Solution: During the formation of a molecule, the extent of bonding that occurs between two atoms is represented by the bond strength of the molecule. As the bond strength increases, the bond...
Although geometries of 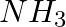 and
and 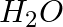 molecules are distorted tetrahedral, bond angle in water is less than that of Ammonia. Discuss.
molecules are distorted tetrahedral, bond angle in water is less than that of Ammonia. Discuss.
Solution: Ammonia's central atom (N) has one lone pair and three bond pairs. In water, the central atom (O) has two lone pairs and two bond pairs. As a result, the two bond pairs repel the two lone...
Discuss the shape of the following molecules using the VSEPR model: 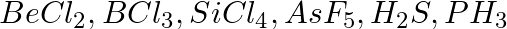
Solution: $BeCl_2$ The central atom does not have a lone pair, but it does have two bond pairs. As a result, its shape is AB2, or linear. $BCl_3$ The central atom has three bond pairs but no lone...
Write the favourable factors for the formation of an ionic bond.
Solution: Ionic bonds are formed when one or more electrons are transferred from one atom to another. As a result, the ability of neutral atoms to lose or gain electrons is required for the...
Equipotential surfaces a) are closer in regions of large electric fields compared to regions of lower electric fields b) will be more crowded near sharp edges of a conductor c) will be more crowded near regions of large charge densities d) will always be equally spaced
The correct answer is a) are closer in regions of large electric fields compared to regions of lower electric fields b) will be more crowded near sharp edges of a conductor c) will be more crowded...
Define the octet rule. Write its significance and limitations
Solution: “Atoms can combine either by transferring valence electrons from one atom to another or by sharing their valence electrons in order to achieve the closest inert gas configuration by having...
Draw the Lewis structures for the following molecules and ions : 
Solution: The lewis dot structures are:
Write Lewis symbols for the following atoms and ions: Sand 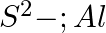 and
and 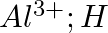 and
and 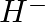
Solution: For S and S2- A sulphur atom has only 6 valence electrons, which is a very small number. As a result, the Lewis dot symbol for the letter S is The presence of a...
Consider a uniform electric field in the z direction. The potential is a constant a) in all space b) for any x for a given z c) for any y for a given z d) on the x-y plane for a given z
The correct answer is b) for any x for a given z c) for any y for a given z d) on the x-y plane for a given z
The measurement of an unknown resistance R is to be carried out using Wheatstone bridge. Two students perform an experiment in two ways. The first student take R2 = 10Ω and R1 = 5Ω. The other student takes R2 = 1000 Ω and R1 = 500 Ω. In the standard arm, both take R3 = 5 Ω. Both find R = R2/R1 R3 = 10 Ω within errors.
a) the errors of measurement of the two students are the same
b) errors of measurement do depend on the accuracy with which R2 and R1 can be measured
c) if the student uses large values of R2 and R1, the currents through the arms will be feeble. This will make determination of null point accurately more difficult
d) Wheatstone bridge is a very accurate instrument and has no errors of measurement
The correct answer is b) errors of measurement do depend on the accuracy with which R2 and R1 can be measured c) if the student uses large values of R2 and R1, the currents through the arms will be...
Write Lewis dot symbols for atoms of the following elements :e) N f) Br
Solution: Nitrogen atoms have only five valence electrons in total. As a result, the Lewis dot symbol for N is Bromine, because the atom has only seven valence electrons. As a result,...
A parallel plate capacitor is made of two dielectric blocks in series. One of the blocks has thickness d1 and dielectric constant k1 and the other has thickness d2 and dielectric constant k2 as shown in the figure. This arrangement can be thought of as a dielectric slab of thickness d = d1 + d2 and effective dielectric constant k. The k is a) k1d1 + k2d2/d1+d2 b) k1d1 + k2d2/k1 + k2 c) k1k2 (d1 + d2)/(k1d1 + k2d2) d) 2k1k2/k1 + k2
The correct answer is c) k1k2 (d1 + d2)/(k1d1 + k2d2)
Write Lewis dot symbols for atoms of the following elements :c) B d) O
Solution: Boron atoms have only three valence electrons, which is a very small number. As a result, the Lewis dot symbols for B are as follows: The oxygen atom has only six valence...
Equipotential at a great distance from a collection of charges whose total sum is not zero are approximately a) spheres b) planes c) paraboloids d) ellipsoids
The correct answer is a) spheres
Temperature dependence of resistivity ρ(T) of semiconductors, insulators, and metals is significantly based on the following factors:
a) number of charge carriers can change with temperature T
b) time interval between two successive collisions can depend on T
c) length of material can be a function of T
d) mass of carriers is a function of T
solution:The correct answer is a) number of charge carriers can change with temperature T b) time interval between two successive collisions can depend on T
The electrostatic potential on the surface of a charged conducting sphere is 100V. Two statements are made in this regard: S1: At any point inside the sphere, the electric intensity is zero S2: At any point inside the sphere, the electrostatic potential is 100V Which of the following is a correct statement? a) S1 is true but S2 is false b) Both S1 and S2 are false c) S1 is true, S2 is also true, and S1 is the cause of S2 d) S1 is true, S2 is also true but the statements are independent
The correct answer is c) S1 is true, S2 is also true, and S1 is the cause of S2
Consider a simple circuit in the figure.stands for a variable resistance R’.
R’ can vary from R0 to infinity. r is internal resistance of the battery,
a) potential drop across AB is nearly constant as R’ is varied
b) current through R’ is nearly a constant as R’ is varied
c) current I depends sensitively on R’
d) I ≥V/r+R always
solution: The correct answer is a) potential drop across AB is nearly constant as R’ is varied d) I ≥V/r+R always
Figure shows some equipotential lines distributed in space. A charged object is moved from point A to point B. a) the work done in fig (i) is the greatest b) the work done in fig (ii) is least c) the work done is the same in fig (i), fig (ii), and fig (iii) d) the work done in fig (iii) is greater than fig (ii) but equal to that in fig (i)
The correct answer is c) the work done is the same in fig (i), fig (ii), and fig (iii)
A capacitor of 4μF is connected as shown in the circuit. The internal resistance of the battery is 0.5Ω. The amount of charge on the capacitor plates will be a) 0 b) 4μC c) 16μC d) 8μC
The correct answer is d) 8μC
Kirchhoff’s junction rule is a reflection of
a) conservation of current density vector
b) conservation of charge
c) the fact that the momentum with which a charged particle approaches a junction is unchanged as the charged particle leaves the junction
d) the fact that there is no accumulation of charges at a junction
solution: The correct answer is b) conservation of charge d) the fact that there is no accumulation of charges at a junction
Which of the following characteristics of electrons determines the current in a conductor?
v a) drift velocity alone
b) thermal velocity alone
c) both drift velocity and thermal velocity
d) neither drift nor thermal velocity
solution: The correct answer is a) drift velocity alone
$=\frac{4}{7}\left[ {{16}^{\frac{7}{4}}}-1 \right]$ $=\frac{512}{7}$
$\frac{{{2}^{-4}}}{-4}-\frac{1}{-4}$ $\frac{16-1}{64}$ $\frac{15}{64}$
$\frac{2}{3}\left[ {{4}^{\frac{3}{2}}}-1 \right]$ $=\frac{14}{3}$
$=\frac{{{3}^{5}}}{5}-\frac{1}{5}$ $\frac{243-1}{5}$ $=\frac{243}{5}$
A metal rod of length 10 cm and a rectangular cross-section of 1 cm × 1/2 cm is connected to battery across opposite faces. The resistance will be
a) maximum when the battery is connected across 1 cm × 1/2 cm faces
b) maximum when the battery is connected across 10 cm × 1 cm faces
c) maximum when the battery is connected across 10 cm × 1/2 cm faces
d) same irrespective of the three faces
solution:The correct solution is a) maximum when the battery is connected across 1 cm × 1/2 cm faces
Two cells of emf’s approximately 5V and 10V are to be accurately compared using a potentiometer of length 400 cm.
a) the battery that runs the potentiometer should have voltage of 8V
b) the battery of potentiometer can have a voltage of 15V and R adjusted so that the potential drop across the wire slightly exceeds 10V
c) the first portion of 50 cm of wire itself should have a potential drop of 10V
d) potentiometer is usually used for comparing resistances and not voltages
Solution: The correct solution is b) the potentiometer's battery can be set to 15V and R adjusted so that the potential drop across the wire is a little higher than 10V.
A resistance R is to be measured using a meter bridge. Student chooses the standard resistance S to be 100Ω. He finds the null point at l1 = 2.9 cm . He is told to attempt to improve the accuracy. Which of the following is a useful way?
a) he should measure l1 more accurately
b) he should change S to 1000 Ω and repeat the experiment
c) he should change S to 3 Ω and repeat the experiment
d) he should give up hope of a more accurate measurement with a meter bridge
solution:The correct answer is c) he should change S to 3 Ω and repeat the experiment
Two batteries of emf ε1 and ε2 and internal resistances r1 and r2 respectively are connected in parallel as shown in the figure.a) the equivalent emf εeq of the two cells is between ε1 and ε2 that is ε1 < εeq < ε2
b) the equivalent emf εeq is smaller than ε1
c) the εeq is given by εeq = ε1 + ε2 always
d) εeq is independent of internal resistances r1 and r2
solution: The correct answer is a) the equivalent emf εeq of the two cells is between ε1 and ε2 that is ε1 < εeq < ε2
Consider a current-carrying wire in the shape of a circle. Note that as the current progresses along the wire, the direction of j changes in an exact manner, while the current I remain unaffected. The agent that is essentially responsible for is
a) source of emf
b) electric field produced by charges accumulated on the surface of wire
c) the charges just behind a given segment of wire which push them just the right way by repulsion
d) the charges ahead
solution: The correct answer is b) electric field produced by charges accumulated on the surface of wire
Construct a  matrix whose elements are given by
matrix whose elements are given by 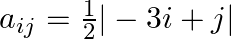
Solution: It is a (3 $x 4)$ matrix. Therefore, it has 3 rows and 4 columns. Given that $a_{i j}=\frac{|-a \|+| l}{2}$ Therefore, $a_{11}=1 . a_{12}=\frac{1}{2}, a_{13}=0, a_{13}=\frac{1}{2}$...
Construct a  matrix whose elements are
matrix whose elements are 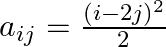
Solution: It is a $(2 \times 3)$ matrix. Therefore, it has 2 rows and 3 columns. Given that $a_{i j}=\frac{\left(t-2 \rho^{2}\right.}{2}$ Therefore, $a_{11}=\frac{1}{2}, a_{12}=\frac{9}{2},...
Construct a  matrix whose elements are
matrix whose elements are 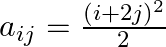
Solution: It is a $(2 \times 2)$ matrix. So, it has 2 rows and 2 columns. Given that $a_{i j}=\frac{(i+2 j)^{2}}{2}$ Therefore, $a_{11}=\frac{9}{2}, a_{12}=\frac{25}{2}$. $a_{21}=8 . a_{22}=18$...
Construct a  matrix whose elements are given by
matrix whose elements are given by 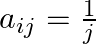
Solution: It is $(4 \times 3)$ matrix. Therefore it has 4 rows and 3 columns Given that$a_{i j}=\frac{i}{j}$ Therefore, $a_{11}=1 . a_{12}=\frac{1}{2}, a_{13}=\frac{1}{3}$ $\begin{array}{l}...
Construct a  matrix whose elements are given by
matrix whose elements are given by 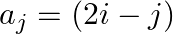
Solution: It is given that: $a_{i j}=(2 \mid-j)$ Now, $a_{11}=(2 \times 1-1)=2-1=1$ $\begin{array}{l} a_{12}=2 \times 1-2=2-2=0 \\ a_{21}=2 \times 2-1=4-1=3 \\ a_{22}=2 \times 2-2=4-2=2 \\ a_{31}=2...
Find all possible orders of matrices having 7 elements.
Solution: No. of entries $=($ No. of rows) $x$ (No. of columns) $=7$ If order is $(\mathrm{a} \times \mathrm{b})$ then, No. of entries = $\mathrm{a} \times \mathrm{b}$ Therefore now a $x b=7$ (in...
If a matrix has 18 elements, what are the possible orders it can have?
Solution: No. of entries $=$ (No. of rows) $x$ (No. of columns) $=18$ If order is $(a \times b)$ then, No. of entries = $a \times b$ Therefore now $a \times b=18$ (in this case) Possible cases are...
Write the order of each of the following matrices:
i. ![Rendered by QuickLaTeX.com E=\left[\begin{array}{c}-2 \\ 3 \\ 0\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-d6b4b616a18471e5e1da412182f3cfc8_l3.png)
ii, ![Rendered by QuickLaTeX.com F=[6]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-f1b7ee74e8d279705a2649b7c1427d2a_l3.png)
Solution: i. $E=\left[\begin{array}{c}-2 \\ 3 \\ 0\end{array}\right]$ Order of matrix $=$ Number of rows $x$ Number of columns $\begin{array}{l} =(3 \times 1) \\ \text { ii, } F=[6] \end{array}$...
Write the order of each of the following matrices:
i. ![Rendered by QuickLaTeX.com \mathrm{C}=\left[\begin{array}{lll}7-\sqrt{2} & 5 & 0\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-2eb7b03611cf5621ee3d31e5a86b8236_l3.png)
ii. ![Rendered by QuickLaTeX.com \mathrm{D}=[8-3]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-40c5e53abc7a937eeaeba1db8b071d85_l3.png)
Solution: i. $C=[7-\sqrt{2} \quad 5 \quad 0]$ Order of matrix $=$ Number of rows $x$ Number of columns $=(1 \times 4)$ ii. $D=[8-3]$ Order of matrix $=$ Number of rows $x$ Number of columns $=(1...
Write the order of each of the following matrices: i. ![Rendered by QuickLaTeX.com A=\left[\begin{array}{cccc}3 & 5 & 4 & -2 \\ 0 & \sqrt{3} & -1 & \frac{4}{9}\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-f969e23c5c5287707729c241fef79e75_l3.png) ii.
ii. ![Rendered by QuickLaTeX.com B=\left[\begin{array}{cc}6 & -5 \\ \frac{1}{2} & \frac{3}{4} \\ -2 & -1\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-ec1233b19429c118c718399789e144a6_l3.png)
Solution: i. $A=\left[\begin{array}{cccc}3 & 5 & 4 & -2 \\ 0 & \sqrt{3} & -1 & \frac{4}{9}\end{array}\right]$ Order of matrix $=$ Number of rows $x$ Number of columns $=(2...
If ![Rendered by QuickLaTeX.com A=\left[\begin{array}{cccc}5 & -2 & 6 & 1 \\ 7 & 0 & 8 & -3 \\ \sqrt{2} & \frac{3}{5} & 4 & 3\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-1db7451bba1582e7fdf8b71d478bfc78_l3.png) then write
then write
i. the elements 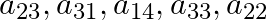 of
of  .
.
Solution: (i) $a_{i j}=$ element of $t^{\text {th }}$ row and $j^{\text {th }}$ column $\begin{array}{l} a_{23}=8 \\ a_{31}=\sqrt{2} \\ a_{14}=1 \\ a_{33}=4 \\ a_{22}=0 \end{array}$
If ![Rendered by QuickLaTeX.com A=\left[\begin{array}{cccc}5 & -2 & 6 & 1 \\ 7 & 0 & 8 & -3 \\ \sqrt{2} & \frac{3}{5} & 4 & 3\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-1db7451bba1582e7fdf8b71d478bfc78_l3.png) then write
then write
i. the order of the matrix  ,
,
ii. the number of all entries in  ,
,
Solution: (i) Order of matrix $=$ Number of rows $x$ Number of columns $=(3 \times 4)$ (ii) Number of entries = (Number of rows) $x$ (Number of columns) $=3 \times 4$ $=12$
If ![Rendered by QuickLaTeX.com A=\left[\begin{array}{cccc}5 & -2 & 6 & 1 \\ 7 & 0 & 8 & -3 \\ \sqrt{2} & \frac{3}{5} & 4 & 3\end{array}\right]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-1db7451bba1582e7fdf8b71d478bfc78_l3.png) then write
then write
i. the number of rows in  ,
,
ii. the number of columns in  ,
,
Solution: (i) Number of rows $=3$ (ii) Number of columns = 4
Let 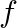 and
and 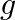 be two functions from
be two functions from 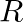 into
into 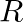 , defined by
, defined by 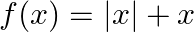 and
and 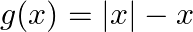 for all
for all 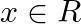 . Find f o
. Find f o 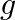 and g o
and g o 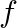 .
.
Solution: $\begin{array}{ll} \mathrm{f}(\mathrm{x})=|\mathrm{x}|+\mathrm{x} \quad & \text { (given }) \\ \mathrm{g}(\mathrm{x})=|\mathrm{x}|-\mathrm{x} \quad & \text { (given) } \end{array}$...
Let 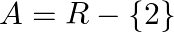 and
and 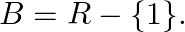 If
If 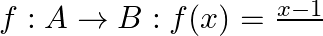 , show that
, show that 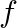 is one-one and onto. Hence, find
is one-one and onto. Hence, find 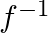
Solution: $\mathrm{f}(\mathrm{x})=\frac{x-1}{x-2} \quad \text { (as given) }$ One-One function Suppose $\mathrm{p}, \mathrm{q}$ be two arbitrary elements in $\mathrm{R}$ Therefore, $f(p)-f(q)$...
Let 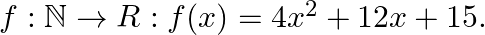 Show that f:
Show that f: 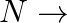 range
range 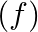 is invertible. Find
is invertible. Find 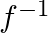
Solution: $\mathrm{f}(\mathrm{x})=4 \mathrm{x} 2+12 \mathrm{x}+15 \quad \text { (as given) }$ $\mathrm{f}(\mathrm{x})$ is invertible if $\mathrm{f}(\mathrm{x})$ is a bijection (i.e one-one onto...
Let 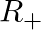 be the set of all positive real numbers. show that the function
be the set of all positive real numbers. show that the function ![Rendered by QuickLaTeX.com f: R_{4} \rightarrow[-5, \infty]: f(x)=\left(9 x^{2}+6 x-5\right)](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-b71d72438933be58e65dbddebe7345c9_l3.png) is invertible. Find
is invertible. Find 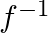 .
.
Solution: $f(x)=9 x_{2}+6 x-5 \text { (as given) }$ $\mathrm{f}(\mathrm{x})$ is invertible if $\mathrm{f}(\mathrm{x})$ is a bijection (i.e one-one onto function) One-One function Suppose p,q be two...
Show that the function 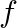 on
on  into itself, defined by
into itself, defined by 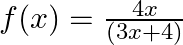 is one-one and onto. Hence, find
is one-one and onto. Hence, find 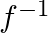
Solution: $\mathrm{f}(\mathrm{x})=\frac{4 x}{3 x+4} \quad$ (as given) One-One function Suppose $\mathrm{p}, \mathrm{q}$ be two arbitrary elements in $\mathrm{R}$ Therefore, $f(p)=f(q)$...
Show that the function 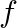 on
on 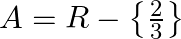 , defined as
, defined as 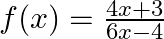 is one-one and onto. Hence, find
is one-one and onto. Hence, find 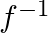 ,
,
Solution: $\mathrm{f}(\mathrm{x})=\frac{4 x+3}{6 x-4} \quad \text { (given) }$ $\mathrm{f}(\mathrm{x})$ is invertible if $\mathrm{f}(\mathrm{x})$ is a bijection (i.e one-one onto function) One-One...
If 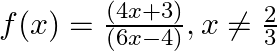 , show that (f
, show that (f 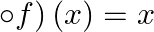 for all
for all 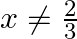 .
.
Solution: $\mathrm{f}(\mathrm{x})=\frac{4 x+3}{6 x-4}, \mathrm{x} \neq \frac{2}{3} \text { (given) }$ We need to Show: fof $(x)=x$ for all $x \neq \frac{2}{3}$ It is known that fof $(x)=f(f(x))$...
Let 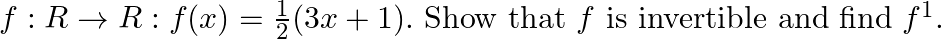
Solution: $\mathrm{f}(\mathrm{x})=\frac{1}{2}(3 \mathrm{x}+1) \quad$ (given) $\mathrm{f}(\mathrm{x})$ is invertible if $\mathrm{f}(\mathrm{x})$ is a bijection (i.e one-one onto function) One-One...
Let f : Q → Q : f(x) = 3x —4. Show that f is invertible and find 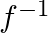 .
.
Solution: $\mathrm{f}(\mathrm{x})=3 \mathrm{x}-4 \quad$ (as given) $\mathrm{f}(\mathrm{x})$ is invertible if $\mathrm{f}(\mathrm{x})$ is a bijection (i.e one-one onto function) One-One function...
Show that the function f : R → R : f(x) = 2x + 3 is invertible and find 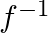 .
.
Solution: $\mathrm{f}(\mathrm{x})=2 \mathrm{x}+3$ (as given) $\mathrm{f}(\mathrm{x})$ is invertible if $\mathrm{f}(\mathrm{x})$ is a bijection (i.e one-one onto function) One-One function Suppose...
Let A = {2, 3, 4, 5} and B = {7, 9, 11, 13}, and let f = {(2, 7), (3, 9), (4, 11), (5, 13)}. Show that f is invertible and find 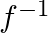 .
.
Solution: A function is invertible if it is a bijection. (i.e. One-One Onto function) One-One function $\mathrm{f}=\{(2,7),(3,9),(4,11),(5,13)\}$ It is observed that different elements of A have...
Write a test to differentiate between pentan-2-one and pentan-3-one.
Solution: One can differentiate between pentan-2-one and pentan-3-one by iodoform test. Pentan-2-one have a –CO-CH3 group and therefore forms a yellow precipitate of Iodoform. Pentan-2-one gives a...
Why is there a large difference in the boiling points of butanal and butane-1-ol?
Solution: Butanal has no intermolecular hydrogen bonding but butan-1-ol has intermolecular hydrogen bonding. This bonding in butan-1-ol makes it more stable at a higher temperature than butanal.
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A) :
Solution: Option (A) and (B) are the answers. Reason:
Benzophenone can be obtained by ____________. (i) Benzoyl chloride + Benzene + AlCl3 (ii) Benzoyl chloride + Diphenyl cadmium (iii) Benzoyl chloride + Phenyl magnesium chloride (iv) Benzene + Carbon monoxide + ZnCl2
Solution: Option (i) and (ii) are the answers Reason: Benzophenone can be obtained by the Friedel-Craft acylation reaction. The reaction is shown as
Through which of the number of the following reactions of carbon atoms can be increased in the chain? (i) Grignard reaction (ii) Cannizaro’s reaction (iii) Aldol condensation (iv) HVZ reaction
Solution: Option (i) and (iii) are the answers. Reason: Grigned reaction and aldol condensation is used to increase the number of carbon attom in the chain as follows:
Write Lewis dot symbols for atoms of the following elements :
a) Mg
b) Na
Solution: Only two valence electrons exist in the magnesium atom. As a result, the Lewis dot symbols for Mg are as follows: Only one valence electron exists in the sodium atom. As a...
Which of the following conversions can be carried out by Clemmensen Reduction? (i) Benzaldehyde into benzyl alcohol (ii) Cyclohexanone into cyclohexane (iii) Benzoyl chloride into benzaldehyde (iv) Benzophenone into diphenylmethane
Solution: Option (ii) and (iv) are the answers. Reason: The carbonyl group of aldehydes and ketones is reduced to CH2 group on treatment with zinc amalgam and concentrated hydrochloric acid...
Explain the formation of a chemical bond.
Answer: "A chemical bond is an attractive force that holds a chemical species' constituents together." For chemical bond formation, many theories have been proposed, including valence shell electron...
Treatment of compound with NaOH solution yields(i) Phenol (ii) Sodium phenoxide (iii) Sodium benzoate (iv) Benzophenone
Solution: Option (ii) and (iii) are the answers. Reason: Treatment of compound with NaOH yields sodium phenoxide and sodium by means of nucleophilic substitution reaction as follows
13. Which of the following compounds do not undergo aldol condensation?
Solution: Option (ii) and (iv) are the answers. reason: Aldehydes and ketones and having at least one alpha-hydrogen undergo a reaction in the presence of dilute alkali as catalyst to beta-hydroxy...
In Clemmensen Reduction carbonyl compound is treated with _____________. (i) Zinc amalgam + HCl (ii) Sodium amalgam + HCl (iii) Zinc amalgam + nitric acid (iv) Sodium amalgam + HNO3
Solution: Option (i) is the answer. Reason: From the above reaction carbonyl group is treated with Zn−Hg(Zinc Amalgum) and HCl
Which of the following compounds will give butanone on oxidation with alkaline KMnO4 solution? (i) Butan-1-ol (ii) Butan-2-ol (iii) Both of these (iv) None of these
Solution: Option (ii) is the answer.
Which is the most suitable reagent for the following conversion?(i) Tollen’s reagent (ii) Benzoyl peroxide (iii) I2 and NaOH solution (iv) Sn and NaOH solution
Solution: Option (iii) is the answer. Reason: This reaction is called as lodoform reaction.
Compound A and C in the following reaction are :_____________
Solution: Option (ii) is the answer. Reason:
Structure of ‘A’ and type of isomerism in the above reaction are respectively. (i) Prop–1–en–2–ol, metamerism (ii) Prop-1-en-1-ol, tautomerism (iii) Prop-2-en-2-ol, geometrical isomerism (iv) Prop-1-en-2-ol, tautomerism
Solution: Option (iv) is the answer. reason: Structure of A and the type of isomerism in the above reaction are Prop-1-en-2-ol, tautomerism respectively. Enol form tautomerises into keto...
Which product is formed when the compoundis treated with concentrated aqueous KOH solution?
Solution: Option (ii) is the answer. Reason: Benzaldhyde C6H5CHO on treatment with KOH yields the corresponding alcohol and acid. In this reaction, there is no alpha hydrogen atom present in...
Cannizaro’s reaction is not given by _____________.
Solution: Option (iv) is the answer. Reason: CH3CHO will not give Cannizzaro’s reaction because it contains a-hydrogen while other three compounds have no a-hydrogen. Hence, they will give...
The reagent which does not react with both, acetone and benzaldehyde. (i) Sodium hydrogen sulphite (ii) Phenyl hydrazine (iii) Fehling’s solution (iv) Grignard reagent
Solution: Option (iii) is the answer. Reason: Aromatic aldehydes and ketones does not respond to Fehling's test. Sodium hydrogen sulphite,phenyl hydrazine, grignard reaction are common for carbonyl...
Compound can be prepared by the reaction of _____________.
Solution: Option (ii) is the answer. Reason:
The correct order of increasing acidic strength is _____________. (i) Phenol < Ethanol < Chloroacetic acid < Acetic acid (ii) Ethanol < Phenol < Chloroacetic acid < Acetic acid (iii) Ethanol < Phenol < Acetic acid < Chloroacetic acid (iv) Chloroacetic acid < Acetic acid < Phenol < Ethanol
Solution: Option (iii) is the answer. Reason: The correct order of increasing acidic strength is Ethanol < Phenol < Acetic acid < Chloroacetic acid. Phenol is more acidic than ethanol...
A ball is dropped and its displacement vs time graph is as shown in the figure where displacement x is from the ground and all quantities are positive upwards. a) Plot qualitatively velocity vs time graph b) Plot qualitatively acceleration vs time graph
a) At t=0 and v=0 , v-t graph is: b) At x = 0, a-t graph is:
Which of the following compounds is most reactive towards nucleophilic addition reactions?
Solution: Option (i) is the answer.
A positively charged particle is released from rest in a uniform electric field. The electric potential energy of the charge a) remains a constant because the electric field is uniform b) increases because the charge moves along the electric field c) decreases because the charge moves along the electric field d) decreases because the charge moves opposite to the electric field
The correct answer is c) decreases because the charge moves along the electric field
Addition of water to alkynes occurs in acidic medium and the presence of Hg2+ ions as a catalyst. Which of the following products will be formed on addition of water to but-1-one under these conditions.
Solution: Option (ii) is the answer. Reason: Addition of water to but-1-yne in the presence of H2SO4 and HgSO4 gives 2-butaone. The addition takes place by markovnikoff's rule....
Arrange the following compounds in increasing order of dipole moment. CH3CH2CH3, CH3CH2NH2, CH3CH2OH
Solution: CH3CH2CH3 < CH3CH2NH2 < CH3CH2OH The dipole moment of CH3CH2OH is greater than that of CH3CH2NH2. CH3CH2CH3 has the least dipole moment among the three given compounds because it is...
Predict the product of the reaction of aniline with bromine in a non-polar solvent such as CS2.
Solution: The products formed in the reaction of aniline with bromine in a non-polar solvent such as CS2 are 4-Bromoaniline and 2-Bromoaniline where 4-Bromoaniline is the major product.
Under what reaction conditions (acidic/basic), the coupling reaction of aryldiazonium chloride with aniline is carried out?
Solution: This reaction is carried out in a mild basic medium. This is an electrophilic substitution reaction. Aryldiazonium chloride reacts with aniline to form a yellow dye of p-Aminoazobenzene.
Explain why MeNH2 is a stronger base than MeOH?
Solution: MeNH2 is a stronger base than MeOH because of the lower electronegativity and the presence of the lone pair of electrons on the nitrogen atom in MeNH2.
Why does the acetylation of —NH2 group of aniline reduce its activating effect?
Solution: The acetylation of —NH2 group of aniline reduces its activating effect because the lone pair of electrons on the nitrogen of acetanilide interacts with oxygen atom due to resonance.
Why is benzene diazonium chloride not stored and is used immediately after its preparation?
Solution: At high temperatures, benzene diazonium chloride is highly soluble in water, and at low temperatures, it is a very stable compound in water. Because it is unstable, it should be used as...
What is Hinsberg reagent?
Solution: Hinsberg's reagent is benzene sulphonyl chloride, also known as $C_6H_5SOCl$. To distinguish between primary, secondary, and tertiary amines, Hinsberg's reagent is used.
What is the best reagent to convert nitrile to primary amine?
Solution: LiAlH4 and Sodium/Alcohol are the best reagents for converting nitrile to primary amine. The nitriles can be converted into a corresponding primary amine through reduction.
What is the product when C6H5CH2NH2 reacts with HNO2?
Solution: The main product is $C_6H_5CH_2OH$ When $C_6H_5CH_2NH_2$ reacts with HNO2, it produces an unstable diazonium salt, which is then converted to alcohol. When $C_6H_5CH_2NH_2$ reacts with...
Why is NH2 group of aniline acetylated before carrying out nitration?
Solution: The NH2 group of aniline is acetylated before nitration to control the nitration reaction and the formation of tarry oxidation products and nitro derivatives. P-nitroaniline is the main...
What is the role of HNO3 in the nitrating mixture used for nitration of benzene?
Solution: The nitration of organic compounds is done with a nitration mixture, which is a 1:1 solution of HNO3 and H2SO4. In the nitration of benzene, it acts as a base and provides electrophile.
Which of the following reactions belong to electrophilic aromatic substitution?
(i) Bromination of acetanilide
(ii) Coupling reaction of aryldiazonium salts
(iii) Diazotisation of aniline
(iv) Acylation of aniline
Solution: Option (i) and (ii) are the answers. Reason:...
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc. H2SO4 + conc. HNO3
(ii) Acetic anhydride/pyridine followed by conc. H2SO4 + conc. HNO3
(iii) Dil. HCl followed by reaction with conc. H2SO4 + conc. HNO3
(iv) Reaction with conc. HNO3 + conc.H2SO4
Solution: Option (i) and (ii) are the answers. Reason: In addition to the nitro derivatives, direct nitration of aniline produces tarry oxidation products. Furthermore, in a strongly acidic...
Which of the following reactions are correct?
Solution: Option (i) and (iii) are the answers. Reason:
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methyl benzylamine
(iv) Aniline
Solution: Option (i) and (ii) are the answers. Reason: Gabriel synthesis is used for the preparation of primary amines. Phthalimide on treatment with ethanolic potassium hydroxide forms potassium...
Arenium ion involved in the bromination of aniline is __________.
Solution: Option (i), (ii) and (iii) are the answers. Reason: Arenium ion involved in the bromination of aniline are as follows:
The product of the following reaction is __________.
Solution: Option (A) and (B) is the answer. Reason:
The reagents that can be used to convert benzene diazonium chloride to benzene are __________.
(i) 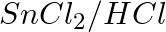
(ii) 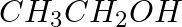
(iii) 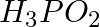
(iv) 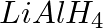
Solution: Option (ii) and (iii) are the answers. Reason: Hypophosphorous acid (phosphinic acid) and ethanol, for example, reduce diazonium salts to arenes, which are then oxidised to phosphorous...
Which of the following species are involved in the carbylamine test?
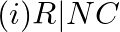
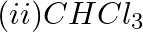
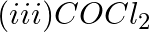
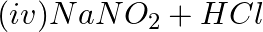
Solution: Option (i) and (ii) are the answers. Reason: Only RNC and CHCl3 are involved in carbylamine reaction.
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) Sn/HCl
(ii) Fe/HCl
(iii) H2-Pd
(iv) Sn/NH4OH
Solution: Option (i), (ii), and (iii) are the answers. Reason: They are reducing agents.
A brick manufacture has two depots, 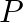 and
and 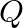 , with stocks of 30000 and 20000 bricks respectively. He receives order from three building
, with stocks of 30000 and 20000 bricks respectively. He receives order from three building 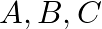 , for 15000,20000 and 15000 bricks respectively. The costs of transporting 1000 bricks to the building from the depots are given below. How should the manufacture fulfill the orders so as to keep the cost of transportation minimum?
, for 15000,20000 and 15000 bricks respectively. The costs of transporting 1000 bricks to the building from the depots are given below. How should the manufacture fulfill the orders so as to keep the cost of transportation minimum?
Let $x$ bricks be transported from $P$ to $A$ and y bricks be transported from $P$ to $B$. Therefore, $30000-(x+y)$ will be transported to $C$. Also, (15000-x) bricks, ( $20000-y)$ bricks and...
Two godowns, A and B, have a grain storage capacity of 100 quintals and 50 quintals respectively. Their supply goes to three ration shops, D, E and 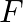 , whose requirements are 60,50 and 40 quintals respectively. The costs of transportation per quintal from the godowns to the shops are given in the following table.
, whose requirements are 60,50 and 40 quintals respectively. The costs of transportation per quintal from the godowns to the shops are given in the following table.
$$ \begin{tabular}{|c|c|c|} \hline & \multicolumn{2}{|c|}{ Cost of transportation (in 2 perquintal) } \\ \hline To & From & B & B \\ \hline$D$ & $6.00$ & 400 \\ \hline$E$ & $3.00$ & $2.00$ \\...
A gardener has a supply of fertilizers of the type 1 which consist of 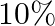 nitrogen and
nitrogen and 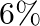 phosphoric acid, and of the type II which consist of
phosphoric acid, and of the type II which consist of 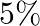 nitrogen and
nitrogen and 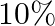 phosphoric acid. After testing the soil condition, he finds that he needs at least
phosphoric acid. After testing the soil condition, he finds that he needs at least 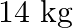 of nitrogen and
of nitrogen and 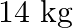 of phosphoric acid for his crop. If the type – I fertilizer costs 60 paise per kg and the type – II fertilizer costs 40 paise per kg, determine how many kilograms of each type of fertilizer should be used so that the nutrient requirement are met at a minimum cost. What is the minimum cost?
of phosphoric acid for his crop. If the type – I fertilizer costs 60 paise per kg and the type – II fertilizer costs 40 paise per kg, determine how many kilograms of each type of fertilizer should be used so that the nutrient requirement are met at a minimum cost. What is the minimum cost?
Let $x$ and $y$ be number of kilograms of fertilizer I and II, $\therefore$ According to the question, $0.10 x+0.05 y \geq 14,0.06 x+0.10 y \geq 14, x \geq 0, y \geq 0$ Minimize $Z=0.60 x+0.40 y$...
A publisher sells a hardcover edition of a book for 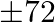 and a paperback edition of the same for
and a paperback edition of the same for 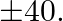 Costs to minutes of printing time although the hardcover edition requires 10 minutes of binding time and the paperback edition requires only 2 minutes. Both the printing and binding operations have 4800 minutes available each week. How many of each type of books should be produced in order to maximize the profit? Also, find the maximum profit per week.
Costs to minutes of printing time although the hardcover edition requires 10 minutes of binding time and the paperback edition requires only 2 minutes. Both the printing and binding operations have 4800 minutes available each week. How many of each type of books should be produced in order to maximize the profit? Also, find the maximum profit per week.
Let $x$ and $y$ be number of hardcover and paperback edition of the book. $\therefore$ According to the question, $5 x+5 y \leq 4800,10 x+2 y \leq 4800, x \geq 0, y \geq 0$ Maximize $Z=(72 x+40...
A man owns a field area 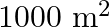 . He wants to plant fruit trees in it. He has a sum of
. He wants to plant fruit trees in it. He has a sum of 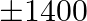 to purchase young trees. He has the choice of two types of trees. Type A requires
to purchase young trees. He has the choice of two types of trees. Type A requires 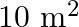 of ground per trees and costs
of ground per trees and costs 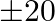 per tree, and type B requires
per tree, and type B requires 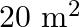 of ground per tree and costs
of ground per tree and costs 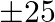 per tree. When full grown, a type A tree produces an average of
per tree. When full grown, a type A tree produces an average of 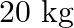 of fruit which can be sold at a profit
of fruit which can be sold at a profit 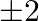 per
per 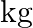 and type
and type 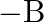 tree produces an average of
tree produces an average of 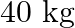 of fruit which can be sold at a profit of
of fruit which can be sold at a profit of 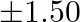 per
per 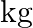 . How many of each type should be planted to achieve maximum profit when tree are full grown? What is the maximum profit?
. How many of each type should be planted to achieve maximum profit when tree are full grown? What is the maximum profit?
Let $x$ and $y$ be number of $A$ and B trees. $\therefore$ According to the question, $20 x+25 y \leq 1400,10 x+20 y \leq 1000, x \geq 0, y \geq 0$ Maximize $Z=40 x+60 y$ The feasible region...
A manufacture makes two product, A and B. product A sells at 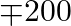 each and takes
each and takes 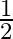 hour to make. Product B sells at
hour to make. Product B sells at 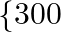 each and takes 1 hour to make. There is a permanent order for 14 of product
each and takes 1 hour to make. There is a permanent order for 14 of product  and 16 of product B. A working week consist of 40 hours of production and the weekly turnover must not be less than \mp10000. If the profit on each of the product
and 16 of product B. A working week consist of 40 hours of production and the weekly turnover must not be less than \mp10000. If the profit on each of the product  is
is 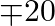 and on product
and on product 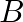 , it is
, it is 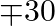 then how many of each should be produced so that the profit is maximum? Also, find the maximum profit.
then how many of each should be produced so that the profit is maximum? Also, find the maximum profit.
Let $x$ and $y$ be number of $A$ and $B$ products. $\therefore$ According to the question. $0.5 x+y \leq 40,200 x+300 y \geq 10000, x \geq 14, y \geq 16$ Maximize $Z=20 x+30 y$ The feasible region...
A manufacture makes two types, A and B, of teapots. Three machines are needed for the manufacture and the time required for each teapot on the machines is given below. Each machine is available for a maximum of 6 hours per day. If the profit on each teapot of type  is 75 paise and that on each teapot of type
is 75 paise and that on each teapot of type 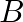 is 50 paise, show that 15 teapots of type
is 50 paise, show that 15 teapots of type  and 30 of type B should be manufactured in a day to get the maximum profit.
and 30 of type B should be manufactured in a day to get the maximum profit.
$$ \begin{tabular}{|l|l|l|l|} \hline Machine & \multicolumn{2}{|l|}{ Time (in minutes) } \\ \hline Type & I & II & III \\ \hline A & 12 & 18 & 6 \\ \hline B & 6 & 0 & 9 \\ \hline \end{tabular} $$...
Which of the following cannot be prepared by Sandmeyer’s reaction?
(i) Chlorobenzene
(ii) Bromobenzene
(iii) Iodobenzene
(iv) Fluorobenzene
Solution: Option (iii) and (iv) are the answers. Reason: Sandmeyer's reaction is used for the preparation of chlorobenzene and bromobenzene.
A small firm manufactures gold rings and chains. The combined number of rings and chains manufactured per day is at most 24. It takes 1 hour to make a ring and half an hour for a chain. The maximum number of hour to available per day is 16 . If the profit on a ring is 300 and that on a chain is 190, how many of each should be manufactured daily so as to maximize the profit?
Let $x$ and $y$ be number of gold rings and chains. $\therefore$ According to the question, $x+y \leq 24, x+0.5 y \leq 16, x \geq 0, y \geq 0$ Maximize $Z=300 x+190 y$ The feasible region determined...
A company producing soft drinks has a contrast which requires a minimum of 80 units of chemical  and 60 , units of chemical
and 60 , units of chemical 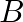 to go in each bottle of the drink. The chemical are available in a prepared mix from two different suppliers. Supplier
to go in each bottle of the drink. The chemical are available in a prepared mix from two different suppliers. Supplier 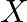 has a mix of 4 units of
has a mix of 4 units of  and 2 units of
and 2 units of 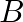 that costs Rs.10, and the supplier
that costs Rs.10, and the supplier 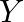 has a mix of 1 unit of
has a mix of 1 unit of  and 1 unit of
and 1 unit of 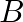 that costs \mp4. How many mixes from
that costs \mp4. How many mixes from 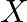 and
and 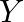 should the company purchase to honor the contract requirement and yet minimize the cost?
should the company purchase to honor the contract requirement and yet minimize the cost?
Let $x$ and $y$ be number of mixes from suppliers $X$ and $Y$. $\therefore$ According to the question, $4 x+y \geq 80,2 x+y \geq 60, x \geq 0, y \geq 0$ Minimize $Z=10 x+4 y$ The feasible region...
Which of the following methods of preparation of amines will not give the same number of carbon atoms in the chain of amines as in the reactant?
(i) The reaction of nitrite with LiAlH4.
(ii) The reaction of the amide with LiAlH4 followed by treatment with water.
(iii) Heating alkyl halide with potassium salt of phthalimide followed by hydrolysis.
(iv) Treatment of amide with bromine in the aqueous solution of sodium hydroxide.
Solution: Option (iv) is the answer. Reason: In Hoffmann Bromide degradation as the word, suggest, the amide is reduced to an amine with 1- carbonless, so this is the method in which we don't get...
Which of the following should be most volatile?
Solution: Option (ii) is the answer. Reason: The order of boiling points of isomeric amines is 1 amine > 2 amines > 3 amines. 3 amines have no intermolecular association because there are no H...
A small manufacture has employed 5 skilled men and 10 semiskilled men and makes an article in two qualities, a deluxe model and an ordinary model. The making of a deluxe model requires 2 hours work by a skilled man and 2 hours work by a semiskilled man. The ordinary model requires 1 hour by a skilled man and 3 hours by a semiskilled man. By union rules, no man can work more than 8 hours per day. The manufacture gains \mp 15 on the deluxe model and \mp 10 on the ordinary model. How many of each type should be made in order to maximize his total daily profit? Also, find the maximum daily profit.
Let $x$ and $y$ be number of deluxe article manufactured and ordinary article manufactured. $\therefore$ According to the question, $2 x+y \leq 40,2 x+3 y \leq 80, x \geq 0, y \geq 0$ Maximize $Z=15...
A toy company manufactures two types of dolls, A and B. Each doll of type B take twice as long to produce as one of type A, and the company would have time to make a maximum of 2000 per day, if it produces only type A. the supply of plastic is sufficient to produce 1500 dolls per day (both A and B combined). Type B requires a fancy dress of which there are only 600 per day available. If the company makes profit of Rs.3 and 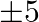 per dolls respectively on dolls A and B, how many of each should be produced per day in order to maximize the profit? Also, find the maximum profit.
per dolls respectively on dolls A and B, how many of each should be produced per day in order to maximize the profit? Also, find the maximum profit.
Let $x$ and $y$ be number of doll A manufactured and doll B manufactured. $\therefore$ According to the question, $x+y \leq 1500, x+2 y \leq 2000, y \leq 600, x \geq 0, y \geq 0$ Maximize $Z=3 x+5...
Among the following amines, the strongest Brönsted base is __________.
Solution; Option (iv) is the answer. Reason: Option (iv) is the strongest Bronsted base as there is no delocalization of lone pair of electron of the atom which is not possible in aniline and in...
The correct decreasing order of basic strength of the following species is _______. H2O, NH3, OH–, NH2– (i) NH2– > OH – > NH3 > H2O (ii) OH– > NH2– > H2O > NH3 (iii) NH3 > H2O > NH2– > OH– (iv) H2O > NH3> OH– > NH2–
Solution: Option (i) is the answer. Reason: NH3 is more basic than H2O, therefore NH2− (Conjugate base of weak acid NH3) is a stronger base than OH−.
Among the following amines, the strongest Brönsted base is __________.
Solution; Option (iv) is the answer. Reason: Option(iv)is the strongest Bronsted base as there is no delocalisation of lone pair of electron of N atom which is not possible in aniline and in...
Which of the following compounds is the weakest Brönsted base?
Solution: Option (iii) is the answer. Reason: A Bronsted Lowry base is a proton acceptor or hydrogen ion acceptor. Amines have a stronger tendency to accept protons and are strong Bronsted bases....
A manufacture of a line of patent medicines is preparing a production plan on medicines  and
and 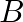 . There are sufficient ingredients available to make 20000 bottles of
. There are sufficient ingredients available to make 20000 bottles of  and 40000 bottles of B but there are only 45000 bottles into which either of the medicines can be put. Furthermore, it takes 3 hours to prepare enough material to fill 1000 bottles of A and it takes 1 hour to prepare enough material to fill 1000 bottles of
and 40000 bottles of B but there are only 45000 bottles into which either of the medicines can be put. Furthermore, it takes 3 hours to prepare enough material to fill 1000 bottles of A and it takes 1 hour to prepare enough material to fill 1000 bottles of 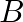 , and there are 66 hours available for this operation. The profit is
, and there are 66 hours available for this operation. The profit is 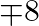 per bottle for
per bottle for  and
and 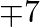 per bottle for
per bottle for 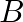 . How should the manufacture schedule the production in order to maximize his profit? Also, find the maximum profit.
. How should the manufacture schedule the production in order to maximize his profit? Also, find the maximum profit.
Let $x$ and $y$ be number of bottles of medicines $A$ and $B$ be prepared. $\therefore$ According to the question, $x+y \leq 45000,3 x+y \leq 66000, x \leq 20000, y \leq 40000 \cdot x \geq 0, y \geq...
Which of the following compound will not undergo an azo coupling reaction with benzene diazonium chloride.
(i) Aniline
(ii) Phenol
(iii) Anisole
(iv) Nitrobenzene
Solution: Option (iv) is the answer. Reason: Diazonium cation is a weak electrophile and hence it reacts with electron-rich compounds containing electron-donating groups such as −OH, -$NH_2$ and...
Evaluate the following integrals: 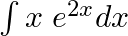
As per the given question,
A manufactures produces two types of soap bars using two machines, A and B. A is operated for 2 minutes and 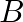 for 3 minutes to manufacture the first type, while it takes 3 minutes on machine
for 3 minutes to manufacture the first type, while it takes 3 minutes on machine  and 5 minutes on machine B to manufacture the second type. Each machine can be operated at the most for 8 hours per day. The two types of soap bars are sold at a profit of
and 5 minutes on machine B to manufacture the second type. Each machine can be operated at the most for 8 hours per day. The two types of soap bars are sold at a profit of 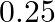 and
and 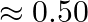 each. Assuming that the manufacture can sell all the soap bars he can manufacture, how many bars of soap of each type should be manufactured per day so as to maximize his profit?
each. Assuming that the manufacture can sell all the soap bars he can manufacture, how many bars of soap of each type should be manufactured per day so as to maximize his profit?
Let $x$ and $y$ be number of soaps be manufactured of $1^{\text {st }}$ and $2^{\text {nd }}$ type. $\therefore$ According to the question, $2 x+3 y \leq 480,3 x+5 y \leq 480, x \geq 0, y \geq 0$...
Evaluate the following integrals: 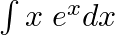
As per the given question,
The best method for preparing primary amines from alkyl halides without changing the number of carbon atoms in the chain is
(i) Hoffmann Bromamide reaction
(ii) Gabriel phthalimide synthesis
(iii) Sandmeyer reaction
(iv) Reaction with 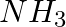
Solution: Option (ii) is the answer. Reason: Best method for preparing primary aminos form alkyl halides without changing the number of carbon atoms in the chain is Gabriel synthesis. Because this...
Evaluate the following integrals: 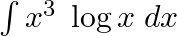
As per the given question,
A firm manufactures two types of products,  and
and 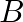 , and sells them at a profit of
, and sells them at a profit of  on type
on type  and B. Each product is processed on two machines,
and B. Each product is processed on two machines, 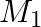 and
and 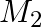 . Type A requires one minute of processing time on
. Type A requires one minute of processing time on 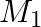 and two minutes on
and two minutes on 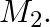 Type
Type 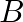 requires one minute on
requires one minute on 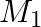 and one minute on
and one minute on 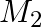 is available for not more than 6 hours 40 minutes while
is available for not more than 6 hours 40 minutes while 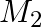 is available for at most 10 hours a day. Find how many products of each type the firm should produce each day in order to get maximum profit.
is available for at most 10 hours a day. Find how many products of each type the firm should produce each day in order to get maximum profit.
Let the firm manufacture $x$ number of Aand y number of $B$ products. $\therefore$ According to the question, $X+y \leq 400,2 x+y \leq 600, x \geq 0, y \geq 0$ Maximize $Z=2 x+2 y$ The feasible...
Evaluate the following integrals: 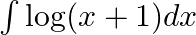
As per the given question,
The reaction Ar + N2Cl– → (Cu/HCl)– ArCl + N2 + CuCl is named as _________.
(i) Sandmeyer reaction
(ii) Gatterman reaction
(iii) Claisen reaction
(iv) Carbylamine reaction
Solution: Option (ii) is the answer. Reason: Diazonium salts in the presence of copper powder and halogen acid give aryl halide. Gattermann reaction is a variation of Sandmeyer reaction in which...
A dealer wishes to purchase a number of fans and sewing machines. He has only 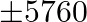 to invest and space and
to invest and space and 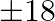 on a sewing machine. Assuming that he can sell all the items he can buy, how should he invest the money in order to maximize the profit?
on a sewing machine. Assuming that he can sell all the items he can buy, how should he invest the money in order to maximize the profit?
Let the number of fans bought be $x$ and sewing machines bought be $y$. $\therefore$ According to the question, $360 x+240 y \leq 5760, x+y \leq 20, x \geq 0, y \geq 0$ Maximize $Z=22 x+18 y$ The...
Evaluate the following integrals: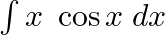
As per the given question,
Question while B can stitch 10 shirts and 4 pairs of trousers per day. How many days should each of them work if it is desired to produce at least 60 shirts and 32 pairs of trousers at a minimum labor cost?
Let the total number of days tailor $A$ work be $x$ and tailor $B$ be $y$. $\therefore$ According to the question, $6 x+10 y \geq 60,4 x+4 y \geq 32, x \geq 0, y \geq 0$ Minimize $Z=300 x+400 y$ The...
Acid anhydrides on reaction with primary amines give ____________.
(i) amide
(ii) imide
(iii) secondary amine
(iv) imine
Solution: Option (i) is the answer Reason: When acid anhydrides react with primary amines, they produce amide. The H atom of the amino group is replaced with an acyl group in this nucleophilic...
Evaluate the following integrals: 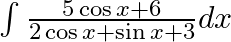
As per the given question,
Evaluate the following integrals: 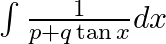
As per the given question,
A manufacture produces nuts and bolts for industrial machinery. It takes 1 hour of work on machine  and 3 hours on machine B to produces a packet of nuts while it takes 3 hours on machine
and 3 hours on machine B to produces a packet of nuts while it takes 3 hours on machine  and 1 hours on machine B to produce a packet of bolts. He earns a profit \mp17.50 per packet on nuts and \mp7 per packet on bolts. How many packets of each should be produced each day so as to maximize his profit if he operates, each machine for at the most 12 hours a day? Also find the maximum profit.
and 1 hours on machine B to produce a packet of bolts. He earns a profit \mp17.50 per packet on nuts and \mp7 per packet on bolts. How many packets of each should be produced each day so as to maximize his profit if he operates, each machine for at the most 12 hours a day? Also find the maximum profit.
Let the number of packets of nuts and bolts be $x$ and y respectively. $\therefore$ According to the question, $x+3 y \leq 12,3 x+y \leq 12, x \geq 0, y \geq 0$ Maximize $Z=17.50 x+7 y$ The feasible...
Evaluate the following integrals: 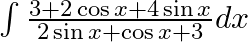
As per the given question,
A man has 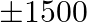 to purchase rice and wheat. A bag of rice and a bag of wheat cost \mp 180 and 120 respectively. He has storage capacity of 10 bags only. He earns a profit of
to purchase rice and wheat. A bag of rice and a bag of wheat cost \mp 180 and 120 respectively. He has storage capacity of 10 bags only. He earns a profit of 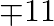 and 78 per bag of rice and wheat respectively. How many bags of each must he buy to make maximum profit?
and 78 per bag of rice and wheat respectively. How many bags of each must he buy to make maximum profit?
Let the number of wheat and rice bags be $x$ and $y$. $\therefore$ According to the question, $120 x+180 y \leq 1500, x+y \leq 10, x \geq 0, y \geq 0$ Maximize $Z=8 x+11 y$ The feasible region...
Evaluate the following integrals: 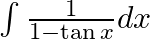
As per the given question,
Evaluate the following integrals: 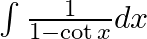
As per the given question,
A small firm manufactures necklace and bracelets. The total number of necklace and bracelet that it can handle per day is at most 24. It takes 1 hour to make a bracelet and half an hour to make a necklace. The maximum number of hours available per day is 16 . If the profit on a necklace is 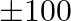 and that on a bracelet is
and that on a bracelet is 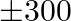 , how many of each should be produced daily to maximize the profit? It is being given that at least one of each must be produced.
, how many of each should be produced daily to maximize the profit? It is being given that at least one of each must be produced.
Let the firm manufacture $x$ number of necklaces and y number of bracelets a day. $\therefore$ According to the question, $x+y \leq 24,0.5 x+y \leq 16 x \geq 1, y \geq 1$ Maximize $Z=100 x+300 y$...
Reduction of aromatic nitro compounds using Fe and HCl gives __________.
(i) aromatic oxime
(ii) aromatic hydrocarbon
(iii) aromatic primary amine
(iv) aromatic amide
Solution: Option (iii) is the answer. Reason: Reduction of nitro aryl compounds in presence of Fe and HCl gives aromatic primary amines.
Mr.Dass wants to invest Rs 12000 in public provident fund (PPF) and in national bonds. He has to invest at 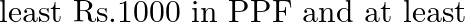 bonds is
bonds is 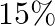 per annum, how should he invest the money to eam maximum annual income? Also find the maximum annual income.
per annum, how should he invest the money to eam maximum annual income? Also find the maximum annual income.
Let the invested money in PPF be $x$ and in national bonds be $y$. $\therefore$ According to the question, $\begin{array}{l} \mathrm{X}+\mathrm{y} \leq 12000 \\ \mathrm{x} \geq 1000, \mathrm{y} \geq...
Evaluate the following integrals: 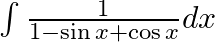
As per the given question,
In the nitration of benzene using a mixture of conc. 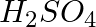 and conc.
and conc. 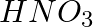 ,the species which initiates the reaction is __________.
,the species which initiates the reaction is __________.
(i) 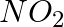
(ii) 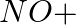
(iii) 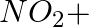
(iv) 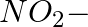
Solution: Option (iii) is the answer. Reason:
Evaluate the following integrals: 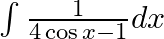
As per the given question,
Evaluate the following integrals: 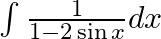
As per the given question,
Find the maximum and minimum values of 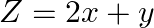 , subject to the constraints
, subject to the constraints 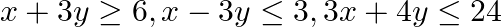
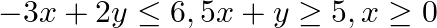 and
and 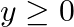
The feasible region determined by $x+3 y \geq 6, x-3 y \leq 3,3 x+4 y \leq 24$ $-3 x+2 y \leq 6,5 x+y \geq 5, x \geq 0$ and $y \geq 0$ is given by The corner points of the feasible region are $A(4 /...
The gas evolved when methylamine reacts with nitrous acid is __________.
(i) 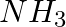 (ii)
(ii) 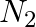 (iii)
(iii) 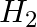 (iv)
(iv) 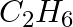
Solution: Option (ii) is the answer. Reason:
Evaluate the following integrals: 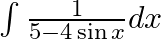
As per the given question,
Maximize 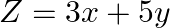 , subject to the constraints
, subject to the constraints 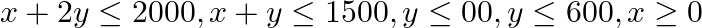 and
and 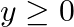
The feasible region determined by the $X+2 y \leq 2000, x+y \leq 1500, y \leq 600, x \geq 0$ and $y \geq 0$ is given by The corner points of the feasible region are $A(0,0), B(0,600), C(800,600),...
Evaluate the following integrals: 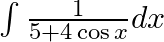
As per the given question,
Evaluate the following integrals: 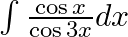
As per the given question,
Minimize 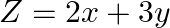 , subject to the constraints
, subject to the constraints 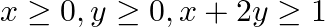 and
and 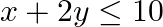
The feasible region determined by the $x \geq 0, y \geq 0, x+2 y \geq 1$ and $x+2 y \leq 10$ is given by The corner points of the feasible region is $A\left(0 \frac{1}{2}\right), B(0,5), C(10,0),...
Evaluate the following integrals: 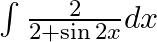
As per the given question,
Methylamine reacts with HNO2 to form _________.
(i) 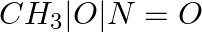 (ii)
(ii) 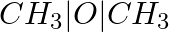 (iii)
(iii) 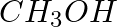 (iv)
(iv) 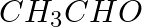
Solution: Option (iii) is the answer. Reason:
Evaluate the following integrals: 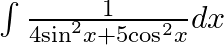
As per the given question,
Evaluate the following integrals: 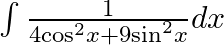
As per the given question,
Evaluate the following integrals: 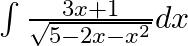
As per the given question,
The correct increasing order of basic strength for the following compounds is _________.
(i) II < III < I
(ii) III < I < II
(iii) III < II < I
(iv) II < I < III
Solution: Option (iv) is the answer. Reason: Electron donating: group increases the basicity while electron-withdrawing group decreases the basicity of...
Evaluate the following integrals: 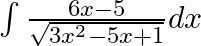
As per the given question,
Find the minimum value of 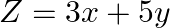 , subject to the constraints
, subject to the constraints  and
and 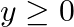
The feasible region determined by the $-2 x+y \leq 4, x+y \geq 3, x-2 y \leq 2, x \geq 0$ and $y_{z} 0$ is given by Here the feasible region is unbounded. The vertices of the region are $A(0,4),...
Evaluate the following integrals: 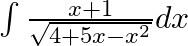
As per the given question,
Evaluate the following integrals: 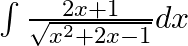
As per the given question,
Maximize 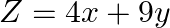 , subject to the constraints
, subject to the constraints 
The feasible region determined by the constraints $x \geq 0, y \geq 0, x+5 y \leq 200,2 x+3 y \leq 134$ is given by The corner points of feasible region are $A(10,38), B(0,40), C(0,0), D(67,0)$. The...
Evaluate the following integrals: 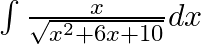
As per the given question,
Evaluate the following integrals: 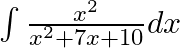
As per the given question, Hence,
Find the maximum value of 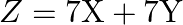 , subject to the constraints.
, subject to the constraints. 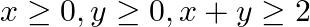 and
and 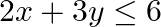
The feasible region determined by the constraints $x \geq 0, y \geq 0$, $x+y \geq 2,2 x+3 y \leq 6$ is given by The corner points of the feasible region is $A(0,2), B(2,0), C(3,0)$. The values of...
Evaluate the following integrals: 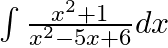
As per the given question,
Find the linear constraints for which the shaded area in the figure given is the solution set.
Solution: Consider A: Given line $x-y=1$ $\Rightarrow y=x-1$ As the region given in the figure is above the $y$ - intercept's coordinates $(0,-1)$, $\begin{array}{l} \Rightarrow y \geq x-1 \\...
Show that the solution set of the following linear constraints is empty: 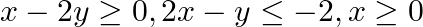 and
and 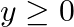
Consider the inequation $x-2 y \geq 0$ $\begin{array}{l} \Rightarrow x \geq 2 y \\ \Rightarrow y \leq \frac{x}{2} \end{array}$ consider the equation $y=\frac{x}{2}$. This equation's graph is a...
Hoffmann Bromamide Degradation reaction is shown by __________.
(i) 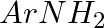
(ii) 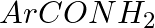
(iii) 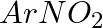
(iv) 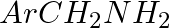
Solution: Option (ii) is the answer. Reason: Where the aryl amide is converted to arylamine in the presence of $Br_2$ and $NaOH$ .
Evaluate the following integrals: 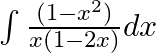
As per the given question,
Evaluate the following integrals: 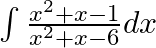
\[\Rightarrow ~1\text{ }=\text{ }\left( A\text{ }+\text{ }B \right)\text{ }x\text{ }+\text{ }\left( 3A\text{ }\text{ }2B \right)\] $⇒$ Then $A\;+\;B\;=\;0\;… (1)$ And $3A\;–\;2B\;=\;1\;… (2)$...
Solve each of the following systems of simultaneous inequations: 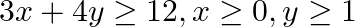 and
and 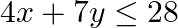
Consider the inequation $3 x+4 y \geq 12$ $\Rightarrow 4 y \geq 12-3 x$ $\Rightarrow y \geq 3-\frac{3}{4} x$ Consider the equation $y=3-\frac{3}{4} x$ Finding points on the coordinate axes: If...
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanolamine is _____.
(i) excess H2 (ii) Br2 in aqueous NaOH (iii) iodine in the presence of red phosphorus (iv) LiAlH4 in ether
Solution: Option (iv) is the answer. Reason:
Evaluate the following integrals: 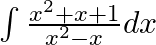
As per the given question,
Solve each of the following systems of simultaneous inequations: 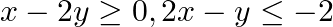
Consider the inequation $x-2 y \geq 0$ : $\begin{array}{l} \Rightarrow x \geq 2 y \\ \Rightarrow y \leq \frac{x}{2} \end{array}$ consider the equation $y=\frac{x}{2}$. This equation's graph is a...
Amongst the given set of reactants, the most appropriate for preparing 2° amine is _____.
(i) 2° R—Br + NH3
(ii) 2° R—Br + NaCN followed by 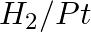
(iii) 1° R—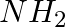 + RCHO followed by %H_2/Pt
+ RCHO followed by %H_2/Pt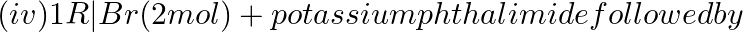 H_3O+$/heat
H_3O+$/heat
Solution: Option (iii) is the answer. Reason:
Write the order and degree (if defined) of each of the following equations.
Solution: $y=\frac{dy}{dx}+\frac{5}{\left( \frac{dy}{dx} \right)}$ On rearranging - $y*\frac{dy}{dx}={{\left( \frac{dy}{dx} \right)}^{2}}+5$ $Order\;=\;2$ (represents the order of the top-most...
Let 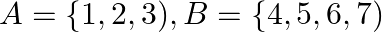 and let
and let 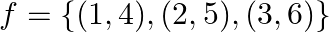 be a function from
be a function from  to
to 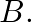 State whether
State whether 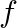 is one-one.
is one-one.
Solution: We need to state: Whether $\mathrm{f}$ is one-one Given that: $f=\{(1,4),(2,5),(3,6)\}$ Here the function is defined from $A \rightarrow B$ For a function to be one-one if the images of...
Let 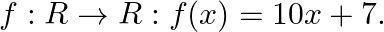 Find the function
Find the function 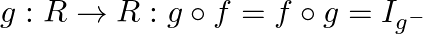
Solution: We need to find: the function $g: R \rightarrow R: g \circ f=f$ o $g=I_{g}$ Formula used: (i) $g$ o $f=g(f(x))$ (ii) f o $g=f(g(x))$ Given that: $f: \mathbb{R} \rightarrow \mathbb{R}:...
The source of nitrogen in Gabriel synthesis of amines is _____________.
(i) Sodium azide, NaN3
(ii) Sodium nitrite, NaNO2
(iii) Potassium cyanide, KCN
(iv) Potassium phthalimide
Solution: Option (iv) is the answer. Reason: Gabriel synthesis :The reaction is given to the image.Source of nitrogen atom is Gabriel synthesis is Potassium phthalamide.
Let 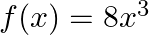 and
and 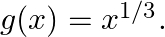 Find
Find 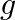 o
o 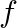 and
and 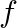 o
o 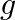 .
.
Solution: We need to find: $g$ o f and f o $\mathrm{g}$ Formula used: (i) f o $\mathrm{g}=\mathrm{f}(\mathrm{g}(\mathrm{x}))$ (ii) $\mathrm{g} \circ \mathrm{f}=\mathrm{g}(\mathrm{f}(\mathrm{x}))$...
Let 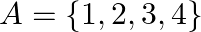 and
and 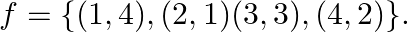 Write down (f of).
Write down (f of).
Solution: We need to find: f of Formula used: $f o f=f(f(x))$ Given that: (i) $f=\{(1,4),(2,1)(3,3),(4,2)\}$ We have, $\text { fof }(1)=f(f(1))=f(4)=2$ $\begin{array}{l}...
Solve each of the following systems of simultaneous inequations: 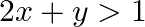 and
and 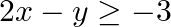
Consider the inequation $2 x+y>1:$ $ \Rightarrow y>1-2 x$ Consider the equation $y=1-2 x$ Finding points on the coordinate axes: If $x=0$, the y value is 1 i.e, $y=1$ $\Rightarrow$ the point...
Let 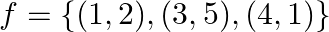 and
and 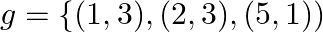 . Write down
. Write down 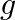 o f.
o f.
Solution: We need to find: g of Formula used: $g$ o $f=g(f(x))$ Given that: (i) $f=\{(1,2),(3,5),(4,1)\}$ (ii) $g=\{(1,3),(2,3),(5,1)\}$ We have, $\begin{array}{l}...
To prepare a 1° amine from an alkyl halide with simultaneous addition of one 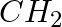 group in the carbon chain, the reagent used as a source of nitrogen is ___________.
group in the carbon chain, the reagent used as a source of nitrogen is ___________.
(i) Sodium amide, NaNH2
(ii) Sodium azide, NaN3
(iii) Potassium cyanide, KCN
(iv) Potassium phthalimide
Solution: Option (iii) is the answer. Reason:
Let 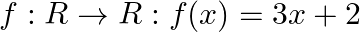 , find
, find 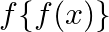
Solution: We need to find: $f\{f(x)\}$ Formula used: (i) $f \circ f=f(f(x))$ Given that: (i) $f: R \rightarrow R: f(x)=3 x+2$ We have, $\begin{array}{l} f\{f(x)\}=f(f(x))=f(3 x+2) \\ \text { fo }...
Write the order and degree (if defined) of each of the following equations.
Solution: $Order\;=\;1$ (represents the order of the top-most derivative involved in the equation derivative involved in the equation) $Degree\;=\;1$ (degree is the highest power to which a...
Let 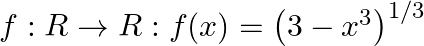 . Find f of.
. Find f of.
Solution: We need to find: f o f Formula used: (i) f o $f=f(f(x))$ Given that: (i) $f: R \rightarrow R: f(x)=\left(3-x^{3}\right)^{1 / 3}$ We have, $\begin{array}{l} \text { fo }...
Graph the solution sets of the following inequations: 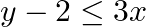
Given $y-2 \leq 3 x$ $\Rightarrow y \leq 3 x+2$ Consider the equation $y=3 x+2$ Finding points on the coordinate axes: If $x=0$, the $y$ value is 2 i.e, $y=2$ $\Rightarrow$ the point on $Y$ axis is...
Let 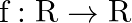 and
and 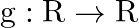 defined by
defined by 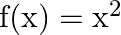 and
and 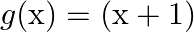 . Show that
. Show that 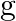 o
o 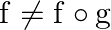 .
.
Solution: We need to prove: $g$ o $f \neq f$ o $g$ Formula used: (i) f o $\mathrm{g}=\mathrm{f}(\mathrm{g}(\mathrm{x}))$ (ii) $g$ of $=g(f(x))$ Given that: (i) $f: R \rightarrow R: f(x)=x^{2}$ (ii)...
Graph the solution sets of the following inequations: 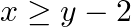
Given $x \geq y-2$ $\Rightarrow \mathrm{y} \leq \mathrm{x}+2$ Consider the equation $y=x+2$ Finding points on the coordinate axes: If $x=0$, the $y$ value is 2 i.e, $y=2$ $\Rightarrow$ the point on...
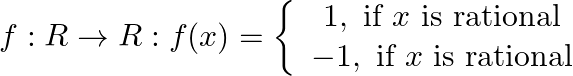 Show that
Show that 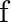 is many-one and into.
is many-one and into.
Solution: We need to prove: function is many-one and into It is given that: $f: R \rightarrow R: f(x)=\left\{\begin{array}{c}1, \text { if } x \text { is rational } \\ -1, \text { if } x \text { is...
Write the order and degree (if defined) of each of the following equations.
Solution: ${{(y'')}^{3}}+{{(y')}^{2}}+\sin y'+1=0$ $Order\;=\;3$ (represents the order of the top-most derivative involved in the equation derivative involved in the equation) $Degree\;=\;2$ (degree...
Let 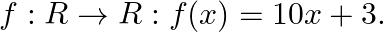 Find
Find 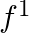
Solution: We need to find: $\mathrm{f}^{-1}$ It is given that: $f: R \rightarrow R: f(x)=10 x+3$ We have, $f(x)=10 x+3$ Suppose $f(x)=y$ such that $y \in R$ $\begin{array}{l} \Rightarrow y=10 x+3 \\...
6. Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine? (i) H2 (excess)/Pt (ii) LiAlH4 in ether (iii) Fe and HCl (iv) Sn and HCl
Solution: Option (ii) is the answer. Reason: LiAlH4/ether reduces aryl nitro compounds to azo compounds 2C6H5NO2→LiAIH4C6H5N=N-C6H5
Graph the solution sets of the following inequations: 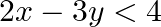
Given $2 x-3 y<4$ $\begin{array}{l} \Rightarrow 2 x-4<3 y \\ \Rightarrow 3 y>2 x-4 \\ \Rightarrow y>\frac{2}{3} x-\frac{4}{3} \end{array}$ Consider the equation $y=\frac{2}{3}...
Let 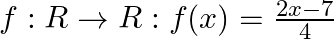 be an invertible function. Find
be an invertible function. Find 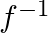
Solution: We need to find: $\mathrm{f}^{-1}$ It is given that: $f: R \rightarrow R: f(x)=\frac{2 x-7}{4}$ We have, $f(x)=\frac{2 x-7}{4}$ Suppose $f(x)=y$ such that $y \in R$ $\begin{array}{l}...
Show that the function 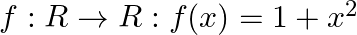 is many-one into.
is many-one into.
Solution: We need to prove: function is many-one into It is given that: $f: R \rightarrow R: f(x)=1+x^{2}$ We have, $f(x)=1+x^{2}$ For, $f\left(x_{1}\right)=f\left(x_{2}\right)$ $\begin{array}{l}...
Write the order and degree (if defined) of each of the following equations.
Solution: $\sqrt{1-{{y}^{2}}}dx+\sqrt{1-{{x}^{2}}}dy=0$ On rearranging - $\frac{dy}{dx}=-\frac{\sqrt{1-{{y}^{2}}}}{\sqrt{1-{{x}^{2}}}}$ $Order\;=\;1$ (represents the order of the top-most derivative...
Let 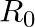 be the set of all nonzero real numbers. Then, show that the function
be the set of all nonzero real numbers. Then, show that the function 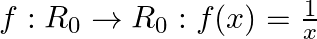 is oneone and onto.
is oneone and onto.
Solution: We need to prove: function is one-one and onto It is given that: $f: R_{0} \rightarrow R_{0}: f(x)=\frac{1}{x}$ We have, $f(x)=\frac{1}{x}$ For, $f\left(x_{1}\right)=f\left(x_{2}\right)$...

![Rendered by QuickLaTeX.com \int{_{0}^{16}{{x}^{\frac{3}{4}}}dx=\left[ \frac{4}{7}{{x}^{\frac{7}{4}}} \right]}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a5289eb5bb96809ad1fc7747e6a02654_l3.png)
![Rendered by QuickLaTeX.com \int{_{1}^{2}{{x}^{-5}}dx=\left[ \frac{{{x}^{-4}}}{-4} \right]}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-b793472d6e2b499501665d78886a1d89_l3.png)
![Rendered by QuickLaTeX.com \int{_{1}^{4}\sqrt{x}dx=\left[ \frac{2}{3}{{x}^{\frac{3}{2}}} \right]}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-7f4a1195fbd44abd6c01848b17dba0d5_l3.png)
![Rendered by QuickLaTeX.com \int{_{1}^{3}{{x}^{4}}dx=\left[ \frac{{{x}^{5}}}{5} \right]}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-062aa044351dc600fbdc6e340df27499_l3.png)