Solution: (c) When fermented K2Cr2O7 arrangement is added to Sn2+ salt, Sn2+ changes to Sn4+. The response is given underneath:
Which of the accompanying assertion isn’t right? (a) Copper frees hydrogen from acids (b) In its higher oxidation states, manganese structures stable mixtures with oxygen and fluorine (c) Mn3+ and Co3+ are oxidizing specialists in watery arrangement (d) Ti2+ and Cr2+ are decreasing specialists in watery arrangement
Solution: (a) Copper doesn't free hydrogen from acids. \[\begin{array}{*{35}{l}} Cu\text{ }+\text{ }2H2S04\text{ }\text{ }>\text{ }CuSO4\text{ }+\text{ }S02\text{ }+\text{ }2H2O \\ ~ \\...
KMn04 goes about as an oxidizing specialist in soluble medium. When soluble KMnO4 is treated with KI, iodide particle is oxidized to (a) I2 (b)Io–(c) I03 (d) I04
Solution: \[\left( c \right)\text{ }2KMnO4\text{ }+\text{ }KI\text{ }+\text{ }H2O\text{ }\text{ }>\text{ }2K0H\text{ }+\text{ }2MnO2\text{ }+\text{ }KI03\]
The attractive second is related with its twist rakish energy and orbital precise force. Twist just attractive second worth of Cr3+ particle is (a) 2.87 B.M. (b) 3.87 B.M. (c) 3.47 B.M. (d) 3.57 B.M.
Solution: (b) The attractive second is related with its twist precise force and orbital rakish energy.
Interstitial mixtures are shaped when little particles are caught inside the gem cross section of metals. Which of coming up next isn’t the trademark property of interstitial mixtures? (a) They have high liquefying focuses in contrast with unadulterated metals (b) They are exceptionally hard (c) They hold metallic conductivity (d) They are synthetically extremely receptive.
Solution: (d) Interstitial mixtures are synthetically idle.
Gadolinium has a place with 4f series. Its nuclear number is 64. Which of the following is the right electronic arrangement of gadolinium? (I) [Xe] 4f 75d16s2 (ii) [Xe] 4f 65d26s2 (iii) [Xe] 4f 86d2 (iv) [Xe] 4f 95s1
Solution: (a) 64Gd: [Xe] 4f7 5d1 6s2
Which of coming up next is amphoteric oxide? Mn2O7, CrO3, Cr2O3, CrO, V2O5, V2O4 (a) V2O5, Cr2O3 (b)Mn2O7, CrO3 (c) CrO, V2O5 (d) V2O5, V2O4
Solution: (a) V2O5 and Cr2O3 are amphoteric oxides on the grounds that both respond with alkalies just as acids. Keep in mind: In lower oxides, the fundamental person is transcendent while in higher...
KMnO4 goes about as an oxidizing specialist in acidic medium. The quantity of moles of KMnO4 that will be expected to respond with one mole of sulfide particles in acidic arrangement is (I) 2/5 (ii) 3/5 (iii) 4/5 (iv) 1/5
Solution:
There are 14 components in actinoid series. Which of the accompanying component doesn’t have a place with this series? (a) U (b) Np (c) Tm (d) Fm
Solution: (c) Tm (Thulium) is a lanthanoid.
At the point when KMnO4 arrangement is added to oxalic corrosive arrangement, the decolourisation is delayed to start with yet becomes momentary after some time on the grounds that (a) CO2 is shaped as the item (b) Reaction is exothermic (c) Mn04 catalyzes the response (d) Mn2+ goes about as autocatalyst
Solution: (d) When KMnO4 arrangement is added to oxalic corrosive arrangement, the decolourisation is delayed first and foremost yet becomes immediate after some time in light of the fact that Mn2+...
Which of the accompanying responses are disproportionation responses?
Solution:
Which of the accompanying oxidation state is normal for all lanthanoids? (a) +2 (b) +3 (c) +4 (d) +5
Solution: (b) +3 oxidation state is generally normal for all lanthanoids.
The attractive idea of components rely upon the presence of unpaired electrons. Distinguish the arrangement of change component, which shows the most elevated attractive second. (a) 3d7 (b) 3d5 (c) 3d8 (d) 3d2
Solution: (b) The more noteworthy the quantity of unpaired electron, the higher will be its worth of attractive second. Since, 3d5 has 5 unpaired electrons subsequently most noteworthy attractive...
On expansion of modest quantity of KMnO4 to concentrated H2SO4, a green slick compound is acquired which is exceptionally touchy in nature. Distinguish the compound from the accompanying: (a) Mn2O7 (b) MnO2 (c) MnSO4 (d) Mn2O3
Solution: \[\left( a \right)\text{ }2KMnO4\text{ }+\text{ }2H2SO4\left( Conc\text{ } \right)\text{ }\text{ }>\text{ }Mn2O7\text{ }+\text{ }2KHSO4\text{ }+\text{ }H2O\]
By and large, progress components structure shaded salts because of the presence of unpaired electrons. Which of the accompanying compound will be shaded in strong state? (a) Ag2SO4 (b) CUF2 (c) ZnF2 (d) Cu2Cl2
Solution: (b) Cu2+ has 1 unpaired electron in CuF2, consequently, it is hued in strong state.
Metallic radii of some change components are given underneath. Which of these components will have most noteworthy thickness?
Solution: (d) On moving passed on to directly along period, metallic span diminishes while mass increments. Diminishes in metallic span combined with expansion in nuclear mass outcomes in...
The electronic setup of Cu(II) is 3d9 while that of Cu(I) is 3d10. Which of coming up next is right? (I) Cu(II) is more steady (ii) Cu(II) is less steady (iii) Cu(I) and Cu(II) are similarly steady (iv) Stability of Cu(I) and Cu(II) relies upon the idea of copper salts
Solution: (a) Cu(II) is more steady because of more noteworthy compelling atomic charge of Cu(II).
Electronic design of a change component X in +3 oxidation state is [Ar]3d5. What is its nuclear number? (I) 25 (ii) 26 (iii) 27 (iv) 24
Solution:
The second and third lines of progress components look like each other significantly more than they take after the primary line. Clarify why?
Solution: Because of helpless f orbital safeguarding, powerful atomic charge increments and there is a compression in the size of the third-column components. This compression in size is called...
In spite of the fact that +3 oxidation states are the trademark oxidation condition of lanthanoids cerium shows +4 oxidation state too. Why?
Solution: Ce – [Xe] 4f1 5d1 6s2. For the most part, lanthanoids lose the 5d and 6s electrons and show +3 oxidation state, yet Cerium loses the one 4f electron additionally to achieve Xenon's...
In spite of the fact that Zr has a place with 4d and Hf has a place with 5d progress series yet it is very hard to isolate them. Why?
Solution: Because of the lanthanoid compression helpless f, orbital safeguarding prompts an expansion in powerful atomic charge, which lessens the size of Hf. So the nuclear radii of both Zr and Hf...
Ionization enthalpies of Ce, Pr and Nd are higher than Th, Pa and U. Why?
Solution: Th, Pa and U, 5f electrons begin filling and they have infiltration lower than 4f electrons for Ce, Pr and Nd. Eliminating 4f electrons will be troublesome, so ionization enthalpy for Th,...
In spite of the fact that Cr3+ and Co2+ particles have similar number of unpaired electrons the attractive snapshot of Cr3+ is 3.87 B.M. furthermore, that of Co2+ is 4.87 B.M. Why?
Solution: Cr3+ has an even electron appropriation and will just have turn attractive second commitment though Co2+ has no balanced circulation of electrons so it will have an orbital attractive...
In spite of the fact that fluorine is more electronegative than oxygen, the capacity of oxygen to balance out higher oxidation states surpasses that of fluorine. Why?
Solution: Fluorine has one unpaired electron and structures a solitary bond, while Oxygen has two unpaired electrons and can shape various bonds along these lines balancing out higher oxidation...
Out of Cu2Cl2 and CuCl2, which is more steady and why?
Solution: CuCl2 is more steady in light of the fact that Cu2+ has a higher electron thickness than Cu+. Cu2+ is more modest in size, has higher powerful atomic charge and subsequently a higher...
Change components show high dissolving focuses. Why?
Solution: Change components have solid metallic bonds. Breaking those bonds becomes more diligently that implies dissolving these components will be troublesome. Thus, liquefying focuses are higher...
Why E° esteems for Mn, Ni and Zn are more negative than anticipated?
Solution: Mn2+(3d5) and Zn2+(3d10) have half-filled and filled d orbitals which give them dependability and accordingly like to remain as such and not get diminished. With respect to Ni2+(3d8), it...
For what reason does copper not supplant hydrogen from acids?
Solution: A positive decrease potential means the diminished type of Cu is more steady than hydrogen. Consequently, Cu is less receptive than hydrogen and can't dislodge it from acids.
Between sodium hydrogen carbonate and magnesium hydroxide which is a superior acid neutralizer and why?
Solution: Magnesium hydroxide is a superior acid neutralizer in light of the fact that it keeps a pH level in the stomach and insoluble in the stomach. It doesn't make stomach antacid though sodium...
What is the essential contrast among germ-killers and sanitizers?
Solution: 1. Germicides are applied to living tissues though sanitizers are applied on non-living substances 2. Germicides are antimicrobial though sanitizers are against microbial as well 3....
What is the logical clarification for the sensation of sorrow?
Solution: A chemical called Noradrenaline controls the emotional episodes. Sorrow can be caused because of the low degree of noradrenaline which hampers the sign exercises in the cerebrum.
What are analgesics?
Solution: The analgesics drug is a neurologically dynamic medication which is utilized to diminish torment. They don't have any incidental effects.
For what reason is it more secure to utilize cleanser according to the ecological perspective?
Solution: When contrasted with the cleansers are more secure to utilize which is biodegradable. It doesn't have a contaminating nature.
How does the expanding of the hydrocarbon chain of manufactured cleansers influence their biodegradability?
Solution: Lesser the expanding lesser is the non-dirtying nature of the cleanser and expanded in fanning causes the contaminating idea of the cleanser to increment.
Draw the outline showing micelle development by the accompanying cleanser. CH3(CH2)10CH2OSO3-Na+
Solution: Micelle formation of the detergent can be shown as:
Dishwashing cleansers are engineered cleansers. What is their compound nature?
Solution: Cleansers are manufactured cleansers which contain non-ionic cleansers that have a purging property. They consolidate with the soil and debasements and make them dissolvable.
Hair shampoos have a place with which class of manufactured cleanser?
Solution: Cationic cleansers are utilized in hair shampoos. eg: cetyltrimethylammonium bromide. Cationic cleansers are quarternary ammonium salts of acetic acid derivations, chlorides or bromides.
Which class of the manufactured cleansers is utilized in toothpaste?
Solution: Anionic cleansers are utilized in toothpaste to clean teeth and structure reasonable froth. Eg: sodium or ammonium lauryl sulfate
Clarify why a few times frothing is found in waterway water close to where sewage water is poured after treatment?
Solution: The froth is because of the non-biodegradable cleansers which are available in water after sewage treatment. A cleanser is a water-solvent purifying specialist which joins with...
In the event that the cleanser has high antacid substance it disturbs the skin. How could the measure of abundance soluble still up in the air? What can be the wellspring of overabundance antacid?
Solution: Overabundance of antacid can be discovered utilizing corrosive base titration. The salt that is shaped during the hydrolyses of oil during cleanser readiness might be a reason for...
What is a delicate cleanser?
Solution: They are effectively solvent which contains potassium salts of unsaturated fats as a significant part.
The two acid neutralizers and antiallergic drugs are antihistamines yet they can’t supplant one another. Clarify why?
Solution: Stomach settling agents are utilized for the treatment of corrosive in the stomach and antihistamines repress the activity of histamine in the body. The two stomach settling agents and...
Anti-inflamatory medicine is an aggravation assuaging antipyretic medication yet can be utilized to forestall coronary failure. Clarify.
Solution: Ibuprofen forestalls blood thickening in the heart as it has against blood-coagulating activity. This activity helps in forestalling coronary failure.
Which class of medications is utilized in dozing pills?
Solution: It contains sedatives as a medication which is intended for the treatment of dread, uneasiness and mental interruptions.
What is the shared trait between the anti-toxin arsphenamine and azodye?
Solution: The kind of linkage moved by the anti-microbial arsphenamine is like that of azodye. anti-toxin arsphenamine gangs – As=As-linkage which is like – N=N-linkage in azodye.
What sort of powers are associated with restricting of substrate to the dynamic site of a catalyst?
Solution: I) Van der Waal power ii) hydrogen holding iii) dipole cooperations iv) ionic securities and so forth are engaged with the limiting of substrate.
Which site of a protein is called allosteric site?
Solution: This is the site other than the dynamic site in which the medications can tie and cause its activity. They control substance responses happening in the human body.
What is the destructive impact of hyperacidity?
Solution: Hyperacidity can cause a ulcer or gastric refluxes in the stomach. The fundamental driver of hyperacidity is the discharge of corrosive in an overabundance sum.
Where are receptors found?
Solution: Receptors are found pon the cell surface layer or inside the cytoplasm. They are organic transducers.
Which sort of medications goes under antimicrobial medications?
Solution: Mostly the antimicrobial medications are utilized to treat the microbial capacities. Models like germicides, sulpha medications and anti-toxins go under this class.
What are germ-killers?
Solution: Germicides are those which are applied to the living body to forestall the development of microorganisms. It is utilized on account of cuts or wounds.
Compose the employments of medications.
Solution: Medications play a significant part in our everyday life. It fixes sicknesses. There are different sorts of medications present as tablets, syrups, salves and so on It advances and keeps...
What is the normal atomic mass of medications?
Solution: Medications have a normal atomic mass of 100-500u.
Which of the accompanying assertions are right? (a) Cationic cleansers have germicidal properties. (b) Bacteria can corrupt the cleansers containing profoundly spread chains. (c) Some engineered cleansers can give froth even in super cold water. (d) Synthetic cleansers are not cleansers.
Solution: (a, c, d) (a) Cationic cleansers are quaternary ammonium salts of amines with acetic acid derivations, chlorides or bromides as anions. These cleansers have germicidal properties. (b)...
Which of coming up next are anionic cleansers? (a) Sodium salts of sulphonated long chain liquor. (b) Ester of stearic corrosive and polyethylene glycol. (c) Quaternary ammonium salt of amine with acetic acid derivation particle. (d) Sodium salts of sulphonated long chain hydrocarbons.
Solution: (a, d) Sodium salts of sulphonated long chain liquor and sodium salts of sulphonated long chain hydrocarbons are anionic cleansers e.g., Sodium laurylsulphate CH3(CH2)10CH2OSO3–Na+ and...
Veronal and Luminal are subsidiaries of barbituric corrosive which are (i) Tranquillizers (ii) Non-narcotic analgesic (iii) Antiallergic drugs (iv) Neurologically active drugs
Solution: (a, d) Tranquilizers are neurologically dynamic medications. Veronal and luminal are subordinates of barbituric corrosive utilized as sedatives.
Amongst the accompanying antihistamine ,which are insect acids? (a)Ranitidine (b) Brompheniramine (c)Terfenadine (d)Cimetidine
Solution: (a, d) Ranitidine and cimetidine are antihistamines which are utilized as subterranean insect acids. These medication bring about arrival of lesser measure of corrosive. Brompheniramine...
Which of the accompanying mixtures are controlled as subterranean insect acids? (a) Sodium carbonate (b)Sodium Hydrogen carbonate (c)Aluminium carbonate (d)Magnism Hydroxide
Solution: (b,d) Sodium Hydrogen carbonate and Magnism Hydroxide ,both are gentle alkalies ,are utilized as subterranean insect acids.
Which of the accompanying assertions are inaccurate with regards to penicillin? (a) An antibacterial organism. (b) Ampicillin is its engineered adjustment. (c) It has bacteriostatic impact. (d) It is a wide range anti-infection.
Solution: (c, d) Penicillin annihilates microbes by obliterating the cell mass of the microorganism or kill the microscopic organisms along these lines, it has bacteriocidal impact. Penicillin has a...
Which of coming up next are antidepressants? (a) Iproniazid (b) Phenelzine (c) Equanil (d) Salvarsan
Solution: (a, b, c) (a) Iproniazid is a hydrazine drug utilized as an upper. (b) Phenelzine is otherwise called Nardil. It is utilized in the treatment of significant burdensome problem. (c) Equanil...
Which of coming up next are sulpha drugs? (a) Sulphapyridine (b) Prontosil (c) Salvarsan (d) Nardil
Solution: (a, b) (a) Sulphapyridine is a sulphonamide antibacterial medication. (b) Prontosil is additionally called sulphamidochrysoidine. (c) Salvarsan is arsenic based antibacterial medication....
Which of the accompanying assertions are right with regards to barbiturates? (a) Hypnotics or rest creating specialists. (b) These are sedatives. (c) Non-opiate analgesics. (d) Pain diminishing without upsetting the sensory system.
Solution: (a, b) Barbiturates are sedatives which are utilized as hypnotics or rest instigating specialists.
Mixtures with clean properties are (a) CHCl, (b) CHI3 (c) Boric corrosive (d) 0.3 ppm watery arrangement of Cl2
Solution: (b, c) (a) CHCl3 (chloroform) was utilized as a sedation in medical procedure yet presently it is utilized in the creation of the Freon refrigerant R-22. (b) Iodoform (CFH3) produces...
Which of coming up next are not utilized as food additives? (a) Table salt (b) Sodium hydrogen carbonate (c) Cane sugar (d) Benzoic corrosive
Solution: (b, d) Table salt and raw sweetener are utilized, as food additives while sodium hydrogen carbonate and benzoic corrosive are not utilized as food additives.
Which of the accompanying assertions are erroneous with regards to receptor proteins? (a) Majority of receptor proteins are installed in the cell layers. (b) The dynamic site of receptor proteins opens within locale of the cell. (c) Chemical couriers are gotten at the limiting locales of receptor proteins. (d) Shape of receptor doesn’t change during connection of courier.
Solution: (b, d) Receptor proteins are implanted in the cell film and their dynamic destinations project outside area of the cell layer. State of the receptor changes during the connection of...
Which of the accompanying won’t improve dietary benefit of food? (a) Minerals (b) Artificial sugars (c) Vitamins (d) Amino acids
Solution: (b) Artificial sugars are non-caloric substitutes for sugar. They are frequently strongly more sweet than sugar however don't upgrade healthy benefit of food. Nutrients and minerals are...
Which of the accompanying synthetic can be added for improving of food things at cooking temperature and doesn’t give calories? (a) Sucrose (b) Glucose (c) Aspartame (d) Sucralose
Solution: (d) Sucralose is a fake improving specialist which is multiple times better than sucrose and doesn't give calories.
Which of the accompanying assertion isn’t correct with regards to chemical inhibitors? (a) Inhibit the synergist movement of the chemical. (b) Prevent the limiting of substrate. (c) Generally, a solid covalent bond is shaped between an inhibitor and a compound. (d) Inhibitors can be cutthroat or non-serious.
Solution: (c) Inhibitors are synthetic substances which will in general lessen the action of a specific chemical. For the most part, a powerless bond, for example, H-holding, van der Waals...
Which of coming up next isn’t an objective particle for drug work in body? (a) Carbohydrates (b) Lipids (c) Vitamins (d) Proteins
Solution: (c) Drugs generally associate with biomolecules like starches, lipids, proteins and nucleic acids. These are called drug targets. Nutrients are not an objective atom for drug work in...
Polyethyleneglycols are utilized in the readiness of which kind of cleansers? (a) Cationic cleansers . (b) Anionic cleansers (c) Non-ionic cleansers (d) Soaps
Solution: (c) Polyethyleneglycols are utilized in the planning of non-ionic cleansers.
Which of coming up next is an illustration of fluid dishwashing cleanser?
Solution: (b) Liquid dishwashing cleansers are non-ionic cleansers.
Glycerol is added to cleanser. It capacities (a) as a filler (b) to increment leathering (c) to forestall quick drying (d) to make cleanser granules
Solution: (c) Glycerol is added to shaving cleanser to forestall quick drying while to upgrade the leathering property of cleanser, a gum called rosin is added to them. It structures sodium rosinate...
Which of the accompanying improves washed property of cleanser? (a) Sodium carbonate (b) Sodium rosinate (c) Sodium stearate (d) Trisodium phosphate
Solution: (b) Shaving cleansers contain glycerol to forestall quick drying. A gum called rosin is included these cleansers which structures sodium rosinate which upgrades washed property of...
A gum rosin added to cleanser to make it foam well. Bithional is added to cleansers to bestow germ-free properties to cleanser. Equanil is (a) fake sugar (b) sedative (c) antihistamine (d) antifertility medication
Solution: (b) Equanil is a sedative utilized in controlling sadness and hypertension.
Compound which is added to cleanser to confer disinfectant properties is (a) sodium laurylsulphate (b) sodium dodecylbenzenesulphonate (c) rosin (d) bithional
Solution: (d) All cleansers are made by bubbling fats or oils with reasonable hydroxide. Varieties are made by adding distinctive natural substances. Sodium laurylsulphate and sodium...
The compound that causes general upper activity on the focal sensory system has a place with the class of (a) analgesics (b) sedatives (c) opiate analgesics (d) antihistamines
Solution: (b) The compound that causes general stimulant activity on the focal sensory system has a place with the class of sedatives.
A limited range anti-microbial is dynamic against (a) gram positive or gram negative microbes (b) gram negative microscopic organisms as it were (c) single creature or one sickness (d) both gram positive and gram negative microbes
Solution: (a) A tight range anti-microbial is dynamic against gram positive or gram negative microorganisms.
Salvarsan is arsenic containing drug which was first utilized for the treatment of (a) syphilis (b) typhoid (c) meningitis (d) loose bowels
Solution: (a) Salvarsan is arsenic containing drug which was first utilized for treatment of syphilis. Syphilis is an intense and ongoing contaminations sickness brought about by the bacterium...
Which of the accompanying assertion is right? (a) Some sedatives work by hindering the compounds which catalyze the debasement of noradrenaline. (b) Tranquilizers are opiate drugs. (c) Tranquilizers are substance intensifies that don’t influence the message move from nerve to receptor. (d) Tranquilizers are substance intensifies that can calm torment and fever.
Solution: (a) Tranquilizers are neurologically dynamic medications. A few sedatives are antidepressants and the capacities by hindering the proteins which catalyze the corruption of noradrenaline....
The most valuable order of medications for restorative scientific experts is (a) based on synthetic design (b) based on drug activity (c) based on sub-atomic targets (d) based on pharmacological impact
Solution: (c) The most valuable grouping of medications for therapeutic physicists. It is based on sub-atomic targets. Target particles are typically biomolecules like carbs, lipids, proteins and...
Which proclamation about ibuprofen isn’t correct? (a) Aspirin has a place with opiate analgesics. (b) It is powerful in diminishing torment. (c) It has antiblood thickening activity. (d) It is a neurologically dynamic medication.
Solution: (a) Aspirin restrains the combination of mixtures known as prostaglandins which invigorate irritation in the tissues and cause torment. Thus, it is viable in easing torment. Ibuprofen has...
Which is the right assertion about anti-conception medication pills? (a) Contain estrogen as it were (b) Contain progesterone as it were (c) Contain a combination of estrogen and progesterone subsidiaries (d) Progesterone upgrades ovulation
Solution: (c) Birth control pills contain a combination of estrogen and progesterone subsidiaries. Both of these are sex chemicals. Progesterone stifles ovulation and estrogen control the feminine...
Which of the accompanying assertion isn’t right? (a) Some germ-killers can be added to cleansers. (b) Dilute arrangements of certain sanitizers can be utilized as germ-free. (c) Disinfectants are antimicrobial medications. (d) Antiseptic prescriptions can be ingested.
Solution: (d) Antiseptics are applied to the living tissues like injuries, cuts and unhealthy skin surfaces. Disinfectant prescriptions, for example, anti-microbials can't be ingested.
For what reason does benzene go through electrophilic replacement responses effectively and nucleophilic replacements with trouble?
Solution: Benzene is a planar atom with electrons delocalized under or more the ring plane. Henceforth, it is a material wealthy in electrons. As an outcome, electron-inadequate species, i.e.,...
Organize benzene, n-hexane and ethyne in expanding request of their acidic conduct. Likewise, give a justification behind this conduct.
Solution: Acidic person of an animal groups is characterized based on the simplicity with which it can lose its H–iotas. The hybridization condition of carbon in the given compound is: As the s –...
Record the results of ozonolysis of 1,2-dimethylbenzene (o-xylene). How does the outcome uphold Kekulé structure for benzene?
Solution: o-xylene has two reverberation structures, which are as per the following: Each of the three items are gotten from two Kekule constructions of o-xylene, i.e., methyl glyoxal, 1,...
Expansion of HBr to propene yields 2-bromopropane, while within the sight of benzoyl peroxide, a similar response yields 1-bromopropane. Clarify and give system.
Solution: The expansion of HBr to propene is an illustration of an electrophilic replacement response. Being a corrosive, the hydrogen bromide gives an electrophile, H+. This electrophile assaults...
What impact does fanning of an alkane chain has on its limit?
Solution: Alkanes experience Van-der Waals powers between atoms. The higher the alkane's force, the more prominent is the limit. As the atom stretching expands, the surface region diminishes which...
In the alkane H3C–CH2 – C(CH3)2 – CH2 – CH(CH3)2, distinguish 1°, 2°, 3° carbon particles and give the quantity of H iotas clung to every last one of these.
Solution: Essential carbon particles (1o): Carbon iotas clung to a solitary molecule of carbon are called essential iotas of carbon. The game plan given has comparing five 1o carbon iotas and...
How might you convert benzene into acetophenone
Solution: Benzene changed over to acetophenone
How might you convert benzene into p – nitrotoluene
Solution: Benzene changed over to p – nitrotoluene
How might you convert benzene into m-nitrochlorobenzene
Solution: Benzene changed over to m-nitrochlorobenzene
How might you convert benzene into p – nitrobromobenzene
Solution: Benzene changed over to p – nitrobromobenzene
Clarify why the accompanying framework isn’t fragrant?
Solution: Cyclo-octatetraene isn't planar yet is tub-molded. It is, in this manner, a non-planar framework having 8 n-electrons. In this manner, the atom isn't sweet-smelling as it doesn't contain a...
Clarify why the accompanying framework isn’t fragrant?
Solution: Because of the presence of sp3–hybridized carbon, the framework isn't planar. Further, it contains just four n-electrons, subsequently, the framework isn't fragrant on the grounds that it...
Clarify why the accompanying framework isn’t fragrant?
Solution: Because of the presence of a sp3-hybridized carbon, the framework isn't planar. It contains six n-electrons yet the framework isn't completely formed since all the six n-electrons don't...
What are the important conditions for any framework to be sweet-smelling?
Solution: The fundamental conditions for any fragrant framework are as per the following: (I) Firstly, the compound course of action or construction ought to be planar. (ii) The n-electrons are...
For what reason is benzene exceptionally steady however it contains three twofold bonds?
Solution: Benzene is a mixture of the resounding designs and it is displayed as: Every one of the six carbon molecules in benzene are hybridized to sp2.In benzene, every carbon particle's two...
Draw the cis and trans constructions of hex-2-ene. Which isomer will have higher b.p. what’s more, why?
Solution: Hex-2-ene is addressed as displayed beneath: Mathematical isomers of hex-2-ene are as per the following: A cis intensifies dipole second is equivalent to the amount of the C – CH3...
Compose synthetic conditions for the burning response of Toluene
Solution: Burning responses might be characterized as an oxygen or oxygen response of a compound.
Compose synthetic conditions for the burning response of Hexyne
Solution: Burning responses might be characterized as an oxygen or oxygen response of a compound.
Compose synthetic conditions for the burning response of Pentene
Solution: Burning responses might be characterized as an oxygen or oxygen response of a compound.
Compose synthetic conditions for the burning response of Butane
Solution: Burning responses might be characterized as an oxygen or oxygen response of a compound.
Propanal and pentan-3-one are the ozonolysis results of an alkene? What is the primary equation of the alkene?
Solution: From the given data, the two ozonolysis results of an alkene are pentan-3-one and propanal. Assume the alkene given is A. The converse of the ozonolysis response is the thing that we get,...
An alkene ‘A’ contains three C – C, eight C – H σ bonds and one C – C π bond. ‘A’ on ozonolysis gives two moles of an aldehyde of molar mass 44 u. Compose IUPAC name of ‘A’.
Solution: From the data given it gives two moles of an aldehyde of molar mass 44 u when ' A ' goes through ozonolysis. The arrangement of an aldehyde's two moles recommends that the presence of...
An alkene ‘A’ on ozonolysis gives a combination of ethanal and pentan-3-one. Compose construction and IUPAC name of ‘A’.
Throughout ozonolysis, an ozonide is created as a halfway that has a cyclic construction; it goes through cleavage to give the eventual outcomes. Ethanal and pentan-3-one are acquired from the...
Compose IUPAC names of the items acquired by the ozonolysis 1 – Phenylbut-1-ene
Solution: Ozonolysis of 1 – Phenylbut-1-ene is displayed as The resulting IUPAC names of the items are : Item (I) benzaldehyde, and Item (II) propanal
Compose IUPAC names of the items acquired by the ozonolysis 2-Ethylbut-1-ene
Solution: Ozonolysis of 2-Ethylbut-1-ene is displayed as: The resulting IUPAC names of the items are: Item (I) pentan-3-one, and Item (II) methanal
Compose IUPAC names of the items acquired by the ozonolysis 3, 4 – Dimethyl-hept-3-ene
Solution: Ozonolysis of 3, 4-Dimethylhept-3-ene is displayed as: The item names resulting to IUPAC are: Item (I) butan-2-one, and Item (II) Pentan-2-one
Compose IUPAC names of the items acquired by the ozonolysis Pent-2-ene
Solution: Ozonolysis of Pent-2-ene is displayed as: The accompanying Product IUPAC names are: Item (I) ethanal, and Item (II) propanal.
compose primary equations and IUPAC names for all potential isomers having the quantity of the twofold or triple bond as demonstrated C5H8 (one triple bond)
Solution: The resulting primary isomers are likely for C5H8 with one triple bond: The IUPAC name of Compound (I) is Pent-1-yne, Compound (II) is Pent-2-yne, and Compound (III) is...
compose primary equations and IUPAC names for all potential isomers having the quantity of the twofold or triple bond as demonstrated C4H8 (one twofold bond)
Solution: The subsequent primary isomers with one twofold bond are plausible for C4H8: The IUPAC name of Compound (I) is But-1-ene, Compound (II) is But-2-ene, and Compound (III) is...
Write the IUPAC names of
Solution: 4-Ethyldeca-1, 5, 8-triene is the necessary IUPAC name
Write the IUPAC names of
Solution: 5-(2-Methylpropyl)- decane is the necessary IUPAC name
Write the IUPAC name of
Solution: 2-Methyl phenol is the necessary IUPAC name
Write the IUPAC name of
Solution: 4-Phenyl but-1-ene is the necessary IUPAC name
Write the IUPAC name of
Solution: 1, 3-Butadiene or Buta-1,3-diene is the necessary IUPAC name
Write the IUPAC names of
Solution: Pen-1-ene-3-yne is the necessary IUPAC name
Write the IUPAC nameof ![Rendered by QuickLaTeX.com \[\mathbf{C}{{\mathbf{H}}_{\mathbf{3}}}~\mathbf{CH}\text{ }=\text{ }\mathbf{C}\text{ }{{(\mathbf{C}{{\mathbf{H}}_{\mathbf{3}}})}_{\mathbf{2}}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c63b9b35d0c7e5500238909182cb534b_l3.png)
Solution: 2-Methylbut-2-ene is the necessary IUPAC name
How would you represent the arrangement of ethane during chlorination of methane?
Solution: The methane chlorination measure works through a free extreme chain instrument. Stage 1: Initiation: The outcome starts with the hemolytic cleavage of Cl – Cl bond as: Stage 2:...
Think about the components: Cs, Ne, I and F Identify the component that displays neither negative nor positive oxidation state?
Solution: Ne displays neither negative nor positive oxidation no. That is 0.
Think about the components: Cs, Ne, I and F Identify the component that displays both negative and positive oxidation states.
Solution: I displays both negative and positive oxidation no. That is - 1, +1, +3, +5 and +7.
Consider the elements: Cs, Ne, I and F Identify the element that exhibits only positive oxidation.
Solution: Cs displays just certain oxidation no. That is +1.
Think about the components: Cs, Ne, I and F Identify the component that displays just adverse oxidation.
Solution: F exhibits only negative oxidation number. That is -1.
Assign oxidation number to the underlined elements 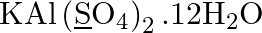
Solution: $\mathrm{KAl}\left(\underline{\mathrm{S}} \mathrm{O}_{4}\right)_{2} .12 \mathrm{H}_{2} \mathrm{O}$ Let expect oxidation number of S is x. \[\begin{array}{*{35}{l}}...
Assign the oxidation number to the underlined component 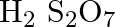
Solution: $\mathrm{H}_{2} \mathrm{~S}_{2} \mathrm{O}_{7}$ Let accept oxidation number of S is x. \[\begin{array}{*{35}{l}} Oxidation\text{ }number\text{ }of\text{ }O\text{ }=\text{...
Assign the oxidation number to the underlined component NaBH4
Solution: NaBH4 Let accept oxidation number of B is x. \[\begin{array}{*{35}{l}} Oxidation\text{ }number\text{ }of\text{ }Na\text{ }=\text{ }+1 \\ ~ \\ Oxidation\text{ }number\text{...
Assign the oxidation number to the underlined component CaO2
Solution: CaO2 Let expect oxidation number of O is x. \[Oxidation\text{ }number\text{ }of\text{ }Ca\text{ }=\text{ }+2\] Then, at that point, we have: \[\begin{array}{*{35}{l}}...
Assign the oxidation number to the underlined component K2MnO4
Solution: K2MnO4 Let expect oxidation number of Mn is x. \[\begin{array}{*{35}{l}} Oxidation\text{ }number\text{ }of\text{ }K\text{ }=\text{ }+1 \\ ~ \\ Oxidation\text{ }number\text{...
Assign the oxidation number to the underlined component H4P2O7
Solution: H4P2O7 Let expect oxidation number of P is x. \[\begin{array}{*{35}{l}} Oxidation\text{ }number\text{ }of\text{ }H\text{ }=\text{ }+1 \\ ~ \\ Oxidation\text{ }number\text{...
Assign the oxidation number to the underlined component NaHSO4
Solution: NaHSO4 Let expect oxidation number of S is x. \[\begin{array}{*{35}{l}} Oxidation\text{ }number\text{ }of\text{ }Na\text{ }=\text{ }+1 \\ ~ \\ Oxidation\text{ }number\text{...
Assign the oxidation number to the underlined component NaH2PO4
Solution: NaH2PO4 Let expect oxidation number of P is x. We realize that, \[\begin{array}{*{35}{l}} Oxidation\text{ }number\text{ }of\text{ }Na\text{ }=\text{ }+1 \\ ~ \\...
Thermodynamically the most steady type of carbon is (a) Diamond (b) Graphite (c) Fullerenes (d) Coal
Solution: (b) Graphite is thermodynamically the most steady type of carbon.
The kind of hybridization of boron in diborane is (a) sp (b)sp2 (c) sp3 (d) dsp2
Solution: (c) Boron in diborane is sp3 hybridized.
A watery arrangement of borax is (a) Neutral (b) Amphoteric (c) Basic (d) Acidic
Solution: (b) Borax is a strong base salt (NaOH) and a frail corrosive (H3BO3). Accordingly, it is an essential thing in nature.
What do you comprehend by Catenation?
Solution: Catenation A few components or particles (like carbon) may interface with one another through solid covalent bonds to shape long chains or branches. This trademark is known as catenation....
What do you comprehend by Allotropy
Solution: Allotropy Allotropy is the presence of a component in more than one structure, having distinctive actual properties yet similar synthetic properties. The various types of a component are...
What do you comprehend by Inert pair impact
Solution: Inert pair impact As one actions down the gathering, s-block electrons decline their propensity to take part in substance holding. This impact is known as the dormant pair impact. The...
In a portion of the responses, thallium takes after aluminum, while in others it looks like with the gathering I metals. Backing this assertion by giving some proof.
Solution: Thallium is a piece of the intermittent table gathering 13. For this gathering, the most widely recognized oxidation state is + 3. Heavier individuals from this gathering, nonetheless,...
Clarify why would that be an incredible decline in ionization enthalpy from carbon to silicon?
Solution: Carbon ionizing enthalpy (the principal component in bunch 14) is exceptionally high (1086 kJ/mol). That is normal due to its little size. Nonetheless, there is a sharp decline in enthalpy...
Give reasons: Aluminium wire is utilized to make transmission links.
Solution: Aluminum wire is utilized to make transmission links. Aluminium wires are good conductors of electricity and also it is cheap metal easily available, light in weight and also very ductile....
Give reasons: Aluminum utensils ought not be kept in water for the time being.
Solution: Aluminum utensils ought not be kept in water for the time being. The oxygen in water responds to make a dainty layer of aluminum oxide with aluminum. This layer keeps further response from...
Give reasons: Aluminum compounds are utilized to make airplane body.
Solution: Aluminum compounds are utilized to make airplane body. Aluminum has a high protection from elastic and is light. It might likewise be alloyed to various metals like Si, Mg, Cu, Mn and Zn....
Give reasons: Diamond is utilized as a rough
Solution: Diamond is utilized as a rough Carbon is sp3 hybridized in Diamond. With the assistance of solid covalent bonds, every carbon molecule is bound to four other carbon iotas. These covalent...
Give reasons: Graphite is utilized as the grease.
Solution: Graphite is utilized as the grease. Graphite has a layered construction, and the powers of frail van der Waals tie various layers of graphite together. These layers might slide one over...
Give reasons: A combination of weaken NaOH and aluminum pieces is utilized to open channel.
Solution: A combination of weaken NaOH and aluminum pieces is utilized to open the channel. Sodium hydroxide and aluminum respond to shape aluminate (III) sodium tetra hydroxy and hydrogen gas....
Give reasons: Conc.HNO3 can be moved in an aluminum holder.
Solution: Conc.HNO3 can be shipped in an aluminum holder. As it responds with aluminum to frame a slight defensive oxide layer on the aluminum surface, concentrated HNO3 can be put away and shipped...
How is the exorbitant substance of CO2 answerable for a dangerous atmospheric devation?
Solution: Carbon dioxide is a gas that is vital for our endurance. The expanded CO2 content in the climate, nonetheless, represents a genuine danger. An increment in petroleum product ignition,...
Recommend an explanation with regards to why CO is toxic.
Solution: Given its capacity to frame a complex with hemoglobin, carbon monoxide is profoundly toxic. The previous hinders restricting with oxygen by Hb. Subsequently, an individual passes on...
Give a short portrayal of the standards of the accompanying procedures taking a model. Chromatography
Solution: Chromatography It is generally utilized for the partition and cleansing of natural mixtures. Rule: The guideline on which it works is that singular parts of a blend move at various speeds...
Give a short portrayal of the standards of the accompanying procedures taking a model. Distillation
Solution: Distillation This strategy is utilized to isolate non-unpredictable fluids from unstable pollutants. It is likewise utilized when the parts have an extensive distinction in their edges of...
Give a short portrayal of the standards of the accompanying procedures taking a model. Crystallization
Solution: Crystallization Crystallization is utilized to decontaminate strong natural mixtures. Standard: The guideline on which it works is the distinction in the dissolvability of the compound and...
Classify the following reactions in one of the reaction type studied in this unit. 
Solution: Substitution (nucleophilic) response since modification of particles happens.
Classify the following reactions in one of the reaction type studied in this unit. 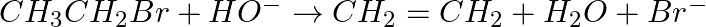
Solution: Elimination (bimolecular) response since response hydrogen and bromine are taken out to shape ethene.
Classify the following reactions in one of the reaction type studied in this unit.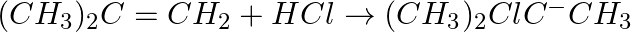
Solution: Addition (electrophilic) response since two reactant particles joins to shape a solitary item.
Classify the following reactions in one of the reaction type studied in this unit.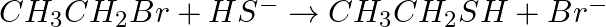
Solution: Substitution (nucleophilic) response since bromine bunch gets subbed by – SH bunch.
Identify the reagents shown in bold in the following equations as nucleophiles or electrophiles: 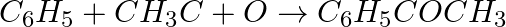
Solution: A nucleophile is a reagent that has an electron pair and will give it away. It is otherwise called a core adoring reagent. An electrophile is a reagent which needs an electron pair and is...
Identify the reagents shown in bold in the following equations as nucleophiles or electrophiles: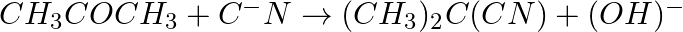
Solution: A nucleophile is a reagent that has an electron pair and will give it away. It is otherwise called a core adoring reagent. An electrophile is a reagent which needs an electron pair and is...
Identify the reagents shown in bold in the following equations as nucleophiles or electrophiles: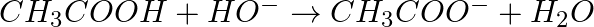
Solution: A nucleophile is a reagent that has an electron pair and will give it away. It is otherwise called a core adoring reagent. An electrophile is a reagent which needs an electron pair and is...
What are electrophiles and nucleophiles? Clarify with models.
Solution: A nucleophile is a reagent that has an electron pair and will give it away. It is otherwise called a core cherishing reagent. Ex: NC–, OH–, R3C–(carbanions), and so on An electrophile is a...
Which of the two: O2NCH2CH2O–or CH3CH2O–is relied upon to be more steady and why?
Solution: Since NO2 has a place with the electron-pulling out bunch, it shows – I impact. NO2 attempts to diminish the negative charge on the compound by pulling out the electrons toward it. This...
Draw recipes for the initial five individuals from each homologous series starting with the accompanying mixture H–CH=CH2
Solution: The initial five individuals from each homologous series starting with the given mixtures are H–CH=CH2: Ethene CH3–CH=CH2: Propene CH3–CH2–CH=CH2: 1-Butene CH3–CH2–CH2–CH=CH2: 1-Pentene...
Draw recipes for the initial five individuals from each homologous series starting with the accompanying mixture CH3COCH3
Solution: CH3COCH3: Propanone CH3COCH2CH3: Butanone CH3COCH2CH2CH3 : Pentan-2-one CH3COCH2CH2CH2CH3: Hexan-2-one CH3COCH2CH2CH2CH2CH3 : Heptan-2-one
Draw recipes for the initial five individuals from each homologous series starting with the accompanying mixture H–COOH
Solution: The initial five individuals from each homologous series starting with the given mixtures are H–COOH: Methanoic corrosive CH3–COOH: Ethanoic corrosive CH3–CH2–COOH: Propanoic corrosive...
Which of the accompanying addresses the right IUPAC name for the mixtures concerned? But-3-yn-1-ol or But-4-ol-1-yne
Solution: Out of the two utilitarian gatherings present in the given compound, the alcoholic gathering is the central practical gathering. Consequently, the parent chain will have an – ol postfix....
Which of the accompanying addresses the right IUPAC name for the mixtures concerned? 2-Chloro-4-methylpentane or 4-Chloro-2-methylpentane
Solution: If the substituents in the chain are in identical positions, then, at that point, the lower number is given to the substituent bunch in sequential request. Along these lines, the right...
Which of the accompanying addresses the right IUPAC name for the mixtures concerned? 2,4,7-Trimethyloctane or 2,5,7-Trimethyloctane
Solution: The locant number should begin from the base. Here, 2,4,7 is lower than 2,5,7. Subsequently, the right IUPAC name would be 2,4,7-Trimethyloctane.
Which of the accompanying addresses the right IUPAC name for the mixtures concerned? 2,2-Dimethylpentane or 2-Dimethylpentane
solution: The prefix di shows that there are two methyl bunches in the chain. Along these lines, the right IUPAC name would be 2,2-Dimethylpentane.
What are the hybridisation conditions of every carbon molecule in the accompanying compound? ![Rendered by QuickLaTeX.com \[C6H6\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-250e7c087123a8d0267c095925b18800_l3.png)
Solution: C6H6 All the 6 carbon particles in benzene are sp2 hybridized
What are the hybridisation conditions of every carbon molecule in the accompanying compound? ![Rendered by QuickLaTeX.com \[CH2=CHCN\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-52ef84ac98a604743dfb23732dfbb165_l3.png)
Solution: C-1 is sp2 hybridized. C-2 is sp2 hybridized. C-3 is sp hybridized.
What are the hybridisation conditions of every carbon molecule in the accompanying compound? ![Rendered by QuickLaTeX.com \[\left( CH3 \right)2CO\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a2bea77220b7e99546204fa04a862cfa_l3.png)
Solution: C - 1 and C-3 are sp3 hybridized. C-2 is sp2 hybridized.
What are the hybridisation conditions of every carbon molecule in the accompanying compound? ![Rendered by QuickLaTeX.com \[CH3CH=CH2\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-6a2ef16862caa7e37fd3861c7df53de4_l3.png)
Solution: C-1 is sp3 hybridized. C-2 is sp2 hybridized. C-3 is sp2 hybridized.
What are the hybridisation conditions of every carbon molecule in the accompanying compound? ![Rendered by QuickLaTeX.com \[CH2=C=O\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-3ffdae7fcb2301f4db1b66d768c446ab_l3.png)
Solution: C-1 is sp2 hybridized. C-2 is sp hybridized.
How does H2 O2 act as a dying specialist?
Solution: Hydrogen peroxide goes about as a solid oxidizing specialist both in fundamental and acidic media. When added to a material, it breaks the substance obligations of the chromophores...
Knowing the properties of H2O and D2O, do you imagine that D2O can be utilized for drinking purposes?
Solution: D2O is referred to as weighty water which goes about as an arbitrator (dials back the pace of response). Because of this property, it can't be utilized for drinking reason since it dials...
Portray the helpfulness of water in biosphere and organic frameworks.
Solution: Water is extremely important for all types of life which establish 65% of human body and 95% of plants.it assumes an indispensable part in the biosphere because of its → Thermal...
Is demineralised or refined water valuable for drinking purposes? If not, how might it be made valuable?
Solution: Water is extremely fundamental for our life. It comprises of many broke down supplements that are needed for ourselves and furthermore for plants and creatures. Demineralised water is...
What is implied by ‘demineralised’ water and how might it be acquired ?
Solution: This water is liberated from every one of the solvent mineral salts and it doesn't contain any cation or anion. It is acquired progressively by going the water through anion trade and...
What causes the impermanent and super durable hardness of water?
Solution: Because of the presence of dissolvable salts of magnesium and calcium as chlorides in water, hardness stays super durable in water. Because of the presence of dissolvable salts of calcium...
Portray the construction of the normal type of ice.
Solution: For the most part, ice is the translucent type of water. It visibles in a hexagonal structure in case it is solidified at climatic strain. At the point when the temperature is...
Analyze the designs of H2O and H2 O2.
Solution: The water atom will be shown with a bond point of 104 .5o has a bowed structure in vaporous stage. The O-H bond length is 95.7 pm. Structure : Hydrogen peroxide has a non-planar design...
Organize the accompanying NaH, MgH2 and H2O arranged by expanding diminishing property.
Solution: Ionic hydrides are solid diminishing specialists. NaH can undoubtedly give its electrons. Henceforth, it is most diminishing in nature. Both, MgH2 and H2O are covalent hydrides. H2O is...
Organize the accompanying H–H, D–D and F–F arranged by expanding bond separation enthalpy.
Solution: The bond pair in D–D bond is more emphatically drawn in by the core than the bond pair in H–H bond. This is a direct result of the greater atomic mass of D2. The more grounded the...
Organize the accompanying LiH, NaH and CsH arranged by expanding ionic person.
Solution: The ionic person of a bond is reliant upon the electro negativities of the molecules in question. The higher the distinction between the electro negativities of molecules, the more modest...
Organize the accompanying CaH2, BeH2 and TiH2 arranged by expanding electrical conductance.
Solution: The electrical conductance of an atom chiefly relies upon its covalent or ionic nature. CaH2 is an ionic hydride, which conducts power in the liquid state. Titanium hydride, TiH2 is...
Among NH3, H2O and HF, which would you hope to have most noteworthy greatness of hydrogen holding and why?
Solution: The degree of hydrogen holding predominantly relies upon (I) Electronegativity (ii) Number of hydrogen iotas accessible for holding. Among oxygen, fluorine and nitrogen, the expanding...
How does the nuclear hydrogen or oxy-hydrogen light capacity for cutting and welding purposes ? Clarify.
Solution: The nuclear hydrogen light is otherwise called oxy-hydrogen light. These molecules are delivered through dihydrogen separation with the assistance of an electric circular segment which...
How would you anticipate that the metallic hydrides should be valuable for hydrogen stockpiling? Clarify
Solution: Metallic hydrides are hydrogen inadequate. They don't keep the law of consistent structure. It has been set up that in the hydrides of Pd, Ac, Ni, and Ce, hydrogen possesses the...
What do you comprehend by the expression “non-stoichiometric hydrides”? Do you anticipate that this type of the hydrides should be framed by soluble base metals? Legitimize your reply.
Solution: Non-Stoichiometric hydrides are hydrogen-insufficient mixtures which is framed by the response of dihydrogen with d-square and f-block components. These hydrides don't observe the law of...
Do you expect the carbon hydrides of the sort (Cn H2n+2 ) to go about as ‘Lewis’ base or corrosive? Legitimize.
Solution: For carbon hydrides which have a place with type (Cn H2n+2), the accompanying hydrides are feasible for \[\begin{array}{*{35}{l}} n\text{ }=\text{ }1\Rightarrow CH4 \\ ~ \\...
What do you comprehend by (I) electron rich – mixtures of hydrogen, (ii) electron-exact, and (iii) electron-lacking? Give avocation appropriate models.
Solution: Sub-atomic hydride is grouped based on the presence of the bonds and absolute number of electrons in their Lewis structures as: Electron-inadequate hydrides Electron-exact hydrides...
Talk about the results of high enthalpy of H–H bond as far as synthetic reactivity of dihydrogen
Solution: The ionization enthalpy of H–H bond is higher (1312 kJ mol–1 ) which shows that hydrogen has a low propensity to frame H+ particles. Its ionization enthalpy esteem is equivalent to that of...
For what reason does hydrogen happen in a diatomic structure instead of in a monoatomic structure under ordinary conditions?
Solution: The ionization enthalpy of hydrogen molecule is higher. In this manner, it is more enthusiastically to eliminate its electron. This outcomes its propensity to exist in the low monoatomic...
Legitimize the situation of hydrogen in the intermittent table based on its electronic design.
Solution: The first component in the occasional table is hydrogen. Hydrogen shows double conduct since it has just 1 electron on its one 'S' shell.(i.e.,) hydrogen takes after the two incandescent...
Work out the entropy change in environmental elements when 1.00 mol of H2O(l) is shaped under standard conditions. ![Rendered by QuickLaTeX.com \[f\text{ }H0=\text{ }\text{ }286\text{ }kJ\text{ }mol1.\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-0876268cab5e7e19237ca8d099603274_l3.png)
Solution: It is given that 286 kJ mol–1 of warmth is advanced on the development of 1 mol of H2O(l). Accordingly, an equivalent measure of warmth will be consumed by the environmental elements....
Comment on the thermodynamic stability of NO(g), given
Solution: The positive worth of ∆rH demonstrates that warmth is assimilated during the development of NO(g). This implies that NO(g) has higher energy than the reactants (N2 and O2)....
The harmony steady for a response is 10. What will be the worth of ![Rendered by QuickLaTeX.com \[G0?\text{ }R\text{ }=\text{ }8.314\text{ }JK1\text{ }mol1,\text{ }T\text{ }=\text{ }300\text{ }K.\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c5355a31e6d1d5f6327e806c31f733f0_l3.png)
Solution: From the articulation, \[\begin{array}{*{35}{l}} G\theta \text{ }=\text{ }\text{ }2.303\text{ }RT\text{ }logKeq \\ ~ \\ G\theta \text{ }for\text{ }the\text{ }response,\text{ }=\text{...
For the response ![Rendered by QuickLaTeX.com \[\begin{array}{*{35}{l}} 2A\left( g \right)\text{ }+\text{ }B\left( g \right)\text{ }\to \text{ }2D\left( g \right) \\ ~ \\ U\theta \text{ }=\text{ }\text{ }10.5\text{ }kJ\text{ }and\text{ }S\theta \text{ }=\text{ }\text{ }44.1\text{ }JK1\text{ }. \\ \end{array}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-cbc82328853f02bdc578b87abcbd9b11_l3.png)
Compute ∆Gθ for the response, and anticipate whether the response might happen suddenly.
![Rendered by QuickLaTeX.com \[\begin{array}{*{35}{l}} 2A\left( g \right)\text{ }+\text{ }B\left( g \right)\text{ }\to \text{ }2D\left( g \right) \\ ~ \\ U\theta \text{ }=\text{ }\text{ }10.5\text{ }kJ\text{ }and\text{ }S\theta \text{ }=\text{ }\text{ }44.1\text{ }JK1\text{ }. \\ \end{array}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-cbc82328853f02bdc578b87abcbd9b11_l3.png)
Solution: For the given response, \[\begin{array}{*{35}{l}} 2\text{ }A\left( g \right)\text{ }+\text{ }B\left( g \right)\text{ }\to \text{ }2D\left( g \right) \\ ~ \\ ng\text{ }=\text{...
For the response, 2Cl(g) → Cl2(g), what are the indications of ∆H and ∆S ?
Solution: ∆H and ∆S are negative The given response addresses the development of chlorine particle from chlorine molecules. Here, bond development is occurring. Along these lines, energy is being...
For the response at 298 K, ![Rendered by QuickLaTeX.com \[\begin{array}{*{35}{l}} 2A\text{ }+\text{ }B\text{ }\to \text{ }C\text{ }H\text{ }=\text{ }400\text{ }kJ\text{ }mol1 \\ ~ \\ what's\text{ }more,\text{ }S\text{ }=\text{ }0.2\text{ }kJ\text{ }K1\text{ }mol1 \\ \end{array}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-dda209e4073843cae616fbc6002155e5_l3.png)
At what temperature will the response become unconstrained believing ∆H and ∆S to be consistent over the temperature range?
![Rendered by QuickLaTeX.com \[\begin{array}{*{35}{l}} 2A\text{ }+\text{ }B\text{ }\to \text{ }C\text{ }H\text{ }=\text{ }400\text{ }kJ\text{ }mol1 \\ ~ \\ what's\text{ }more,\text{ }S\text{ }=\text{ }0.2\text{ }kJ\text{ }K1\text{ }mol1 \\ \end{array}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-dda209e4073843cae616fbc6002155e5_l3.png)
Solution: From the articulation, \[G\text{ }=\text{ }H\text{ }\text{ }TS\] Expecting the response at balance, ∆T for the response would be: \[\begin{array}{*{35}{l}} \left( G\text{...
