{-\mathrm{GM}}{\mathrm{X}}$ Here $M=16...
A progressive wave is represented by 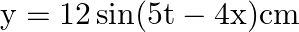 . On this wave, how far away are the two points having phase difference of
. On this wave, how far away are the two points having phase difference of 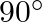 ?
?
A) 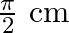
B) 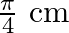
C) 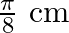
D) 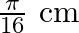
Answer is (C) $y=12 m(5 t-4 y)$ Comparing in $y=A \sin (w t-k x)$ We have $A=12, w=5$ and $k=4$ $(w t-k x)=$ phase difference $=\frac{\pi}{2}$ $\therefore \quad 5 t-4 x=\frac{\pi}{2}$ When $t=0,4...
If the end correction of an open pipe is 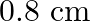 then the inner radius of that pipe will be
then the inner radius of that pipe will be
A) 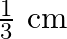
B) 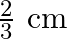
C) 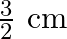
D) 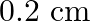
Answer is (B) For open organ pipe $\begin{array}{l} \Delta \mathrm{L}=1.2 \times \mathrm{r} \\ \therefore \mathrm{r}=\Delta \mathrm{L} \times \frac{1}{1.2}=\frac{0.8}{1.2}=\frac{2}{3} \mathrm{~cm} ....
Which of the following quantity does NOT change due to damping of oscillations?
A) Angular frequency
B) Time period
C) Initial phase
D) Amplitude
Answer is (C) $\delta$ is the initial phase. When oscillations of pendulum are damped, $\delta$ doesn't change
Let a steel bar of length ‘ 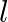 ‘, breadth ‘b’ and depth ‘d’ be loaded at the centre by a load ‘
‘, breadth ‘b’ and depth ‘d’ be loaded at the centre by a load ‘ 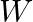 ‘. Then the sag of bending of beam is (
‘. Then the sag of bending of beam is ( 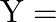 Young’s modulus of material of steel)
Young’s modulus of material of steel)
A) 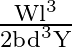
B) 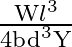
C) 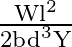
D) 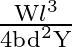
Answer is (B) The sag of bending of beam is given by. $\delta=\frac{W L^{3}}{48 \mathrm{YI}} \quad$ Where $W$ is load, $L$ is length, $Y$ is young's modules and $I$ is $\frac{b d^{3}}{12}$ (area...
A disc of radius ‘  ‘ and thickness
‘ and thickness 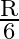 has moment of inertia ‘I’ about an axis passing through its centre and perpendicular to its plane. Disc is melted and recast into a solid sphere. The moment of inertia of a sphere about its diameter is
has moment of inertia ‘I’ about an axis passing through its centre and perpendicular to its plane. Disc is melted and recast into a solid sphere. The moment of inertia of a sphere about its diameter is
A) 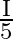
B) 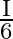
C) 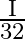
D) 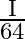
Answer is (A) Volume of disc is $\mathrm{A} \cdot \mathrm{d}=\pi \cdot \mathrm{R}^{2} \times \frac{\mathrm{R}}{6}=\frac{\mathrm{R}^{3} \times \pi}{6}$ Moment of inertia of disc is...
When open pipe is closed from one end then third overtone of closed pipe is higher in frequency by 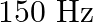 than second overtone of open pipe. The fundamental frequency of open end pipe will be A)
than second overtone of open pipe. The fundamental frequency of open end pipe will be A) 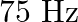 B)
B) 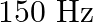 C)
C) 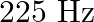 D)
D) 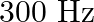
Answer is (D) Let $f_{0}=\frac{v_{0}}{2 L}$ be the fundamental frequency of the open pipe $\therefore$ is second overtone is $\frac{3}{2} f_{0}=\frac{3 v_{0}}{2 L}$ Let...
In a capillary tube of radius ‘ 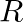 ‘, a straight thin metal wire of radius ‘
‘, a straight thin metal wire of radius ‘ 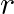 ‘
‘ 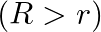 is inserted symmetrically and one end of the combination is dipped vertically in water such that the lower end of the combination is at same level. The rise of water in the capillary tube is
is inserted symmetrically and one end of the combination is dipped vertically in water such that the lower end of the combination is at same level. The rise of water in the capillary tube is  surface tension of water,
surface tension of water,  density of water,
density of water,  gravitational acceleration
gravitational acceleration ![Rendered by QuickLaTeX.com ]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-478d84af1027ee3593251b323a3fb181_l3.png)
A) 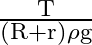
B) 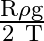
C) 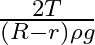
D) 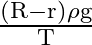
Answer is (C) $\mathrm{h}=\frac{2 \mathrm{~T} \cos \theta}{\rho g(\mathrm{R}-\mathrm{r})}$ For $\cos \theta=0, \mathrm{~h}=\frac{2 \mathrm{~T}}{\rho g(\mathrm{R}-\mathrm{r})}$
The bob of a simple pendulum performs S.H.M. with period ‘ 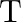 ‘ in air and with period ‘
‘ in air and with period ‘ 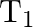 , in water. Relation between ‘
, in water. Relation between ‘ 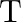 ‘ and ‘
‘ and ‘ 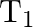 ‘ is (neglect friction due to water, density of the material of the bob is
‘ is (neglect friction due to water, density of the material of the bob is 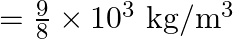 , density of water
, density of water 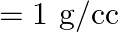 )
)
A) 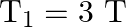
B) 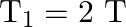
C) 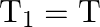
D) 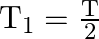
Answer is (A) $\mathrm{T}=2 \pi \sqrt{\frac{\ell}{\mathrm{g}_{\mathrm{eff}}}}$ In air $\mathrm{g}_{\text {eff }}=\mathrm{g}$ where as in water $\begin{array}{l} g_{\text {eff...
Two strings 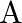 and
and 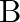 of same material are stretched by same tension. The radius of the string A is double the radius of string B. Transverse wave travels on string A with speed ‘
of same material are stretched by same tension. The radius of the string A is double the radius of string B. Transverse wave travels on string A with speed ‘ 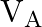 ‘ and on string
‘ and on string 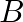 with speed ‘
with speed ‘ 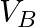 ‘. The ratio
‘. The ratio 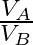 is
is
A) 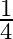
B) 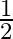
C) 2
D) 4
Answer is (C) The velocity of wave travelling on string is $\begin{array}{l} v=n \lambda=\frac{\lambda}{2 L} \sqrt{\frac{T}{\mu}}=\sqrt{\frac{T}{\mu}} \\ \because \quad n=\frac{1}{2 L}...
A particle moves along a circle of radius ‘r’ with constant tangential acceleration. If the velocity of the particle is ‘ 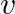 ‘ at the end of second revolution, after the revolution has started then the tangential acceleration is
‘ at the end of second revolution, after the revolution has started then the tangential acceleration is
A) 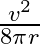
B) 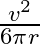
C) 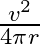
D) 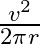
Answer is (A) Using $v^{2}-u^{2}=2 a s$ and $u=u_{1}$ and $s=4 \pi r$ $\therefore \quad 2 \mathrm{as}=\mathrm{v}^{2} \Rightarrow \frac{\mathrm{v}^{2}}{2...
A mass ‘  ‘ connected to a horizontal spring performs S.H.M. with amplitude ‘
‘ connected to a horizontal spring performs S.H.M. with amplitude ‘ ‘. While mass ‘
‘. While mass ‘ 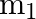 ‘ is passing through mean position another mass ‘
‘ is passing through mean position another mass ‘ 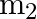 ‘ is placed on it so that both the masses move together with amplitude
‘ is placed on it so that both the masses move together with amplitude 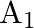 , The ratio of
, The ratio of 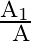 is
is 
A) ![Rendered by QuickLaTeX.com \left[\frac{m_{1}}{m_{1}+m_{2}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-2b29b57ed39eb1f6c58436a799bc7bae_l3.png)
B) ![Rendered by QuickLaTeX.com \left[\frac{\mathrm{m}_{1}+\mathrm{m}_{2}}{\mathrm{~m}_{1}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-f41789fa421a8e3dd967c1a79e94c1ec_l3.png)
C) ![Rendered by QuickLaTeX.com \left[\frac{m_{2}}{m_{1}+m_{2}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-e261477e0e770b6b424c21c834fa7358_l3.png)
D) ![Rendered by QuickLaTeX.com \left[\frac{\mathrm{m}_{1}+\mathrm{m}_{2}}{\mathrm{~m}_{2}}\right]^{\frac{1}{2}}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-17db4e934120c4864100c1c60fc53b04_l3.png)
Answer is (A) P.E of the oscillating mass is given by $E=\frac{1}{2} m \omega x^{2}=\frac{1}{2} k x^{2} \quad \text { Where } k=m \omega^{2}$ When only black is oscillating, at $x=A$ $E=E_{\max...
Assuming the expression for the pressure exerted by the gas on the walls of the container, it can be shown that pressure is
A) ![Rendered by QuickLaTeX.com [\frac{1}{3}]^{rd}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-9f96177eb515481513c54e551e5e8124_l3.png) kinetic energy per unit volume of a gas
kinetic energy per unit volume of a gas
B) ![Rendered by QuickLaTeX.com [\frac{2}{3}]^{rd}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-496e049a5aa2709ddb63a237a5e8e0be_l3.png) kinetic energy per unit volume of a gas
kinetic energy per unit volume of a gas
C) ![Rendered by QuickLaTeX.com [\frac{3}{4}]^{th}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-152c9f0a76e8d19379623b976244d69f_l3.png) kinetic energy per unit volume of a gas
kinetic energy per unit volume of a gas
D) 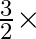 kinetic energy per unit volume of a gas
kinetic energy per unit volume of a gas
Answeris (B) The pressure exerted by the gas on the walls of container is $\mathrm{P}=\mathrm{P}_{0}+\mathrm{P}_{1}+\mathrm{P}_{2}$ i.e. $\quad \mathrm{P}=\mathrm{P}_{0}+\frac{1}{3} 8...
Wire having tension 225 N produces six beats per second when it is tuned with a fork. When tension changes to 256 N, it is tuned with the same fork, the number of beats remain unchanged. The frequency of the fork will be
A) 186 Hz
B) 225 Hz
C) 256 Hz
D) 280 Hz
Answer is (A) For wire vibrating under tension, the fundamental frequency is given by $\mathrm{f}_{1}=\frac{1}{2} \sqrt{\frac{\mathrm{T}}{\mu}}$, Where $\mathrm{T}$ is tension \& $\mathrm{r}$...
When the observer moves towards the stationary source with velocity, ‘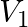 ’, the apparent frequency of emitted note is ‘
’, the apparent frequency of emitted note is ‘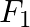 ’. When the observer moves away from the source with velocity ‘
’. When the observer moves away from the source with velocity ‘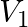 ’, the apparent frequency is ‘
’, the apparent frequency is ‘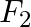 ’. If ‘
’. If ‘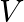 ’ is the velocity of sound in air and
’ is the velocity of sound in air and 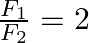 then
then 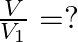
A) 2
B) 3
C) 4
D) 5
Answer is (B) $\frac{\mathrm{f}_{1}}{\mathrm{f}_{2}}=2, \quad$ Speed of approach $=$ Speed of leaving The apparent frequency of sound level by observer when it is approaching source is given by...
A ring and a disc roll on the horizontal surface without slipping with same linear velocity. If both have same mass and total kinetic energy of the ring is 4 J then total kinetic energy of the disc is
A) 3 J
B) 4 J
C) 5 J
D) 6 J
Answer is (A) Total K.E of the rolling disc or ring is given by. $\mathrm{K} . \mathrm{E}=\frac{1}{2} \mathrm{mv}^{2}+\frac{1}{2} \mathrm{I} \omega^{2}$ For ring and disk, translational kinetic...
Two wires having same length and material are stretched by same force. Their diameters are in the ratio 1 : 3. The ratio of strain energy per unit volume for these two wires (smaller to larger diameter) when stretched is
A) 3 : 1
B) 9 : 1
C) 27 : 1
D) 81 : 1
Answer is (B) Strain energy per unit volume $\mathrm{u}=\frac{\mathrm{U}}{\mathrm{v}}=\frac{1}{2} \times$ Stress $\times$ Strain $\begin{array}{l} \frac{U}{V}=\frac{1}{2} \times...
In vertical circular motion, the ratio of kinetic energy of a particle at highest point to that at lowest point is
A) 5
B) 2
C) 0.5
D) 0.2
Answer is (D) $\mathrm{K} . \mathrm{E}=\frac{1}{2} \mathrm{mv}^{2}$ At lowest point in vertical circular motion. $V_{L}=\sqrt{5 r g}$ and at highest point $V_{h}=\sqrt{r g}$ $\therefore \quad...
For a gas 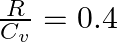 where ‘R’ is the universal gas constant and ‘
where ‘R’ is the universal gas constant and ‘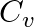 ’ is molar specific heat at constant volume. The gas is made up of molecules which are
’ is molar specific heat at constant volume. The gas is made up of molecules which are
A) rigid diatomic
B) monoatomic
C) non-rigid diatomic
D) polyatomic
1:Answer is (A) $\begin{array}{l} \frac{R}{C_{v}}=0.4 \Rightarrow \quad \frac{C_{p}-C_{v}}{C_{v}}=0.4 \\ \frac{C_{p}}{C_{v}}-1=0.4 \end{array}$ $\therefore \quad \frac{C_{p}}{C_{v}}=1.4 \Rightarrow...
The hydrolysis reaction that takes place at the slowest rate, among the following is
(1) (2) (3) H3C-CH2-Cl (aq. NaOH) H3C-CH2-OH (4) H3C=CH-CH2-Cl (aq. NaOH) H3C=CH-CH2-OH Correct Answer: (1) Explanation: The aryl halides do not give substitution reactions due to partial...
When vapors of a secondary alcohol is passed over heated copper at 573 K, the product formed is
(1) an alkene (2) a carboxylic acid (3) an aldehyde (4) a ketone Correct Answer: (4) a ketone Explanation:
The major products C and D formed in the following reaction respectively are
${{H}_{3}}C-C{{H}_{2}}-C{{H}_{2}}-O-C{{(C{{H}_{3}})}_{3}}\xrightarrow[\Delta ]{excessHI}C+D$ (1) H3C – CH2 – CH2 – OH and HO – C(CH3)3 (2) H3C – CH2 – CH2 – I and I – C(CH3)3 (3) H3C – CH2 – CH2–...
The pH of 0.01 M NaOH (aq) solution will be
(1) 9 (2) 7.01 (3) 2 (4) 12 Correct Answer: (4) 12 Explanation: [OH-] = [NaOH] = 0.01 M = 10-2 M pOH = –log[OH-] = –log(10-2) pH = 14 – pOH = 12
Which of the following cannot act both as Bronsted acid and as Bronsted base?
(1) HSO4- (2) HCO3- (3) NH3 (4) HCl Correct Answer: (4) HCl Explanation: The substance which can release and accept H+ can act as both Bronsted base and acid. HCl cannot accept H+...
The molar solubility of CaF2(Ksp = 5.3 × 10–11) in 0.1 M solution of NaF will be
(1) 5.3 × 10- 10 mol L-1 (2) 5.3 × 10-11 mol L-1 (3) 5.3 × 10-8 mol L-1 (4) 5.3 × 10-9 mol L-1 Correct Answer: (4) 5.3 × 10-9 mol L-1 Explanation: Calculation – Ksp(CaF2) = 5.3 × 10-11...
The oxidation state of Cr in CrO6 is
(1) +4 (2) –6 (3) +12 (4) +6 Correct Answer: (4) +6 Explanation: The most appropriate oxidation state of Cr in CrO6 is +6 although CrO6 has doubtful existence.
The number of hydrogen bonded water molecule(s) associated with CuSO4 . 5H2O is
(1) 5 (2) 3 (3) 1 (4) 2 Correct Answer: (3) 1 Explanation: The actual structure of CuSO4.5H2O is [Cu(H2O)4]SO4.H2O, so only one water molecule is associated with the molecule via hydrogen...
Formula of nickel oxide with metal deficiency defect in its crystal is Ni0.98O. The crystal contains Ni2+ and Ni3+ ions. The fraction of nickel existing as Ni2+ ions in the crystal is
(1) 0.31 (2) 0.96 (3) 0.04 (4) 0.50 Correct Answer: (2) 0.96 Explanation: Ni0.98 O (Ni2+ )x( Ni3+ )0.98-x( O2- )1 Net charge = 0 [X x 2]+ [ (0.98- x) 3]+ [-2x 1]= 0 x = 0.94 Fraction of...
Which of the following statements is correct regarding a solution of two components A and B exhibiting positive deviation from ideal behavior?
(1) Intermolecular attractive forces between A-A and B-B are equal to those between A-B. (2) Intermolecular attractive forces between A-A and B-B are stronger than those between A-B. (3) mixH = 0 at...
In water saturated air, the mole fraction of water vapour is 0.02. If the total pressure of the saturated air is 1.2 atm, the partial pressure of dry air is
(1) 0.98 atm (2) 1.18 atm (3) 1.76 atm (4) 1.176 atm Correct Answer: (4) 1.176 atm Explanation: Calculation - Mole fraction of water vapour = 0.02 Mole fraction of dry air = 1 – 0.02 = 0.98...
The standard electrode potential (E⊖) values of Al3+/Al, Ag+/Ag, K+/K and Cr3+/Cr are –1.66 V, 0.80 V, –2.93 V and –0.74 V, respectively. The correct decreasing order of reducing power of the metal is
(1) Al > K > Ag > Cr (2) Ag > Cr > Al > K (3) K > Al > Cr > Ag (4) K > Al > Ag > Cr Correct Answer: (3) K > Al > Cr > Ag Explanation: The lesser...
The liquified gas that is used in dry cleaning along with a suitable detergent is
(1) CO2 (2) Water gas (3) Petroleum gas (4) NO2 Correct Answer: (1) CO2 Explanation: Liquified CO2 (carbon dioxide) with a suitable detergent is used in dry cleaning.
The Crystal Field Stabilisation Energy (CFSE) for [CoCl6]4– is 18000 cm–1. The CFSE for [CoCl4]2– will be,
(1) 8000 cm-1 (2) 6000 cm-1 (3) 16000 cm-1 (4) 18000 cm-1 Correct Answer: (1) 8000 cm-1 Explanation: Formula Used - t = 4/9 o
When neutral or faintly alkaline KMnO4 is treated with potassium iodide, iodide ion is converted into ‘X’. ‘X’ is
(1) IO- (2) I2 (3) IO4- (4) IO3- Correct Answer: (4) IO3- Explanation: In neutral or faintly alkaline solutions, iodide ion is converted into iodate. Reaction Involved – 2MnO4- + H2O + I-...
Identify the correct formula of ‘oleum’ from the following.
(1) H2S2O8 (2) H2S2O7 (3) H2SO3 (4) H2SO4 Correct Answer: (2) H2S2O7 Explanation: The chemical formula of oleum is H2S2O7
Which of the following oxoacids of phosphorus has strongest reducing property?
(1) H3PO4 (2) H4P2O7 (3) H3PO3 (4) H3PO Correct Answer: (2) Ca3P2 Explanation: Hypo phosphorous acid is a good reducing agent as it contains two P–H bonds.
A compound ‘X’ upon reaction with H2O produces a colorless gas ‘Y’ with rotton fish smell. Gas ‘Y’ is absorbed in a solution of CuSO4 to give Cu3P2 as one of the products. Predict the compound ‘X’.
(1) Ca23(PO4)2 (2) Ca3P2 (3) NH4Cl (4) As2O Correct Answer: (2) Ca3P2 Explanation: Reactions Involved – Ca3P2 + 6H2O 3Ca(OH)2 + 2PH3 2PH3 3CuSO4 Cu3P2 +...
Identify the incorrect statement.
(1) Gangue is an ore contaminated with undesired materials (2) The scientific and technological process used for isolation of the metal from its ore is known as metallurgy (3) Minerals are naturally...
Which of the following compounds is used in cosmetic surgery?
(1) Zeolites (2) Silica (3) Silicates (4) Silicones Correct Answer: (4) Silicones Explanation: Silicones are usually used in cosmetic surgeries.
Aluminium chloride in acidified aqueous solution forms a complex ‘A’, in which hybridisation state of Al is ‘B’. What are ‘A’ and ‘B’, respectively?
(1) [Al(H2O)6]3+ , d2sp3 (2) [Al(H2O)6]3+ , sp3 d2 (3) [Al(H2O)6]3+ , sp3 (4) [Al(H2O)6]3+ , dsp2 Correct Answer: (2) [Al(H2O)6]3+ , sp3 d2 Explanation: Reaction Involved – AlCl3(acidified...
In the following reaction, H3C-C=CH (red hot iron tube at 873 K) →A, the number of sigma () bonds present in the product A, is
(1) 18 (2) 21 (3) 9 (4) 24 Correct Answer: (2) 21 Explanation:
The alkane that gives only one mono-chloro product on chlorination with Cl2 in presence of diffused sunlight is
(1) Isopentane (2) 2, 2-dimethylbutane (3) neopentane (4) n-pentane Correct Answer: (3) neopentane Explanation: The chlorination of neopentane with Cl2 in the presence of diffused sunlight...
The most stable carbocation, among the following, is
Correct Answer: Option (4) Explanation: The stability of carbocation depends upon the number of a-H because of +H effect that is the hyperconjugation effect.
Match the catalyst with the process
Catalyst Process (i) V2O5 (a) the oxidation of ethyne to ethanal (ii) TiCl4+ Al(CH3)3 (b)...
Match the oxide given in column A with its property given in column B
Column-A Column-B (i) Na2O (a) Neutral (ii) Al2O3 (b) Basic (iii) N2O (c) Acidic (iv) Cl2O7 (d)...
Reversible expansion of an ideal gas under isothermal and adiabatic conditions are as shown in the figure.
AB Isothermal expansion AC Adiabatic expansion. Which of the following options is not correct? (1) TC > TA (2) Sisothermal > Sadiabatic (3) TA = TB (4) wisothermal >...
An ideal gas expands isothermally from 10–3 m3 to 10–2 m3 at 300 K against a constant pressure of 105 Nm–2. The work done on the gas is
(1) – 900 kJ (2) + 270 kJ (3) – 900 J (4) + 900 kJ Correct Answer: (3) – 900 J Explanation: Using the formula, W = -Pext (Vf – Vi) W = -900 J
The volume occupied by 1.8 g of water vapour at 374°C and 1 bar pressure will be [Use R = 0.083 bar LK–1 mol–1]
(1) 5.37 L (2) 96.66 L (3) 55.87 L (4) 3.10 L Correct Answer: (1) 5.37 L Explanation: Using the formula, PV = nRT V = 5.37 L
In hydrogen atom, the de Broglie wavelength of an electron in the second Bohr orbit is [Given that Bohr radius, ao = 52.9 pm]
(1) 105.8 pm (2) 211.6 pm (3) 211.6 pm (4) 52.9 pm Correct Answer: (3) 211.6 pm Explanation: n = 2 r = 211.6 2 pm
Orbital having 3 angular nodes and 3 total nodes is
(1) 6 d (2) 5 p (3) 3 d (4) 4 f Correct Answer: (4) 4 f Explanation: Total number of nodes = (n – 1) 3 = n – 1 n = 4
The density of 2 M aqueous solution of NaOH is 1.28 g/cm3. The molality of the solution is [Given that molecular mass of NaOH = 40 g mol-1]
(1) 1.32 m (2) 1.20 m (3) 1.56 m (4) 1.67 m Correct Answer: (4) 1.67 m Explanation: Let, volume of solution = 1 litre Mole of NaOH = 2 Mass of NaOH solution = 1000 × 1.28 = 1280 g Mass of...
The artificial sweetener stable at cooking temperature and does not provide calories is
(1) alitame (2) Saccharin (3) Aspartame (4) Sucralose Correct Answer: (4) Sucralose Explanation: Sucralose is trichloro derivative of sucrose. It is stable at cooking temperature. It does...
The polymer that is used as a substitute for wool in making commercial fibres is,
(1) Buna-N (2) Melamine (3) nylon-6,6 (4) Polyacrylonitrile Correct Answer: (4) Polyacrylonitrile Explanation: Polyacrylonitrile is used as a substitute for wool in making commercial fibre as...
Which structure of protein remain intact during denaturation process?
(1) Tertiary structure only (2) Both secondary and tertiary structures (3) Primary structure only (4) Secondary structure only Correct Answer: (3) Primary structure only Explanation: During...
The amine that reacts with Hinsberg’s reagent to give an alkali insoluble product is
Correct Answer: Option (2) Explanation: Secondary amines react with Hinsberg's reagent to give a product which is insoluble in...
The reaction that does not give benzoic acid as the major product is,
Correct Answer: Option (3) Explanation:
Which of the alkali metal chloride (MCl) forms its dihydrate salt (MCl . 2 H2O) easily?
(1) KCl (2) LiCl (3) CsCl (4) RbCl Correct Answer: (2) LiCl Explanation: Out of alkali metal chlorides only LiCl forms a dihydrate, other metal chlorides do not form hydrates.
Crude sodium chloride obtained by crystallization of brine solution does not contain
(1) CaSO4 (2) MgSO4 (3) Na2SO4 (4) MgCl2 Correct Answer: (2) MgSO4 Explanation: Crude sodium chloride generally obtained by crystallization of brine solution contains Na2SO4, CaSO4, CaCl2...
Which of the following is the correct order of dipole moment?
(1) H2O < NF3 < NH3 < BF3 (2) NH3 < BF3 < NF3 < H2O (3) BF3 < NF3 < NH3 < H2O (4) BF3 < NH3 < NF3 < H2O Correct Answer: (3) BF3 < NF3 < NH3 <...
Which of the following is paramagnetic?
(1) O2 (2) N2 (3) H2 (4) Li2 Correct Answer: (1) O2 Explanation: O2 has 8 + 8 = 16 electrons. In O2 molecule, there are two (2) unpaired electrons, so, it is a "paramagnetic" substance in...
The correct option representing a Freundlich adsorption isotherm is
(1) x/m = kp-1 (2) x/m = kp0.3 (3) x/m = kp2.5 (4) x/m = kp-0.5 Correct Answer: (2) x/m = kp0.3 Explanation: According to Freundlich isotherm, x/m = kp0.3
For a reaction, activation energy Ea = 0 and the rate constant at 200 K is 1.6 × 106 s-1. The rate constant at 400 K will be [Given that gas constant, R = 8.314 J K-1 mol-1]
(1) 3.2 × 106 s-1 (2) 3.2 × 104 s-1 (3) 1.6 × 106 s-1 (4) 1.6 × 103 s-1 Correct Answer: (3) 1.6 × 106 s-1 Explanation: From Arrhenius equation, k400 = k200
A first order reaction has a rate constant of 2.303 × 10-3 s-1. The time required for 40 g of this reactant to reduce to 10 g will be [Given that log102 = 0.3010]
(1) 602 s (2) 230.3 s (3) 301 s (4) 2000 s Correct Answer: (1) 602 s Explanation: For a first order reaction 40 g substance requires 2 half life periods to reduce upto 10 g Time taken in...
Following limiting molar conductivities are given as
$\lambda $mo (H2SO4) =x S cm2 mol-1 $\lambda $mo (K2SO4 )= y S cm2 mol-1 $\lambda $mo (CH3COOK) =z S cm mol-1 $\lambda $mo (in S cm2 mol-1 ) for CH3COOH will be (1)(x-y)/2+z (2) x – y + 2z (3) x +...
Which one is a wrong statement?
(1) The electronic configuration of N atom is, (2) An orbital is designated by three quantum numbers while an electron in an atom is designated by four quantum numbers (3) Total orbital...
Iron exhibits bcc structure at room temperature. Above 900°C, it transforms to fcc structure. The ratio of density of iron at room temperature to that at 900°C (assuming molar mass and atomic radii of iron remains constant with temperature) is,
$\begin{array}{l} (1)\frac{{3\sqrt 3 }}{{4\sqrt 2 }}\\ (2)\frac{{4\sqrt 3 }}{{3\sqrt 2 }}\\ (3)\frac{{\sqrt 3 }}{{\sqrt 2 }}\\ (4)\frac{1}{2} \end{array}$ Correct Answer: (1)\frac{{3\sqrt 3...
Magnesium reacts with an element (X) to form an ionic compound. If the ground state electronic configuration of (X) is 1s2 2s2 2p3, the simplest formula for this compound is
(1) Mg2X (2) MgX2 (3) Mg2X3 (4) Mg3X2 Correct Answer: (4) Mg3X2 Explanation: Formula of compound formed by Mg and X will be Mg3X2.
Consider the following species: Which one of these will have the highest bond order?
(1) CN+ (2) CN- (3) NO (4) CN Correct Answer: (2) CN- Explanation: CN- = (s1s)2, (s* 1s)2, (s2s)2,(s* 2s)2, (p2px)2 = (p2py) 2,(s2pz) 2 Bond Order = 10-4 / 3 => 3
The correction factor ‘a’ to the ideal gas equation corresponds to
(1) Electric field present between the gas molecules (2) Volume of the gas molecules (3) Density of the gas molecules (4) Forces of attraction between the gas molecules Correct Answer: (4)...
The bond dissociation energies of X2, Y2 and XY are in the ratio of 1 : 0.5 : 1. H for the formation of XY is –200 kJ mol–1. The bond dissociation energy of X2 will be
(1) 800 kJ mol-1 (2) 100 kJ mol-1 (3) 200 kJ mol-1 (4) 400 kJ mol-1 Correct Answer: (1) 800 kJ mol-1 Explanation:
