Across a period in the lanthanoid series, there is gradual increase in the atomic number which increases by one. With the increase in atomic number, the number of protons and electrons present in...
Name the oxometal anions of the first series of the transition metals in which the metal exhibits the oxidation state equal to its group number.
The name of the oxometal anions are as follows: (i) Chromate, CrO42- Oxidation state of Cr is + 6. (ii) Permanganate, MnO4- Oxidation state of Mn is + 7. (iii) Vanadate, VO3- Oxidation...
What may be the stable oxidation state of the transition element with the following d electron configurations in the ground state of their atoms: 3d3, 3d5, 3d8 and 3d4?
To what extent do the electronic configurations decide the stability of oxidation states in the first series of the transition elements? Illustrate your answer with examples.
Mn exhibits maximum oxidation states from +2 to +7 and it lies in the first half of the transition series. With an increase in atomic number of the elements, the stability of +2 oxidation state...
Explain briefly how +2 state becomes more and more stable in the first half of the first row transition elements with increasing atomic number?
In the given table, we can find the oxidation numbers of the elements in the first half of the first row of transition elements with increasing atomic number: Here, we can find that except Sc all...
Why are Mn2+ compounds more stable than Fe towards oxidation to their +3 state?
The electronic configuration of Fe2+ and Mn2+ ions are as follows: Fe2+ is [Ar]18 3d6. Mn2+ is [Ar]18 3d5. As it is known the half and completely filled orbitals have more...
Give the IUPAC names of the following compounds:
(i) Cl CH2 C ≡ C CH2 Br (iv) ( CCl 3) 3 CCl (i) 1 – Bromo – 4 – chlorobut – 2 – yne (ii) 2−(Trichloromethyl)−1,1,1,2,3,3,3−heptachloropropane
Give the IUPAC names of the following compounds:
(i) CH3 CH(Cl) CH(Br) CH3 (ii) CH F2 CBr ClF Solution: (i) 2−Bromo−3−chlorobutane (ii) 1−Bromo−1−chloro−1, 2, 2−trifluoroethane
Which one of the following has the highest dipole moment?
(i) CH2Cl2 (ii) CHCl3 (iii) CCl4 Solution: In CHCl3, the resultant of dipole moments of two C – Cl bonds is countered by the resultant of dipole moments of one CH bond and one C – Cl bond, as seen...
Write the structures of the following organic halogen compounds.
(i) 1 – Bromo – 4 – sec – butyl – 2 – methylbenzene (ii) 1 ,4 – Dibromobut – 2 – ene Solution: (i) (ii)
Write down the electronic configuration of:
The electronic configuration of the given elements in their respective oxidation states are: (i) Mn2+: 1s2 2s2 2p6 3s2 3p6 3d5 Or, [Ar]18 3d5 (ii) Th4+:...
Write down the electronic configuration of: (i) Co2+ (ii) Lu2+
The electronic configuration of the given elements in their respective oxidation states are: (i) Co2+: 1s2 2s2 2p6 3s2 3p6 3d7 Or, [Ar]18 3d7 (ii) Lu2+:...
Write the structures of the following organic halogen compounds.
(i) 2 – Bromobutane (ii) 4 – tert – Butyl -3 –iodoheptane Solution: (i) (ii)
Write down the electronic configuration of:
The electronic configuration of the given elements in their respective oxidation states are: (i) Cu+: 1s2 2s2 2p6 3s2 3p6 3d10 Or, [Ar]18 3d10 (ii) Ce+4 :...
Write the structures of the following organic halogen compounds.
(i) 1 -Chloro-4-ethylcyclohexane (ii) 2 – (2 -Chlorophenyl) -1 –iodooctane Solution: (i) (ii)
Write down the electronic configuration of:
(i) Cr+3 (ii) Pm+3 The electronic configuration of the given elements in their respective oxidation states are: (i) Cr+3 : 1s2 2s2 2p6 3s2 3p6 3d3 Or, [Ar]18 3d3 (ii)...
Write the structures of the following organic halogen compounds.
(i) 2 -Chloro-3 -methylpentane (ii) p -Bromochlorobenzene Solution: (i) (ii)
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl ( primary, secondary, tertiary ), vinyl, or aryl halides.
(i) m-ClCH2C6H4CH2C(CH3)3 (ii) o-Br-C6H4CH(CH3)CH2CH3 Solution: (i) 1 – Chloromethyl – 3 − ( 2, 2 – dimethylpropyl ) benzene (Primary benzyl halide) (ii) 1 − Bromo − 2 – ( 1 − methylpropyl ) benzene...
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl ( primary, secondary, tertiary ), vinyl, or aryl halides
(i) CH3C(Cl)(C2H5)CH2CH3 (ii) CH3CH=C(Cl)CH2CH(CH3)2 Solution: (i) 3−Chloro−3–methylpentane (Tertiary alkyl halide) (ii) 3−Chloro−5−methylhex−2−ene (Vinyl halide)
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl ( primary, secondary, tertiary ), vinyl, or aryl halides
(i) CH3CH(CH3)CH(Br)CH3 (ii) CH3C(C2H5)2CH2Br Solution: (i) 2 − Bromo – 3 – methylbutane (Secondary alkyl halide) (ii) 1 − Bromo − 2 − ethyl − 2 – methylbutane (Primary alkyl...
Write structures of the compounds whose IUPAC names are as follows:
(i) Cyclopent – 3 – en – 1 – ol (ii) 4-Chloro-3-ethylbutan-1-ol. Solution: (i) (ii)
Write structures of the compounds whose IUPAC names are as follows:
(i) Cyclohexylmethanol (ii) 3 – Cyclohexylpentan – 3 – ol Solution: (i) (ii)
Write structures of the compounds whose IUPAC names are as follows:
(i) 1 – Ethoxypropane (ii) 2 – Ethoxy – 3 – methylpentane Solution: (i) structure of 1 – Ethoxypropane : (ii) Structure of 2 – Ethoxy – 3 – methylpentane...
Write structures of the compounds whose IUPAC names are as follows:
(i) 3 , 5 – Dimethylhexane – 1 , 3 , 5 – triol (ii) 2, 3 – Diethylphenol Solution: (i) (ii)
Write structures of the compounds whose IUPAC names are as follows:
(i) 2 – Methylbutan – 2 – ol (ii) 1–Phenylpropan–2–ol Solution: (i) (ii)
Give a mechanism for this reaction. (Hint: The secondary carbocation formed in step II rearranges to a more stable tertiary carbocation by a hydride ion shift from 3rd carbon atom)
Solution: The following procedure is involved in the above-mentioned steps of the reaction: Step 1: Protonation Step 2: Forming 2 ° carbonation by eliminating the water molecule Step 3: Rearranging...
Show how would you synthesize the following alcohols from appropriate alkenes?
(i) (ii) Solution: All the alcohols are formed by the hydration of alkenes in the acidic medium. The addition follows Markownikov’s rule. (i) 1-Methylcyclohexene can be used in the reaction. (ii)...
Write equations of the following reactions:(i) Bromination of anisole in ethanoic acid medium (ii) Friedel-Craft’s acetylation of anisole.
(i) (ii)
Write equations of the following reactions: (i) Friedel-Crafts reaction – alkylation of anisole.(ii) Nitration of anisole.
(i) (ii)
Write the mechanism of the reaction of HI with methoxymethane.
Step 1: Protonation of methoxymethane : Step 2: Nucleophilic attack of I – : Step 3: The methanol produced in the previous step is used to combine with another HI molecule, resulting in the...
Explain the fact that in aryl alkyl ethers.
(i) the alkoxy group activates the benzene ring towards electrophilic substitution and (ii) it directs the incoming substituents to ortho and para positions in the benzene ring. (i) The presence of...
Write the equation of the reaction of hydrogen iodide with:
(i) 1-propoxypropane (ii) methoxybenzene (iii) benzyl ethyl ether
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
The production of ethers with alcohol dehydration is a bimolecular process ( SN 2) that involves an alcohol molecule attacking a protonated alcohol molecule. The alkyl group should be free in the...
How is 1-propoxypropane synthesised from propan-1-ol? Write the mechanism of this reaction.
The process of dehydration is used to synthesize 1- propoxypropane from propan –1– ol. In the presence of protic acids (such as H2SO4, H3PO4), propan-1-ol dehydrates to yield 1 – propoxypropane....
How is 1-propoxypropane synthesized from propan-1-ol? Write the mechanism of this reaction.
Dehydration reaction can be used to synthesise 1- propoxypropane from propan –1– ol. In the presence of protic acids (such as H2SO4, H3PO4), propan-1-ol dehydrates to yield 1 –...
Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
Williamson's synthesis is a flexible approach for making symmetrical and unsymmetrical ethers. However, careful reactant selection is required for the synthesis of unsymmetrical ethers. Because the...
Write the names of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
(i) 2-Methoxy-2-methylpropane (ii) 1-Methoxyethane
Write the names of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
(i) 1-Propoxypropane (ii) Ethoxybenzene
Give IUPAC names of the following ethers:
1 -Ethoxy-4 -4 – dimethyl cyclohexane Ethoxybenzene
Give IUPAC names of the following ethers:
(i) 4-Nitroanisole(ii) 1-Methoxypropane
Give IUPAC names of the following ethers:
(i)1-Ethoxy-2-methylpropane(ii) 2-Chlorlo-l-methoxy ethane
Give a reason for the higher boiling point of ethanol in comparison to methoxymethane.
Because of the presence of the – OH group in ethanol, intermolecular H – bonding occurs, resulting in molecule interaction. To break these hydrogen bonds, more energy is required. Methoxymethane, on...
Give a reason for the higher boiling point of ethanol in comparison to methoxymethane.
Because of the presence of the –OH group in ethanol, intermolecular H–bonding occurs, resulting in molecule interaction. To break these hydrogen bonds, more energy is required. Methoxymethane, on...
Question 12.1: What is meant by the following terms? Give an example of the reaction in each case. (iii) Semicarbazone (iv) Aldol
Solution: (iii) Semicabarbazone: Semicarbazone is made up of aldehydes and ketones, and it is generated when a ketone or aldehyde reacts with semicarbazide. The reaction type is condensation....
Question 12.1: What is meant by the following terms? Give an example of the reaction in each case. (ii) Acetal
Acetal refers to gemdialkoxy alkanes with two groups of alkoxy at the terminal carbon atom. The alkyl group is...
15.1 What is meant by the following terms? Give an example of the reaction in each case. (i) Cyanohydrin
Cyanohydrins are organic compounds having the formula RR'C(OH)CN, where R and R ‘ are alkyl or aryl groups. Aldehydes and ketones react with hydrogen cyanide in the presence of excess sodium cyanide...
Figure below shows a 2.0 V potentiometer used for the determination of internal resistance of a 1.5 V cell. The balance point of the cell in open circuit is 76.3 cm. When a resistor of 9.5 Ω is used in the external circuit of the cell, the balance point shifts to 64.8 cm length of the potentiometer wire. Determine the internal resistance of the cell.
Answer – According to the question statement – Internal resistance of the cell is r = 1.5 V cell Balance point of the cell in open circuit is l = 76.3 cm External resistance is R = 9.5 Ω...
Figure shows a potentiometer with a cell of 2.0 V and internal resistance 0.40 Ω maintaining a potential drop across the resistor wire AB. A standard cell which maintains a constant emf of 1.02 V (for very moderate currents upto a few mA) gives a balance point at 67.3 cm length of the wire. To ensure very low currents drawn from the standard cell, a very high resistance of 600 kΩ is put in series with it, which is shorted close to the balance point. The standard cell is then replaced by a cell of unknown emf ε and the balance point found similarly, turns out to be at 82.3 cm length of the wire.
(a) What is the value ε? (b) What purpose does the high resistance of 600 kΩ have? (c) Is the balance point affected by this high resistance (d) Would the method work in the above...
Name the reagents used in the following reactions:
(i) Dehydration of propan-2-ol to propene.(ii) Butan-2-one to butan-2-ol. (i) Concentrated H2SO4 or H3PO4 (ii) catalytic hydrogenation or sodium borohydride (NaBH4) or lithium aluminium hydride...
Name the reagents used in the following reactions:
(i) Bromination of phenol to 2,4,6-tribromophenol.(ii) Benzyl alcohol to benzoic acid. (i) Bromine water (ii) Acidified potassium permanganate
Determine the current drawn from a 12V supply with internal resistance 0.5Ω by the infinite network shown in the figure. Each resistor has 1Ω resistance.
Solution: Let the infinite network's effective resistance be X. Because it is an endless network, adding three 1 resistance resistors will have no effect on the total resistance. X, in other...
Name the reagents used in the following reactions:
(i) Oxidation of primary alcohol to a carboxylic acid.(ii) Oxidation of primary alcohol to aldehyde. (i) NaBH4 or LiAlH4 to acidified KMnO4 (ii) Pyridinium chlorochromate (PCC)
How are the following conversions carried out?
(i) Ethyl magnesium chloride → Propan-1-ol.(ii) Methyl magnesium bromide → 2-Methylpropan-2-ol. (i) When ethyl magnesium chloride reacts with methane, an adduct is formed, resulting in propan–1–ol...
(a) Given n resistors each of resistance R, how will you combine them to get the (i) maximum (ii) minimum effective resistance? What is the ratio of the maximum to minimum resistance?
(b) Given the resistances of 1 Ω, 2 Ω, 3 Ω, how will be combine them to get an equivalent resistance of (i) (11/3) Ω (ii) (11/5) Ω, (iii) 6Ω, (iv) (6/11) Ω?
(c) Determine the equivalent resistance of networks shown in figure
Answer – (a) The total number of resistors is equal to n. Each resistor's resistance is equal to R. (i) When the resistors are connected in series, the maximum effective resistance is...
How are the following conversions carried out?
(i) Propene → Propan-2-ol.(ii) Benzyl chloride → Benzyl alcohol. (i) Propene is converted to propan–2–ol when it is treated with water in the presence of an acid catalyst. (ii) If benzyl chloride is...
Write the mechanism of acid dehydration of ethanol to yield ethene.
The mechanism of acid dehydration of ethanol to yield ethene takes place in the following three steps : Step 1 : Formation of ethyl oxonium by protonation of ethanol : Step 2 : Formation of a...
Explain the following with an example.
(i) Williamson ether synthesis.(ii) Unsymmetrical ether. (i) Williamson's ether synthesis is the process of obtaining ethers by treating an alkyl halide with a suitable sodium alkoxide. The sodium...
Choose the correct alternative:
(a) Alloys of metals usually have (greater/less) resistivity than that of their constituent metals.
(b) Alloys usually have much (lower/higher) temperature coefficients of resistance than pure metals.
(c) The resistivity of the alloy manganin is nearly independent of/ increases rapidly with increase of temperature.
(d) The resistivity of a typical insulator (e.g., amber) is greater than that of a metal by a factor of the order of (1022/1023).
Solution: (a) greater (b) lower (c) nearly independent of (d) 1022
Answer the following questions:
(a) A steady current flows in a metallic conductor of the non-uniform cross-section. Which of these quantities is constant along the conductor: current, current density, electric field, drift speed?
(b) Is Ohm’s law universally applicable for all conducting elements? If not, give examples of elements that do not obey Ohm’s law.
(c) A low voltage supply from which one needs high currents must have very low internal resistance. Why?
(d) A high tension (HT) supply of, say, 6 kV must have a very large internal resistance. Why?
Answer: (a) The current is assumed to be constant. As a result, it's a constant. The area of cross-section has an inverse relationship with current density, electric field, and drift speed. (b) No,...
Explain the following with an example. (i) Kolbe’s reaction, (ii) Reimer-Tiemann reaction.
(i) Kolbe's reaction: When sodium phenoxide is heated with C02 at 400°C under 4-7 atmospheres and then acidified, the main result is 2-hydroxybenzoic acid (salicylic acid), with a minor amount of...
Two wires of equal length, one of aluminium and the other of copper have the same resistance. Which of the two wires is lighter? Hence explain why aluminium wires are preferred for overhead power cables. (ρAl = 2.63 × 10–8 Ω m, ρCu = 1.72 × 10–8 Ω m, Relative density of Al = 2.7, of Cu = 8.9.)
Answer – According to the question statement, some given properties of aliminium are – Length = l1 Resistance = R Resistivity ,ρAI =ρ1= 2.63×10−8 Ωm Relative density , d1 = 2.7 Area...
16.
What conclusion can you draw from the following observations on a resistor made of alloy manganin? Current AVoltage VCurrent A Voltage...
Give equations of the following reactions:
(i) Dilute HNO3 with phenol.(ii) Treating phenol with chloroform in the presence of aqueous NaOH. (i) (ii)
(a) Six lead-acid type of secondary cells each of emf 2.0 V and internal resistance 0.015 Ω are joined in series to provide a supply to a resistance of 8.5 Ω. What is the current drawn from the supply and its terminal voltage?
(b) A secondary cell after long use has an emf of 1.9 V and a large internal resistance of 380 Ω. What maximum current can be drawn from the cell? Could the cell drive the starting motor of a car?
Answer – According to the question statement, (a) Emf of the secondary cells is ε = 2.0 V N is the number of secondary cells = 6 Then total EMF is given by – E = nε = 6 x 2 E = 12 V r =...
Give equations of the following reactions: (i) Oxidation of propan-1-ol with alkaline KMnO4solution, (ii) Bromine in CS2, with phenol.
(i) (ii)
The earth’s surface has a negative surface charge density of 10–9 C m–2. The potential difference of 400 kV between the top of the atmosphere and the surface results (due to the low conductivity of the lower atmosphere) in a current of only 1800 A over the entire
globe. If there were no mechanism of sustaining atmospheric electric field, how much time (roughly) would be required to neutralise the earth’s surface? (This never happens in practice because there is a mechanism to replenish electric charges, namely the continual thunderstorms and lightning in different parts of the globe). (Radius of earth = 6.37 × 106 m.)
Answer – According to the question statement, Surface charge density of the earth is σ = 10−9 cm−2Potential difference between the surface and the top of the atmosphere is V= 400 kVCurrent over...
The number density of free electrons in a copper conductor estimated in Example 3.1 is 8.5 × 1028 m–3. How long does an electron take to drift from one end of a wire 3.0 m long to its other end? The area of cross-section of the wire is 2.0 × 10–6 m2 and it is carrying a current of 3.0 A.
Answer – It is given that Number density of free electrons in a copper conductor is n = 8.5 x 10 28 m – 3 Assume that the Length of the copper wire is denoted by l and we have l...
Explain how does the –OH group attached to a carbon of benzene ring activates it towards electrophilic substitution.
Because the – OH group works as an electron-donating group, the electron density in the benzene ring increases. The resonance structure of phenol, as seen below, clearly demonstrates this. As a...
In a potentiometer arrangement, a cell of emf 1.25 V gives a balance point at 35.0 cm length of the wire. If the cell is replaced by another cell and the balance point shifts to 63.0 cm, what is the emf of the
second cell?
Answer – According to the question statement – Emf of the cell, E 1 = 1.25 V Balance point of potentiometer, given by l 1 = 35 cm In the question, the cell is replaced by another...
Explain why ortho nitrophenol is more acidic than ortho methoxyphenol.
An electron-withdrawing group is a nitro – group. The electron density in the O – H bond is reduced by the presence of this group in the ortho position. As a result, it is simpler to give away a...
A storage battery of emf 8.0 V and internal resistance 0.5 Ω is being charged by a 120 V dc supply using a series resistor of 15.5 Ω. What is the terminal voltage of the battery during charging? What is the
purpose of having a series resistor in the charging circuit?
Answer – According to the question statement – The EMF of storage battery is E = 8.0 V Internal resistance of battery is given by r = 0.5 Ω DC supply voltage is V = 120 V Resistance of the resistor...
Give two reactions that show the acidic nature of phenol. Compare the acidity of phenol with that of ethanol.
The acidic nature of phenol can be proven with the two reactions shown below : (i) Reaction with sodium: Phenol reacts with active metals like sodium to liberate Hydrogen gas. (ii) Reaction with...
A ) In a meter bridge given below, the balance point is found to be at 39.5 cm from the end A, when the resistor S is of 12.5 Ω. Determine the resistance of R. Why are the connections between resistors in a Wheatstone or meter bridge made of thick copper strips?
B ) Determine the balance point of the bridge above if R and S are interchanged. C ) What happens if the galvanometer and cell are interchanged at the balance point of the bridge? Would the...
Show how will you synthesize:
(i) 1-phenylethanol from a suitable alkene.(ii) cyclohexylmethanol using an alkyl halide by an SN2 reaction.(iii) pentan-1-ol using a suitable alkyl halide? (i) 1 – phenylethanol can be made by...
You are given benzene, conc.H2SO4and NaOH. Write the equations for the preparation of phenol using these reagents.
Write a chemical reaction for the preparation of phenol from chlorobenzene.
Chlorobenzene is mixed with NaOH (at 623 K and 320 atm pressure) to make sodium phenoxide, which is then acidified to produce phenol.
Determine the current in each branch of the network shown in the figure:
Answer : The current flowing through the network's numerous branches is depicted in the diagram below: Let I 1 denote the current flowing through the outer circuit Let I 2 denote...
Write the mechanism of hydration of ethene to yield ethanol.
In the mechanism of hydration of ethene to create ethanol, three steps are involved. These are the actions to take: Step 1: Electrophilic action of H3O+ protonates ethene to create carbocation:...
A heating element using nichrome connected to a 230 V supply draws an initial current of 3.2 A which settles after a few seconds to a steady value of 2.8 A. What is the steady temperature of the heating
element if the room temperature is 27.0 °C? Temperature coefficient of resistance of nichrome averaged over the temperature range involved is 1.70 × 10–4 °C–1.
Answer – According to the question; Supply voltage, V = 230 V initial current drawn is given by I 1 = 3.2 A Let the initial resistance be given by R 1, which can be determined...
Give the equations of reactions for the preparation of phenol from cumene.
Cumene is first oxidized in the presence of air to produce cumene hydroperoxide, which is then used to make phenol. The cumene hydroxide was then treated with dilute acid to produce phenol and...
A silver wire has a resistance of 2.1 Ω at 27.5 °C, and a resistance of 2.7 Ω at 100 °C. Determine the temperature coefficient of resistivity of silver.
Answer – According to the question – Temperature T 1 = 27.5 ° C Resistance R 1 is given as, R 1 = 2.1 Ω ( at T 1 ) Also, Temperature T 2 = 100...
While separating a mixture of ortho and para nitrophenols by steam distillation, name the isomer which will be steam volatile. Give reason.
o-nitrophenol and p-nitrophenol both have intramolecular H–bonding. Because intermolecular bonding exists in p–nitrophenol, the molecules are tightly bound together. As a result, o – nitrophenol is...
A negligibly small current is passed through a wire of length 15 m and uniform cross-section 6.0 × 10–7 m2, and its resistance is measured to be 5.0 Ω. What is the resistivity of the material at the temperature of the experiment?
Answer – We are given that, Length of the wire, L = 15 m Area of cross – section is given by a = 6.0 x 10 – 7 m 2 Let the resistance of the wire material be, R = 5.0 Ω The...
At room temperature (27.0 °C) the resistance of a heating element is 100 Ω. What is the temperature of the element if the resistance is found to be 117 Ω, given that the temperature coefficient of the material of the resistor is 1.70 × 10–4 °C–1.
Answer – We are given that, Room temperature, T = 27 ° C Resistance of heating element , R = 100 Ω Let the increased temperature of the filament be given by T 1 At T 1, the...
Give the structures and IUPAC names of monohydric phenols of molecular formula, C7H8O.
a) Three resistors 2 Ω, 4 Ω and 5 Ω are combined in parallel. What is the total resistance of the combination?
b) If the combination is connected to a battery of emf 20 V and negligible internal resistance, determine the current through each resistor, and the total current drawn from the battery. Answer – A...
What is meant by the hydroboration-oxidation reaction? Illustrate it with an example.
The hydroboration–oxidation reaction is one in which borane is used to initiate the oxidation process. Propan–1–ol, for example, is made by subjecting propene to a hydroboration – oxidation...
a) Three resistors 1 Ω, 2 Ω, and 3 Ω are combined in series. What is the total resistance of the combination?
b) If the combination is connected to a battery of emf 12 V and negligible internal resistance, obtain the potential drop across each resistor. Answer – a) According to the question, resistors...
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact
Due to the presence of the –OH group, alcohols form H – bonds with water. As a result, alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses.
A battery of EMF 10 V and internal resistance 3 Ω is connected to a resistor. If the current in the circuit is 0.5 A, what is the resistance of the resistor? What is the terminal voltage of the battery when the circuit is closed?
Answer – We are given: The EMF of the battery as E = 10 V The internal resistance of the battery as R = 3 Ω The current in the circuit as I = 0.5 A Let the resistance of the resistor...
Explain why propanol has a higher boiling point than that hydrocarbon, butane.
Propanol undergoes intermolecular H-bonding due to the presence of the – OH group. Butane, on the other hand, does not have the same advantages. In order to dissolve the intermolecular hydrogen...
The storage battery of a car has an emf of 12 V. If the internal resistance of the battery is 0.4 Ω, what is the maximum current that can be drawn from the battery?
Answer – We are given, The EMF of the battery, E = 12 V The internal resistance of the batter, R = 0.4 Ω The amount of maximum current drawn from the battery = I We know that, according to...
(i) Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names. (ii) Classify the isomers of alcohols in question.
(a) CH3-CH2-CH2-CH2-CH2-OH Pentan – 1 – ol (1 °) (ii) Primary alcohol: Pentan – 1 – ol ; 2 – Methylbutan – 1 – ol ; 3 – Methylbutan – 1 – ol ; 2, 2 – Dimethylpropan – 1 – ol Secondary alcohol:...
Write IUPAC names of the following compounds:
(i) 1 – Phenoxyheptane (ii) 2 – Ethoxybutane
Write IUPAC names of the following compounds:
(i) 1 – Methoxy – 2 – methyl propane (ii) Ethoxy benzene
Write IUPAC names of the following compounds:
(i) 2, 5 – Dimethylphenol (ii) 2, 6 – Dimethylphenol
Write IUPAC names of the following compounds:
(i) 2-Methylphenol (ii) 4 – Methyl phenol
Write IUPAC names of the following compounds:
(i) Butane – 2, 3 – diol (ii) Propane – 1, 2, 3 – triol
Write IUPAC names of the following compounds:
Solutions: (i) 2, 2, 4 -Trimethylpentan – 3 – ol (ii) 5 – Ethylheptane – 2, 4 – diol
How the following conversions can be carried out?
(i) tert-Butyl bromide to isobutyl bromide (ii) Aniline to phenylisocyanide Solutions: (i) (ii)
How the following conversions can be carried out?
(i) Chloroethane to butane (ii) Benzene to diphenyl Solutions: (i) (ii)
How the following conversions can be carried out?
(i) Chlorobenzene to p-nitrophenol (ii) 2-Bromopropane to 1-bromopropane Solutions: (i) (ii)
How the following conversions can be carried out?
(i) 2-Chloropropane to 1-propanol (ii) Isopropyl alcohol to iodoform Solution: (i) (ii)
How the following conversions can be carried out?
(i) Ethyl chloride to propanoic acid (ii) But-1-ene to n-butyliodide Solution: (i) (ii)
How the following conversions can be carried out?
(i) 2-Chlorobutane to 3, 4-dimethylhexane (ii) 2-Methyl-1-propene to 2-chloro-2-methylpropane Solution: (i) (ii)
How the following conversions can be carried out?
(i) Ethanol to propanenitrile (ii) Aniline to chlorobenzene Solution: (i) (ii)
How the following conversions can be carried out?
(i) Benzene to 4-bromonitrobenzene (ii) Benzyl alcohol to 2-phenylethanoic acid Solution: (i) (ii)
How the following conversions can be carried out?
(i) 1-Bromopropane to 2-bromopropane (ii) Toluene to benzyl alcohol Solution: (i) (ii)
How the following conversions can be carried out?
(i) Propene to propan-1-ol (ii) Ethanol to but-1-yne Solution: (i) (ii) OR
Predict conditions under which Al might be expected to reduce MgO.
After 13500C, the standard Gibbs free energy of formation of Al2O3 from Al is lower than that of MgO from Mg. As a result, Al can be expected to be able to reduce MgO at temperatures above 13500C.
Outline the principles of refining of metals by Vapour phase refining.
Vapour phase refining : It is a process of metal refining in which the metal is converted into a volatile compound, which is then decomposed to obtain the pure metal.(1) To accomplish this, the...
Outline the principles of refining of metals by Electrolytic refining.
It is the process of employing electricity to refine impure metals. The anode is impure metal, and the cathode is a thin sheet of pure metal in this process. The electrolyte is a metal-specific salt...
Outline the principles of refining of metals by Zone refining.
The premise behind this approach is that contaminants are more soluble in molten metal than in solid metal. A rotating circular heater slowly glides over an impure metal rod or bar in zone refining....
What is the role of graphite rod in the electrometallurgy of aluminium?
The anode in aluminium electrometallurgy is graphite, and the cathode iron is graphite lined. Electrolysis liberates O2, which combines with the graphite anode to produce CO2 and CO. If it hadn't...
Name the processes from which chlorine is obtained as a by-product. What will happen if an aqueous solution of NaCl is subjected to electrolysis?
Chlorine is obtained as a byproduct of the Down process. A fused mixture of CaCl2 and NaCl is electrolyzed at 873 K in this method. At the cathode, sodium is obtained, while Cl2 is released at the...
The choice of a reducing agent in a particular case depends on thermodynamic factor. How far do you agree with this statement? Support your opinion with two examples.
Ans: The graph of Gibbs energy ∆Gθ versus Temperature for the formation of solid oxides is shown above. This graph shows that if the ∆fGθ of a metal's oxide is more negative than the ∆fGθ of another...
Out of C and CO, which is a better reducing agent for ZnO?
At roughly 1673 K, ZnO is converted to Zn. The Gibbs free energy of formation of CO from C is smaller than the Gibbs free energy of formation of ZnO after 1073 K, and the Gibbs free energy of...
The value of ∆fG0 for formation of Cr2 O3 is – 540 kJmol−1 and that of Al2 O3 is – 827 kJmol−1 Is the reduction of Cr2 O3 possible with Al ?
Cr2O3 has a higher value of formation ( −540 kJ mol−1 ) than Al2O3 ( −827 kJ /mol ). Therefore, Al can reduce Cr2O3 to Cr. 2Al + (3/2)O2 ⇒ Al2O3 2Cr + (3/2)O2 ⇒ Cr2O3 Subtracting, 2Al +...
Why is zinc not extracted from zinc oxide through reduction using CO?
CO will not be able to reduce ZnO to Zn because the standard Gibbs free energy of formation of CO to CO2 is larger than that of Zn to ZnO. As a result, CO-assisted zinc oxide reduction is not...
How is leaching carried out in case of low grade copper ores?
When working with low-grade copper ores, bacteria or acids are utilised to leach the copper in the presence of air. Copper is introduced to the solution as Cu2+ ions in this method: Cu + 2H+ + ½...
What is the role of cryolite in the metallurgy of aluminium?
The basic functions of Na3AlF6 (Cryolite) are as follows: 1. Due to the presence of cryolite, the melting point of the solution drops from 2323 K to 1140 K. 2. Cryolite makes alumina a good...
Why copper matte is put in silica lined converter?
Cu2S and FeS are present in copper matte. When a hot blast of air passes through a molten matte in a silica coated converter, the matte's FeS oxidises to FeO. FeSiO3 (slag) is formed when FeO...
Differentiate between “minerals” and “ores”.
Minerals are substances that contain metals or metal compounds and are found in nature. Ores are rocks and minerals that can be mined for metals in a cost-effective and convenient manner. For...
How is ‘cast iron’ different from ‘pig iron”?
The iron obtained from a blast furnace is known as pig iron. It is 4 percent carbon and contains minor levels of additional impurities such as Si, P, S, and Mn.Cast iron is created by using a hot...
Giving examples, differentiate between ‘roasting’ and ‘calcination’.
Calcination is the process of converting carbonate and hydroxide ores to oxides by heating them to temperatures below their melting points while avoiding or limiting the presence of air.This...
How can you separate alumina from silica in a bauxite ore associated with silica? Give equations, if any.
The following steps are used to separate alumina from silica in bauxite ore that contains silica:To begin, the powdered ore is digested in a concentrated NaOH solution at 473 523 K and 35 36 bar...
Describe a method for refining nickel.
Mond's procedure is a nickel refinement technology. Heat is applied to nickel in the presence of carbon monoxide in this process, resulting in nickel tetracarbonyl, a volatile complex. Ni + 4CO ⇒...
What is the criterion followed while selecting the stationary phase of chromatography?
The stationary phase is chosen so that the constituents of the mixture have varying degrees of solubility in it. As a result, distinct elements move at different speeds through the phase, allowing...
“Chromatography”, what do you understand by this term?
Chromatography is a term that refers to a group of scientific procedures that are used to purify, separate, characterise, and identify the species in a mixture, whether coloured or colourless. The...
State the role of silica in the metallurgy of copper.
During roasting, copper pyrites are transformed to a combination of Cu2O and FeO. During the smelting process, acidic silica is introduced to separate the FeO from the mixture. FeSiO3 (slag) is...
Write chemical reactions taking place in the extraction of zinc from zinc blende.
The various steps involved in zinc extraction are : (1) Concentration of oreZinc blende is crushed first to eliminate impurities from the ore, and then froth flotation is used to concentrate the...
Write down the reactions taking place in different zones in the blast furnace during the extraction of iron.
At 500 – 800 K3Fe2O3 +CO → 2Fe3O4 + CO2Fe2O3 +CO → 2FeO + CO2 At 850 KFe3O4 +CO → 3FeO + CO2 At 900 – 1500 KFeO +CO → Fe + CO2CO2 + C → 2CO Above 1570 KC+ O2 → CO2FeO +C → Fe + COCaCO3(s) → CaO(s) +...
Name the common elements present in the anode mud in electrolytic refining of copper. Why are they so present ?
During the electrolytic refinement of copper, frequent components found in the anode mud include selenium, silver, tellurium, platinum, antimony, and gold.These metals are almost unaffected by the...
Out of C and CO, which is a better reducing agent at 673 K ?
CO is a more effective reducing agent at 673 K because the value of ∆G( C,CO ) is bigger than ∆G( CO,CO2 ), implying that CO is more easily reduced to CO2 than C to CO.
Explain Column chromatography.
Column chromatography: This is a great way to purify elements that are only present in trace levels. Column chromatography is commonly used to remove impurities that have chemical properties that...
Explain Zone refining.
Zone refining: The premise behind this approach is that contaminants are more soluble in molten metal than in solid metal. A rotating circular heater slowly glides over an impure metal rod or bar in...
Why is the extraction of copper from pyrites more difficult than that from its oxide ore through reduction?
Copper (I) sulfite has a lower ∆fG (Gibbs free energy of formation) than hydrogen sulfite and carbon disulfite (CS2). As a result, neither C nor H2S will be able to convert Cu2S to Cu.Because...
What is the role of depressant in froth floatation process?
In froth flotation, the job of a depressant is to separate two sulphide ores by selectively preventing one ore from foaming. For example, to separate two sulphide ores, PbS and ZnS, NaCN is...
Copper can be extracted by hydrometallurgy but not zinc. Explain.
Copper's reduction potential is greater than that of zinc or iron. Iron and zinc can thus be employed to remove copper from a solution in hydrometallurgy. Fe(s) + Cu2+(aq) ⇒ Fe2+ (aq) + Cu(s)...
Monochromatic light of wavelength 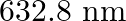 is produced by a helium-neon laser. The power emitted is
is produced by a helium-neon laser. The power emitted is 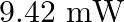 . How fast does a hydrogen atom have to travel in order to have the same momentum as that of the photon?
. How fast does a hydrogen atom have to travel in order to have the same momentum as that of the photon?
We are provided that the momentum of the hydrogen atom is equal to the momentum of the photon, $\mathrm{P}=1.047 \times 10^{-27} \mathrm{~kg} \mathrm{~m} / \mathrm{s}$ Momentum's expression is: $P=m...
Monochromatic light of wavelength 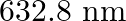 is produced by a helium-neon laser. The power emitted is
is produced by a helium-neon laser. The power emitted is 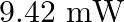 (a) Find the energy and momentum of each photon in the light beam
(a) Find the energy and momentum of each photon in the light beam
(b) How many photons per second, on the average, arrive at a target irradiated by this beam? (Assume the beam to have a uniform cross-section which is less than the target area)
Wavelength of a monochromatic light is given as $\lambda=632.8 \mathrm{~nm}=632.8 \times 10^{-9} \mathrm{~m}$ Power emitted by the laser is, $P=9.42 \mathrm{~mW}=9.42 \times 10^{-3} \mathrm{~W}$...
A Rowland ring of mean radius 15 cm has 3500 turns of wire wound on a ferromagnetic core of relative permeability 800. What is the magnetic field B in the core for a magnetising current of 1.2 A?
Answer- Mean radius of the Rowland ring is given = 15 cm Number of turns = 3500 Relative permeability of the core is given by μr = 800 Magnetizing current is given by I = 1.2 A Magnetic field...
A monoenergetic (18 keV) electron beam initially in the horizontal direction is subjected to a horizontal magnetic field of 0.04 G normal
to the initial direction. Estimate the up or down deflection of the beam over a distance of 30 cm (me
= 9.11 × 10–31 kg). [Note: Data in this exercise are so chosen that the answer will give you an idea of
the effect of earth’s magnetic field on the motion of the electron beam from the electron gun to the screen in a TV set.]
Answer – Energy of the electron beam is given by E = 18 keV Or, E = 18 x 103 eV = 18 x 103 x 1.6 x 10-19 J Magnetic field is given by B = 0.04 G Mass of the electron is given by...
A magnetic dipole is under the influence of two magnetic fields. The angle between the field directions is 60°, and one of the fields has a magnitude of 1.2 × 10–2 T. If the dipole comes to stable equilibrium at an angle of 15° with this field, what is the magnitude of the other field?
Answer- Magnitude of one of the magnetic field is given by B1 = 1.2 × 10–2 T Suppose that the magnitude of the other field is B2 And the angle between the field is given, θ = 60° We...
A compass needle free to turn in a horizontal plane is placed at the centre of circular coil of 30 turns and radius 12 cm. The coil is in a vertical plane making an angle of 45° with the magnetic meridian. When the current in the coil is 0.35 A, the needle points west to east.
(a) Determine the horizontal component of the earth’s magnetic field at the location.
(b) The current in the coil is reversed, and the coil is rotated about its vertical axis by an angle of 90° in the anticlockwise sense looking from above. Predict the direction of the needle. Take the magnetic declination at the places to be zero.
Answer: Number of turns is given = 30 Radius of the coil is given = 12 cm Current in the coil is given = 0.35 A Angle of dip, given by δ = 450 (a) Horizontal component of earth’s magnetic field...
A telephone cable at a place has four long straight horizontal wires carrying a current of 1.0 A in the same direction east to west. The earth’s magnetic field at the place is 0.39 G, and the angle of dip is 35°. The magnetic declination is nearly zero. What are the resultant magnetic fields at points 4.0 cm below the cable?
Answer – First it is important to decide the direction which would best represent the given situation. We are given that BH = B cos δ = 0.39 × cos 35o G Therefore, BH =...
A long straight horizontal cable carries a current of 2.5 A in the direction 10° south of west to 10° north of east. The magnetic meridian
of the place happens to be 10° west of the geographic meridian. The earth’s magnetic field at the location is 0.33 G, and the angle of dip is zero. Locate the line of neutral points (ignore the thickness of the
cable)? (At neutral points, magnetic field due to a current-carrying the cable is equal and opposite to the horizontal component of earth’s magnetic field.)
Answer – Current in the wire is given by 2.5 A The earth’s magnetic field at a location is given by R= 0.33 G = 0.33 x 10-4 T Angle of dip is zero is given by δ = 0 Horizontal...
Answer the following questions:
(a) Explain qualitatively on the basis of domain picture the irreversibility in the magnetisation curve of a ferromagnet.
(b) The hysteresis loop of a soft iron piece has a much smaller area than that of a carbon steel piece. If the material is to go through repeated cycles of magnetisation, which piece will dissipate greater heat energy?
(c) ‘A system displaying a hysteresis loop such as a ferromagnet, is a device for storing memory?’ Explain the meaning of this statement.
(d) What kind of ferromagnetic material is used for coating magnetic tapes in a cassette player, or for building ‘memory stores’ in a modern computer?
(e) A certain region of space is to be shielded from magnetic fields. Suggest a method.
Answer – (a) When a substance is placed in an external magnetic field, the domain aligns in the direction of the magnetic field. The process of alignment consumes some energy. The substance retains...
Answer the following questions:
(a) Why does a paramagnetic sample display greater magnetisation (for the same magnetising field) when cooled?
(b) Why is diamagnetism, in contrast, almost independent of temperature?
(c) If a toroid uses bismuth for its core, will the field in the core be (slightly) greater or (slightly) less than when the core is empty?
(d) Is the permeability of a ferromagnetic material independent of the magnetic field? If not, is it more for lower or higher fields?
(e) Magnetic field lines are always nearly normal to the surface of a ferromagnet at every point. (This fact is analogous to the static electric field lines being normal to the surface of a conductor at
every point.) Why?
(f) Would the maximum possible magnetisation of a paramagnetic sample be of the same order of magnitude as the magnetisation
of a ferromagnet?
Answer – (a) At a lower temperature, thermal motion is reduced, and the tendency to disturb the alignment of the dipoles is reduced. (b) The induced dipole moment is always in the opposite direction...
A short bar magnet of magnetic moment 5.25 × 10–2 J T–1 is placed with its axis perpendicular to the earth’s field direction. At what distance from the centre of the magnet, the resultant field is inclined at 45° with earth’s field on (a) its normal bisector and (b) its axis. The magnitude of the earth’s field at the place is given to be 0.42 G. Ignore the length of the magnet in comparison to the distances involved.
Answer : Magnetic moment of the bar magnet is given by M = 5.25 × 10–2 J T–1 Magnitude of earth’s magnetic field at a place is given by H = 0.42 G = 0.42×10-4 T The magnetic field on the...
If the bar magnet in exercise 5.13 is turned around by 180°, where will the new null points be located?
Answer – The magnetic field at a distance d1 = 14 cm on the axis of the magnet can be written as – ${{B}_{1}}=\frac{{{\mu }_{0}}2M}{4\pi {{d}_{1}}^{3}}=H$ Where, M is the magnetic moment...
A short bar magnet placed in a horizontal plane has its axis aligned along the magnetic north-south direction. Null points are found on the axis of the magnet at 14 cm from the centre of the magnet. The
earth’s magnetic field at the place is 0.36 G and the angle of dip is zero. What is the total magnetic field on the normal bisector of the magnet at the same distance as the null–point (i.e., 14 cm) from the centre of the magnet? (At null points, field due to a magnet is equal and opposite to the horizontal component of earth’s magnetic field.)
Answer : Earth’s magnetic field at the given place is given by H = 0.36 G The magnetic field at distance d from the axis of the magnet can be calculated using the following formula –...
A short bar magnet has a magnetic moment of 0.48 J T–1. Give the direction and magnitude of the magnetic field produced by the magnet at a distance of 10 cm from the centre of the magnet on (a) the axis,
(b) the equatorial lines (normal bisector) of the magnet.
Magnetic moment of the bar magnet is given by M = 0.48 J T–1 Distance, d = 10 cm = 0.1 m The magnetic field at distance d from the magnet's centre on the axis can be calculated using the following...
At a certain location in Africa, a compass points 12° west of the geographic north. The north tip of the magnetic needle of a dip circle placed in the plane of magnetic meridian points 60° above the
horizontal. The horizontal component of the earth’s field is measured to be 0.16 G. Specify the direction and magnitude of the earth’s field at the location.
Answer – We are given the angle of declination, θ = 12° Angle of dip, given by δ = 60° Horizontal component of earth’s magnetic field is given by BH = 0.16 G Earth’s magnetic field at the given...
A magnetic needle free to rotate in a vertical plane parallel to the magnetic meridian has its north tip pointing down at 22° with the horizontal. The horizontal component of the earth’s magnetic field
at the place is known to be 0.35 G. Determine the magnitude of the earth’s magnetic field at the place.
Answer – Horizontal component of earth’s magnetic field is given by BH = 0.35 G Angle made by the needle with the horizontal plane is known as the Angle of dip, given by δ=22° Earth’s magnetic field...
A circular coil of 16 turns and radius 10 cm carrying a current of 0.75 A rests with its plane normal to an external field of magnitude 5.0 × 10–2 T. The coil is free to turn about an axis in its plane perpendicular to the field direction. When the coil is turned slightly and released, it oscillates about its stable equilibrium with a frequency of 2.0 s–1. What is the moment of inertia of the coil about its axis of rotation?
Answer – Number of turns in the circular coil is given by N = 16 Radius of the coil is given by r = 10 cm = 0.1 m Cross-section of the coil, A is given by – $A=\pi {{r}^{2}}=\pi {{(0.1)}^{2}}$...
A closely wound solenoid of 2000 turns and area of cross-section 1.6×10-4 m2, carrying 4.0 A current, is suspended through its centre, thereby allowing it to turn in a horizontal plane.
(a) What is the magnetic moment associated with the solenoid? (b) What is the force and torque on the solenoid if a uniform the horizontal magnetic field of 7.5 × 10–2 T is set up at an angle...
A line charge λ per unit length is lodged uniformly onto the rim of a wheel of mass M and radius R. The wheel has light non-conducting spokes and is free to rotate without friction about its axis. A uniform magnetic field extends over a circular region within the rim. It is given by,
\[B=-{{B}_{0}}k(r\le a;a<R)\] = 0 (otherwise) What is the angular velocity of the wheel after the field is suddenly switched off? Answer – Line charge per unit length is given by the expression –...
(i) We are given a long straight wire and a square loop of given size (refer to figure). Find out an expression for the mutual inductance between both.
(ii) Now, consider that we passed an electric current through the straight wire of 50 A, and the loop is then moved to the right with constant velocity, v = 10 m/s.Find the emf induced in the loop...
We have an air-cored solenoid having a length of 30 cm, whose area is 25cm2 and a number of turns are 500. And the solenoid has carried a current of 2.5 A. Suddenly the current is turned off and the time is taken for it is 10−3s. What would be the average value of the induced back -emf by the ends of the open switch in the circuit? (Neglect the variation in the magnetic fields near the ends of the solenoid.)
Answer – Given, Length of the solenoid is given by l = 30 cm = 0.3 m Area of the solenoid, then becomes A =25 cm2 = 25 × 10−4m2 Number of turns on the solenoid is given by N = 500 Current in...
In the given figure we have a metal rod PQ which is put on the smooth rails AB and these are kept in between the two poles of permanent magnets. All these three (rod, rails and the magnetic field ) are in mutually perpendicular direction. There is a galvanometer ‘G’ connected through the rails by using a switch ‘K’. Given, Rod’s length = 15 cm , Magnetic field strength, B = 0.50 T, Resistance produced by the closed-loop = 9.0mΩ. Let’s consider the field is uniform.
(i) Determine the polarity and the magnitude of the induced emf if we will keep the K open and the rod will be moved with the speed of 12 cm/s in the direction shown in the figure. (ii) When...
We have a powerful loudspeaker magnet and have to measure the magnitude of the field between the poles of the speaker. And a small search coil is placed normal to the field direction and then quickly removed out of the field region, the coil is of 2cm2 area and has 25 closely wound turns. Similarly, we can give the coil a quick 90 degree turn to bring its plane parallel to the field direction. We have measured the total charge flown in the coil by using a ballistic galvanometer and it comes to 7.5 mC. Total resistance after combining the coil and the galvanometer is 0.50Ω. Estimate the field strength of the magnet.
Ans: Given, Coil’s Area is A = 2cm2 = 2×10-4m2 Number of turns on the coil is given by N = 25 Total Charge in the coil is given by Q = 7.5 mC = 7.5×10−3C The combo of coil and galvanometer...
We have a square loop having side as 12 cm and its sides are parallel to x and y-axis is moved with a velocity of 8 cm /s in the positive x-direction in a region which have a magnetic field in the direction of positive z-axis. The field is not uniform whether in case of its space or in the case of time. It has a gradient of 10−3 T cm−1 along the negative x-direction(i.e its value increases by 10−3 T cm−1 as we move from positive to negative direction ), and it is reducing in the case of time with the rate of 10−3 T s−1 . Calculate the magnitude and direction of induced current in the loop (Given: Resistance = 4.50mΩ).
Answer – We have, Side of the Square loop is given by s = 12cm = 0.12m Area of the loop becomes – A = s × s = 0.12 × 0.12 A = 0.0144 m2 Velocity of the loop is given by v = 8 cm-1 =...
Let us assume that the loop in question number 4 is stationary or constant but the current source which is feeding the electromagnet which is producing the magnetic field is slowly decreased. It was having an initial value of 0.3 T and the rate of reducing the field is 0.02 T / sec. If the cut is joined to form the loop having a resistance of 1.6 \Omega1.6Ω. Calculate how much power is lost in the form of heat? What is the source of this power?
Answer – A rectangular loop is given having sides as 8 cm and 2 cm. Therefore, the area of the loop will be will be given by A = L × B A = 8 cm × 2 cm = 16 cm2 A = 16×10−4m2 Value of...
A jet plane is travelling towards the west at a speed of 1800 km/h. What is the voltage difference developed between the ends of the wing having a span of 25 m, if the Earth’s magnetic field at the location
has a magnitude of 5 × 10–4 T and the dip angle is 30°.
Answer – We have been provided the following data – Speed of the plane with which it is moving is given by v = 1800 km/h = 500 m/s Wing span of the jet is given by l = 25 m Magnetic field...
A pair of adjacent coils has a mutual inductance of 1.5 H. If the current in one coil changes from 0 to 20 A in 0.5 s, what is the change of flux linkage with the other coil?
Ans: Given, Mutual inductance µ = 1.5H Current at initial point is given by I1= 0 A Current at final point is given by I2 = 20 A Therefore, change in current becomes dI = I1- I2 = =...
Current in a circuit falls from 5.0 A to 0.0 A in 0.1 s. If an average emf of 200 V induced, give an estimate of the self-inductance of the circuit.
Ans: Current at initial point is given by I1 = 5.0 A Current at final point is given by I2 = 0.0 A Therefore the change in current is given by dI = I1- I2 = 5 A Total time taken is...
Question 12.19: An organic compound contains 69.77% carbon, 11.63% hydrogen and rest oxygen. The molecular mass of the compound is 86. It does not reduce Tollens’ reagent but forms an addition compound with sodium hydrogensulphite and give positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acid. Write the possible structure of the compound.
Solution: The number of carbon, hydrogen, and oxygen atoms in an organic compound can thus be expressed as: Therefore, the empirical formula of the compound is C5H10O. Now, the empirical formula...
Question 12.20: Although phenoxide ion has more number of resonating structures than carboxylate ion, carboxylic acid is a stronger acid than phenol. Why?
Solution: The phenoxide ion has the following resonance structures: The resonance structures of the phenoxide ion show that less electronegative carbon atoms carry a negative charge in II, III, and...
Question 12.18: Give a plausible explanation for: (iii) During the preparation of esters from a carboxylic acid and an alcohol in the presence of an acid catalyst, the water or the ester should be removed as soon as it is formed.
Solution: In the presence of an acid, an ester along with water is generated reversibly from a carboxylic acid and an alcohol. Because the reaction is reversible, if either water or ester is not...
Question 12.18: Give a plausible explanation for: (ii) There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Solution: Only one of the two −NH2 groups, which is connected directly to the carbonyl-carbon atom, undergoes resonance in semicarbazide. As a result, the electron density on the −NH2 group in the...
Question 12.18: Give a plausible explanation for: (i) Cyclohexanone forms cyanohydrin in good yield but 2, 2, 6 trimethylcyclohexanone does not.
Solution: (i) According to the equation below, cyclohexanones produce cyanohydrins. There will be no steric hindrance in this situation, allowing the nucleophile CN– to attack with ease. However,...
Question 12.25: Complete each synthesis by giving missing starting material, reagent or products
Solutions:
Question 12.24: Complete each synthesis by giving missing starting material, reagent or products
Solutions:
Question 12.23: Complete each synthesis by giving missing starting material, reagent or products
Solutions:
A bar magnet of magnetic moment 1.5 J /T lies aligned with the direction of a uniform magnetic field of 0.22 T.
(a) What is the amount of work required by an external torque to turn the magnet so as to align its magnetic moment: (i) normal to the field direction, (ii) opposite to the field direction? (b) What...
If the solenoid in Exercise 5.5 is free to turn about the vertical direction and a uniform horizontal magnetic field of 0.25 T is applied, what is the magnitude of the torque on the solenoid when its axis makes an angle of 30° with the direction of applied field?
Answer – Magnetic field strength is given by B = 0.25 T Magnetic moment is by M = 0.6 T-1 The angle θ between the direction of the applied field...
The photoelectric cut-off voltage in a certain experiment is 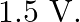 What is the maximum kinetic energy of photoelectrons emitted?
What is the maximum kinetic energy of photoelectrons emitted?
Photoelectric cut-off voltage is given as $\mathbf{V}_{\mathbf{0}}=\mathbf{1 . 5} \mathbf{V}$ The maximum kinetic energy for emitted photoelectrons, $K_{e}=eV$ Where, $e=$ charge on an electron...
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl ( primary, secondary, tertiary ), vinyl, or aryl halides :
(i) (CH3)2CHCH(Cl)CH3 (ii) CH3CH2CH(CH3)CH(C2H5)Cl Solution: (i) 2 − Chloro − 3 – methylbutane (Secondary alkyl halide) (ii) 3 − Chloro − 4 – methyhexane (Secondary alkyl...
The work function of caesium metal is 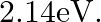 When light of frequency
When light of frequency 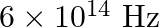 is incident on the metal surface, photoemission of electrons occurs. What is the maximum speed of the emitted photoelectrons?
is incident on the metal surface, photoemission of electrons occurs. What is the maximum speed of the emitted photoelectrons?
$\mathbf{v}$ is the maximum speed of photoelectrons emittedThe kinetic energy relation can be written as:$$\mathrm{K}=\frac{1}{2} m v^{2}$$Where,$$\mathrm{m}=\text { mass of electron }=9.1 \times...
The work function of caesium metal is 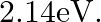 When light of frequency
When light of frequency 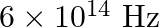 is incident on the metal surface, photoemission of electrons occurs. What is the (a) maximum kinetic energy of the emitted electrons (b) Stopping potential
is incident on the metal surface, photoemission of electrons occurs. What is the (a) maximum kinetic energy of the emitted electrons (b) Stopping potential
Work function of caesium is given as $\Phi_{o}=2.14 \mathrm{eV}$ Frequency of light is given as $\mathbf{v}=6.0 \times 10^{14} \mathrm{~Hz}$ (a) The maximum energy (kinetic) by the photoelectric...
A closely wound solenoid of 800 turns and area of cross-section 2.5 × 10–4 m2 carries a current of 3.0 A. Explain the sense in which the solenoid acts like a bar magnet. What is its associated magnetic moment?
Answer – It is given that the number of turns in the solenoid, n = 800 Area of cross section is given by, A = 2.5×10-4 m2 And the current in the solenoid, I = 3.0 A Because a magnetic field develops...
A short bar magnet of magnetic moment m = 0.32 JT–1 is placed in a uniform magnetic field of 0.15 T. If the bar is free to rotate in the plane of the field, which orientation would correspond to its (a) stable, and (b) unstable equilibrium? What is the potential energy of the magnet in each case?
Answer – Moment of the bar magnet is given by M = 0.32 JT–1 External magnetic field, is given by B = 0.15 T The magnetic field is aligned with the bar magnet. This system is said to be in...
A short bar magnet placed with its axis at 30° with a uniform external magnetic field of 0.25 T experiences a torque of magnitude equal to 4.5×10-2J. What is the magnitude of the magnetic moment of the magnet?
Answer – We are given the magnetic field strength, B = 0.25 T Torque on the bar magnet is given by, T = 4.5×10-2J Θ = 30° is the angle between the external magnetic field and the bar...
Answer the following:
(a) A vector needs three quantities for its specification. Name the three independent quantities conventionally used to specify the earth’s magnetic field. (b) The angle of dip at a location in...
Answer the following:
(a) The earth’s magnetic field varies from point to point in space. Does it also change with time? If so, on what time scale does it change appreciably? (b) The earth’s core is known to...
A horizontal straight wire 10 m long extending from east to west is falling with a speed of 5.0 m s–1, at right angles to the horizontal component of the earth’s magnetic field, 0.30 × 10–4 Wb m–2.
(a) What is the instantaneous value of the emf induced in the wire?
(b) What is the direction of the emf?
(c) Which end of the wire is at the higher electrical potential?
Answer...
A circular coil of radius 8.0 cm and 20 turns is rotated about its vertical diameter with an angular speed of 50 rad s–1 in a uniform horizontal magnetic field of magnitude 3.0 × 10–2 T. Obtain the maximum and average emf induced in the coil. If the coil forms a closed loop of resistance 10 Ω, calculate the maximum value of current in the coil. Calculate the average power loss due to Joule heating. Where does this power come from?
Answer – Maximum emf induced is given = 0.603 V Average emf induced is given by= 0 V Maximum current in the coil = 0.0603 A Power loss (average) is given =...
A rectangular wire loop of sides 8 cm and 2 cm with a small cut is moving out of a region of the uniform magnetic field of magnitude 0.3 T directed normal to the loop. What is the emf developed across the cut if the velocity of the loop is 1 cm s–1 in a direction normal to the (a) longer side, (b) shorter side of the loop? For how long does the induced voltage last in each case?
Answer – Length of the wired loop is given by l = 8 cm = 0.08 m Width of the wired loop is given by b = 2 cm = 0.02 m Since the loop is a rectangle, the area of the wired loop is given by – A = lb =...
A long solenoid with 15 turns per cm has a small loop of area 2.0 cm2 placed inside the solenoid normal to its axis. If the current carried by the solenoid changes steadily from 2.0 A to 4.0 A in 0.1 s, what is the induced emf in the loop while the current is changing?
Answer – We are given - Number of turns – 15 turns / cm = 1500 turns / m Number of turns per unit length is given by n = 1500 turns The solenoid has a small loop of area which is given by A = 2.0...
We are rotating a 1 m long metallic rod with an angular frequency of 400 red s^{-1}s−1 with an axis normal to the rod passing through its one end. And on to the other end of the rod, it is connected with a circular metallic ring. There exist a uniform magnetic field of 0.5 T which is parallel to the axis everywhere. Find out the emf induced between the centre and the ring.
Answer – Length of the rod is given = 1m Angular frequency is given by = ω = 400 rad/sec Magnetic field strength is given by B = 0.5 T The rod has zero linear velocity at one end and a linear...
Predict the direction of induced current in the situations described by the following figures
Answer – Lenz’s law predicts the direction of the induced current in a closed loop. When the North pole of a bar magnet is moved towards a closed loop, a current is induced in the loop which flows...
Question 12.22: Complete each synthesis by giving missing starting material, reagent or products.
Solutions:
Question 12.21: Complete each synthesis by giving missing starting material, reagent or products.
Solutions:
Question 12.20: Complete the synthesis by giving missing starting material, reagent or products.
Solution:
Question 12.19: Describe decarboxylation.
Decarboxylation is the process by which carboxylic acids lose carbon dioxide and create hydrocarbons when their sodium salts are heated with soda-lime. Decarboxylation occurs when alkali metal salts...
Question 12.18: Describe Cross aldol condensation.
Cross-aldol condensation occurs when two different aldehydes, two different ketones, or an aldehyde and a ketone react together. If both reactants contain α-hydrogens, four compounds are produced as...
Question 12.17: Describe Cannizzaro reaction.
The self-disproportionation (oxidation-reduction) reaction of aldehydes with no alpha hydrogens, when treated with concentrated alkalis, is known as the Cannizzaro reaction. In this reaction, one...
Question 12.16: Describe Acetylation.
Acetylation is the process of adding an acetyl function group to a chemical molecule. When this process is carried out, bases such as pyridine, dimethylaniline, and others are present. In this...
