Answer – We know that for ground level, n1 = 1 Suppose E1 is the energy of the level n1. So as we know E1 is related with n1 as – \[{{E}_{1}}=\frac{13.6}{{{n}_{1}}^{2}}eV=-13.6eV\]...
What is the De Broglie wavelength of an electron with a kinetic energy of 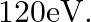
de Broglie wavelength of an electron having a momentum $p$, is given by the relation: $\lambda=\frac{h}{p}$ $=\frac{6.6 \times 10^{-34}}{5.91 \times 10^{-24}}=1.116 \times 10^{-10} \mathrm{~m}=0.112...
The ground state energy of hydrogen atom is –13.6 eV. What are the kinetic and potential energies of the electron in this state?
Ans: We know that the ground state energy of hydrogen atom is given by – E = − 13.6 eV The total energy of hydrogen atom is equal to -13.6 eV. It is understood that the kinetic energy is equal to...
A difference of 2.3 eV separates two energy levels in an atom. What is the frequency of radiation emitted when the atom make a transition from the upper level to the lower level?
Answer – We know that the separation of two energy levels in an atom is given by the relation – \[E=2.3eV=2.3\times 1.6\times {{10}^{-19}}\] \[E=3.68\times {{10}^{-19}}J\] Let v be the frequency of...
What is the: (a) Momentum, (b) Speed of an electron with a kinetic energy of 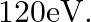
Kinetic energy of the electron is given as $\mathbf{E}_{\mathrm{K}}=\mathbf{1 2 0} \mathrm{eV}$ Planck's constant, $\mathbf{h}=\mathbf{6 . 6} \times \mathbf{1 0}^{-34} \mathrm{~J} \mathrm{~s}$ Mass...
What is the shortest wavelength present in the Paschen series of spectral lines?
Answer – Using the Rydberg’s formula, given the relation – \[\frac{hc}{\lambda }=21.76\times {{10}^{-19}}[\frac{1}{{{n}_{1}}^{2}}-\frac{1}{{{n}_{2}}^{2}}]\] Where h is the Planck’s constant, given =...
Suppose you are given a chance to repeat the alpha-particle scattering experiment using a thin sheet of solid hydrogen in place of the gold foil. (Hydrogen is a solid at temperatures below 14 K.) What results do you expect?
Answer – It is quite known that the mass of the incident alpha particle (6.64 × 10-27kg) is much more than the mass of hydrogen (1.67 × 10-27Kg). Hence, the alpha particle would fail to rebound...
Choose the correct alternative from the clues given at the end of each statement:
(d) An atom shows continuous mass distribution in a ……….but a very high non-uniform mass distribution in ……….(Thomson’s model/ Rutherford’s model.) (e) In ………. the positively charged part of the...
Choose the correct alternative from the clues given at the end of each statement:
(a) In Rutherford’s model the atomic size is ……….. the size of the atom in Thomson’s model. (much greater than/no different from/much less than.) (b) ) In ………. the ground state electrons...
Calculate the (a) momentum, and (b) the de Broglie wavelength of the electrons accelerated through a potential difference of 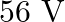 .
.
Potential difference is given in the question as $\mathbf{V}=\mathbf{5 6 V}$ Planck's constant, $\mathbf{h}=6.6 \times 10^{-34} \mathrm{~J} \mathrm{~s}$ Mass of an electron, $\mathbf{m}=\mathbf{9 ....
Light of wavelength 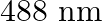 is produced by an argon laser which is used in the photoelectric effect. When light from this spectral line is incident on the emitter, the stopping (cut-off) potential of photoelectrons is
is produced by an argon laser which is used in the photoelectric effect. When light from this spectral line is incident on the emitter, the stopping (cut-off) potential of photoelectrons is 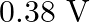 . Find the work function of the material from which the emitter is made.
. Find the work function of the material from which the emitter is made.
Wavelength of light produced by the argon laser is given as $\lambda=488 \mathrm{~nm}=488 \times 10^{-9} \mathrm{~m}$ Stopping potential of the photoelectrons is given as...
What will be the correct order for the wavelengths of absorption in the visible region for the following:
![Rendered by QuickLaTeX.com \[{{[\mathbf{Ni}{{(\mathbf{N}{{\mathbf{O}}_{\mathbf{2}}})}_{\mathbf{6}}}]}^{\mathbf{4}}},\text{ }{{[\mathbf{Ni}{{(\mathbf{N}{{\mathbf{H}}_{\mathbf{3}}})}_{\mathbf{6}}}]}^{\mathbf{2}+}},\text{ }{{[\mathbf{Ni}{{({{\mathbf{H}}_{\mathbf{2}}}\mathbf{O})}_{\mathbf{6}}}]}^{\mathbf{2}+}}?\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c719fbf2dc2de970f85896e939d068d0_l3.png)
Because all of the complexes have the same metal ion, the energy absorption is determined by the ligands' CFSE values. The ligands' CFSE values are in the order of H2O < NH3 < NO2- according...
Light of frequency 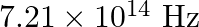 is incident in a metal surface. Electrons with a maximum speed of
is incident in a metal surface. Electrons with a maximum speed of 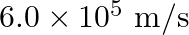 are ejected from the surface. What is the threshold frequency for photoemission of electrons?
are ejected from the surface. What is the threshold frequency for photoemission of electrons?
Frequency of the incident photon is provided as $v=488 \mathrm{~nm}=488 \times 10^{-9} \mathrm{~m}$ Maximum speed of the electrons is $\mathbf{v}=\mathbf{6 . 0} \times \mathbf{1 0}^{5} \mathrm{~m} /...
Amongst the following, the most stable complex is ![Rendered by QuickLaTeX.com \[\left( i \right)\text{ }{{[Fe{{({{H}_{2}}O)}_{6}}]}^{3+}}\left( ii \right)\text{ }{{[Fe{{(N{{H}_{3}})}_{6}}]}^{3+}}~\left( iii \right)\text{ }{{[Fe{{({{C}_{2}}{{O}_{4}})}_{3}}]}^{3}}~~~\left( iv \right)\text{ }{{[FeC{{l}_{6}}]}^{3}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c9055c71fe68a5c25b0ec883d67368e1_l3.png)
Fe has an oxidation state of +3 in all circumstances. The chelating ligand (C2O4)3 is a bidentate chelating ligand that produces chelating rings. As a result, the most stable complex is ( iii ).
The work function for a certain metal is  Will this metal give photoelectric emission for incident radiation of wavelength 330 nm?
Will this metal give photoelectric emission for incident radiation of wavelength 330 nm?
Work function of the metal is given as $\Phi_{o}=4.2 \mathrm{eV}$ Charge on an electron is $\mathbf{e}=\mathbf{1 . 6} \times \mathbf{1 0}^{-19} \mathbf{C}$ Planck's constant, $\mathbf{h}=\mathbf{6 ....
What is the oxidation number of cobalt in K[Co(CO)4]?
The threshold frequency for a certain metal is 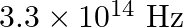 . If the light of frequency
. If the light of frequency 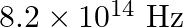 is incident on the metal, predict the cut-off voltage for the photoelectric emission.
is incident on the metal, predict the cut-off voltage for the photoelectric emission.
Threshold frequency of the metal given to us is $\mathbf{v}_{\mathbf{0}}=\mathbf{3 . 3} \times \mathbf{1 0}^{\mathbf{1 4}} \mathrm{Hz}$. Frequency of light incident on the metal is given as...
Amongst the following ions which one has the highest magnetic moment value? ![Rendered by QuickLaTeX.com \[\left( i \right)\text{ }{{[Cr{{({{H}_{2}}O)}_{6}}]}^{3+}}\left( ii \right)\text{ }{{[Fe{{({{H}_{2}}O)}_{6}}]}^{2+}}\left( iii \right)\text{ }{{[Zn{{({{H}_{2}}O)}_{6}}]}^{2+}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a172782a885937508308ebe32dd3d3ec_l3.png)
How many ions are produced from the complex Co(NH3)6Cl2 in solution?
(i) 6 (ii) 4 (iii) 3 (iv) 2
(iii) The answer is 3. The given complex [ Co( NH3)6 ]Cl2 ionizes to give three ions, viz one [ Co( NH3)6] + and two Cl – ions.
What can be inferred from the magnetic moment values of the following complex species?
Example Magnetic Moment (BM)
K4[Mn(CN)6) 2.2
[Fe(H2O)6]2+ 5.3
K2[MnCl4] 5.9
Magnetic moment is calculated by the formula: ν=[n(n+2)]1/2 For value n = 1, ν=[1(1+2)]1/2= (3)1/2 = 1.732 For value n = 2, ν=[2(2+2)]1/2= (8)1/2 = 2.83 For value n = 3, ν=[3(3+2)]1/2 =...
A 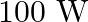 sodium lamp radiates energy uniformly in all directions. The lamp is located at the centre of a large sphere that absorbs all the sodium light which is incident on it. The wavelength
sodium lamp radiates energy uniformly in all directions. The lamp is located at the centre of a large sphere that absorbs all the sodium light which is incident on it. The wavelength
of the sodium light is 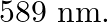 (a) What is the energy per photon associated with the sodium light? (b) At what rate are the photons delivered to the sphere?
(a) What is the energy per photon associated with the sodium light? (b) At what rate are the photons delivered to the sphere?
Power of the sodium lamp is given as $\mathbf{P}=\mathbf{1 0 0 W}$ Wavelength of the emitted sodium light is given as $\lambda=589 \mathrm{~nm}$ $=589 \times 10^{-9} \mathrm{~m}$ Planck's constant,...
Discuss briefly giving an example in each case the role of coordination compounds in:
(i) analytical chemistry and (ii) extraction/metallurgy of metals.
(i) Role in analytical chemistry:Determination of hardness of the water. (ii) Complexes are generated during the extraction of metals from ores, and they play a role in metallurgy and...
In an experiment on the photoelectric effect, the slope of the cut-off voltage versus frequency of incident light is found to be 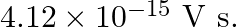 Calculate the value of Planck’s constant.
Calculate the value of Planck’s constant.
The slope of cut-off voltage (V) versus frequency (v) is given as, $\frac{V}{v}=4.12 \times 10^{-15} \mathrm{Vs}$ $\mathrm{V}$ and frequency is related by the equation: $\mathrm{Hv}=\mathrm{eV}$...
Discuss briefly giving an example in each case the role of coordination compounds in:
(i) biological systems (ii) medicinal chemistry
(i) Role in biological systems: There are various essential coordination compounds in the bodies of animals, such as hemoglobin, which is an iron coordination compound. In plants, chlorophyll...
Comment on the statement that elements of the first transition series possess many properties different from those of heavier transition elements.
In many ways, the properties of elements of heavier transition elements differ from those of the first transition series. (a) The elements of the first transition series form low-spin or high-spin...
The energy flux of sunlight reaching the surface of the earth is 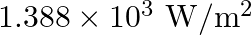 . How many photons are incident on the Earth per second/square meter? Assume an average wavelength of
. How many photons are incident on the Earth per second/square meter? Assume an average wavelength of 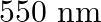
Energy flux of sunlight reaching the surface of the earth: $\phi=1.388 \times 10^{3} \mathrm{~W} / \mathrm{m}^{2}$ Hence, power of sunlight per square metre, $\mathbf{P}=\mathbf{1 . 3 8 8} \times...
What is meant by the chelate effect? Give an example.
The metal-ligand bond becomes more stable when a polydentate or bidentate ligand attaches to a metal ion in such a way that it takes on the structure of a ring. Chelate rings are the name for these...
Explain the violet color of the complex ![Rendered by QuickLaTeX.com \[{{[\mathbf{Ti}{{({{\mathbf{H}}_{\mathbf{2}}}\mathbf{O})}_{\mathbf{6}}}]}^{\mathbf{3}+}}~\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a69eb6951f610e97c75cb1f3a64bd20f_l3.png)
on the basis of crystal field theory.
The degree/level of association among the species involved in a state of equilibrium determines the stability of a coordination molecule in a solution. The formation constant or stability constant...
Write down the number of 3d electrons in each of the following ions: Ti2+, V2+, Cr3+, Mn2+, Fe2+, Fe3+, Co2+, Ni2+ and Cu2+. Indicate how would you expect the five 3d orbitals to be occupied for these hydrated ions (octahedral).
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following points: (i) ionisation enthalpies and (ii) atomic sizes
In each of the three transition series, generally the first ionisation enthalpy increases from left to right. However, there are some exceptions. The first ionisation enthalpies of the third...
Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration, and coordination number. Also, give stereochemistry and magnetic moment of the complex: ![Rendered by QuickLaTeX.com \[\left( i \right)\text{ }[CrC{{l}_{3}}{{\left( py \right)}_{3}}]\left( ii \right)\text{ }Cs[FeC{{l}_{4}}]\left( iii \right)\text{ }{{K}_{4}}[Mn{{\left( CN \right)}_{6}}]\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-94b27372566420147b9890b0383935a6_l3.png)
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following points: (i) electronic configurations (ii) oxidation states
(i) In the 1st, 2nd and 3rd transition series, the 3d, 4d, and 5d orbitals are filled with electrons respectively. We know that elements in the same group generally have similar electronic...
Write down the IUPAC name for each of the following complexes and indicate the oxidation state, electronic configuration and coordination number. Also, give stereochemistry and magnetic moment of the complex:
(i) K[Cr(H2O)2(C2O4)2].3H2O (ii) [Co(NH3)5Cl]Cl2
n = 0.Thus, Magnetic moment = 0
Give the oxidation state, d orbital occupation and coordination number of
the central metal ion in the following complexes:
(i) (NH4)2[CoF4] (ii) [Mn(H2O)6]SO4
Give the oxidation state, d orbital occupation and coordination number of
the central metal ion in the following complexes:
(i) K3[Co(C2O4)3] (iii) (NH4)2[CoF4]
Discuss the nature of bonding in metal carbonyls.
The metal-carbon bond in metal carbonyls has both the and bond characteristics. The carbonyl carbon donates a lone pair of electrons to the metal's vacant orbital, forming a connection. The filled d...
![Rendered by QuickLaTeX.com \[{{[\mathbf{Fe}{{\left( \mathbf{CN} \right)}_{\mathbf{6}}}]}^{\mathbf{4}}}~\mathbf{and}\text{ }{{[\mathbf{Fe}{{({{\mathbf{H}}_{\mathbf{2}}}\mathbf{O})}_{\mathbf{6}}}]}^{\mathbf{2}+}}~\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-3b1c1b754a174142f48bf8ad312b8baa_l3.png)
are of different colours in dilute solutions. Why?
[Fe( H2O)6 ]2+ and [ Fe( CN)6 ]2+ have H2O and CN- ligands respectively. CN- has a higher CFSE (crystal field splitting energy) than water because it is a strong field ligand. As a result,...
Write the electronic configurations of the elements with the atomic numbers 61, 91, 101 and 109.
A solution of [Ni(H2O)6]2+ is green but a solution of [Ni(CN)4]2– is colorless. Explain
[ Ni (H2O)6 ] is made up of the Ni+2 ion, which has a 3d8 electrical structure. Because H2O is a weak ligand, there are two unpaired electrons in this arrangement that cannot pair up. The d – d...
![Rendered by QuickLaTeX.com \[{{[\mathbf{Cr}{{(\mathbf{N}{{\mathbf{H}}_{\mathbf{3}}})}_{\mathbf{6}}}]}^{\mathbf{3}+}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-39b74154417ca7572b9b77e89bc26feb_l3.png)
is paramagnetic while [Ni(CN)4]2– is diamagnetic. Explain why.
In [ Ni ( CN)4 ] 2−, Ni has an oxidation state of +2. Thus, it has d 8configuration.Ni 2+ : Because CN- is a strong field ligand, electrons in 3d orbitals couple. Ni 2+ undergoes dsp2 hybridization...
Name the members of the lanthanoid series which exhibit +4 oxidation states and those which exhibit +2 oxidation states. Try to correlate this type of behaviour with the electronic configurations of these elements.
In the parenthesis are the atomic numbers of the elements are given. Tb after forming Tb4+ attains a stable electronic configuration of [Xe] 4f7. Yb after forming Yb2+ attains a stable...
What is crystal field splitting energy? How does the magnitude of ∆o decide the actual configuration of d orbitals in a coordination entity?
The difference in energy between the two levels ( t2g and eg ) that have split from a degenerated d orbital due to the presence of a ligand is known as crystal-field splitting energy. It is...
Use Hund’s rule to derive the electronic configuration of Ce3+ ion, and calculate its magnetic moment on the basis of ‘spin-only’ formula.
Ce:1s22s22p63s23p63d104s24p64d105s25p64f15d16s2 Ce3+:1s22s22p63s23p63d104s24p64d105s25p64f1 Magnetic moment can be calculated by the formula: μ = [n(n+2)]1/2 Where, n = number of unpaired...
Which is the last element in the series of the actinoids? Write the electronic configuration of this element. Comment on the possible oxidation state of this element.
In the actinoid series, the last element is lawrencium, Lr. The atomic number of the element is 103 and its electronic configuration is [Rn] 5f14 6d1 7s2 . The most common oxidation...
What is the spectrochemical series? Explain the difference between a weak field ligand and a strong field ligand.
The Spectrochemical series is a set of common ligands arranged in ascending order of their crystal-field splitting energy (CFSE). CFSE readings are higher in strong field ligands. Weak field...
Draw a figure to show the splitting of d orbitals in an octahedral crystal field.
The chemistry of the actinoid elements is not so smooth as that of the lanthanoids. Justify this statement by giving some examples from the oxidation state of these elements.
Lanthanoids project 3 different oxidation states which are +2, +3, +4. Among these, the +3 oxidation state is the most common. Lanthanoids exhibit a limited number of oxidation states because of the...
What are inner transition elements? Decide which of the following atomic numbers are the atomic numbers of the inner transition elements : 29, 59, 74, 95, 102, 104.
Inner transition metals are the elements in which the last electron is entering in the f-orbital. The elements in which the 4f and the 5f orbitals are progressively filled are called f-block...
What are alloys? Name an important alloy which contains some of the lanthanoid metals. Mention its uses.
An alloy is defined as a solid solution of two or more elements present in a combined form in a metallic matrix. It can either be a partial solid solution or a complete solid solution. Alloys...
Indicate the steps in the preparation of: (i) KMnO4 from pyrolusite ore.
(i) Potassium permanganate ( KMnO4 ) can be prepared from pyrolusite (MnO2). The ore is fused with KOH in the presence of either atmospheric oxygen or an oxidising agent, such as KNO3 or...
Indicate the steps in the preparation of: (i) K2Cr2O7 from chromite ore. (ii) KMnO4 from pyrolusite ore.
(i) Potassium dichromate ( K2Cr2O7 ) is prepared from chromite ore (FeCr2O4) in the following steps. Step (1): Preparation of sodium chromate \[4FeC{r_2}{O_4} + 16NaOH + 7{O_2} \to...
Give examples and suggest reasons for the following features of the transition metal chemistry: (i) The highest oxidation state is exhibited in oxoanions of a metal.
(i) Oxygen is a strong oxidising agent due to its high electronegativity and small size. So, oxo-anions of a metal have the highest oxidation state. For example, in , the oxidation state of Mn is +7.
Figure below shows a biconvex lens (of refractive index 1.50) in contact with a liquid layer on top of a plane mirror. A small needle with its tip on the principal axis is moved along the axis until its inverted image is found at the position of the needle. The distance of the needle from the lens is measured to be 45.0 cm. The liquid is removed and the experiment is repeated. The new distance is measured to be 30.0 cm. What is the refractive index of the liquid?
Answer – According to the question – Focal length of the given convex lens is f1 = 30 cm Since, the liquid acts as a mirror. Focal length of the liquid is denoted by f2 Total focal length...
Light incident normally on a plane mirror attached to a galvanometer coil retraces backwards as shown in Fig. 9.36. A current in the coil produces a deflection of 3.5° of the mirror. What is the displacement of the reflected spot of light on a screen placed 1.5 m away?
Answer – According to the question – Angle of deflection is θ = 3.5° The distance of the screen from the mirror is D = 1.5 m The deflection undergone by the reflected rays are twice the angle...
A Cassegrain telescope uses two mirrors as shown in Fig. 9.33. Such a telescope is built with the mirrors 20 mm apart. If the radius of curvature of the large mirror is 220 mm and the small mirror is 140 mm, where will the final image of an object at infinity be?
Answer - Below is the diagram of a Cassegrain telescope. It has a concave and a convex mirror. We are given – Distance between the secondary mirror and the objective mirror is d = 20 mm Radius of...
Answer the following –
(a)For a telescope, what is the separation between the objective lens and the eyepiece? (b) If this telescope is used to view a 100 m tall tower 3 km away, what is the height of the image of the...
A small telescope has an objective lens of focal length 140 cm and an eyepiece of focal length 5.0 cm. What is the magnifying power of the telescope for viewing distant objects when
(a) the telescope is in normal adjustment (i.e., when the final image is at infinity)? (b) the final image is formed at the least distance of distinct vision (25 cm)? Answer – According to the...
An angular magnification (magnifying power) of 30X is desired using an objective of focal length 1.25 cm and an eyepiece of focal length 5 cm. How will you set up the compound microscope?
Answer – According to the question – Focal length of the objective lens is fo= 1.25 cm The focal length of the eyepiece is fe = 5 cm Least distance of distinct vision is d = 25 cm Angular...
Answer the following questions:
(a) An object subtends an angle at the eye which is equal to the angle subtended at the eye by the virtual image that is produced by a magnifying glass. Does the magnifying glass provide angular...
The virtual image of each square in the figure is to have an area of 6.25 mm2. Find out, what should be the distance between the object in Exercise 9.30 and the magnifying glass?If the eyes are too close to the magnifier, would you be able to see the squares distinctly?
Answer – According to the question – Area of the virtual image of each square is A = 6.25 mm2 Area of each square is A0 = 1 mm2 Hence, the linear magnification of the object can be determined...
(a)Determine the distance in which the lens should be held from the figure in Exercise 9.29 in order to view the squares distinctly with the maximum possible magnifying power.
(b)Determine the magnification in the following situation. (c) Find if the magnifying power is equal to magnification. Answer – We have – (a) We get the maximum possible magnification when the image...
A large card divided into squares each of size 1 mm2 is being viewed from a distance of 9 cm through a magnifying glass ( converging lens has a focal length of 9 cm) held close to the eye. Determine:
(a) the magnification produced by the lens? How much is the area of each square in the virtual image? (b) the angular magnification (magnifying power) of the lens? (c) Is the magnification in (a)...
A child with normal near point (25 cm) reads a book with small size print using a magnifying glass: a thin convex lens of focal length 5 cm.
(a) What would be the shortest and the longest distance at which the lens should be placed from the page so that the book can be read easily when viewing through the magnifying glass? (b) What is...
A person looking at a cloth with a pattern consisting of vertical and horizontal lines is able to see the vertical lines more distinctly than the horizontal ones. What is this defect due to? How is such a defect of vision corrected?
Answer – The person in the circumstance is having trouble seeing the horizontal lines, but the vertical lines are clearly apparent. When the eye's refracting system does not work in the same way for...
Spectacles of power −1.0 dioptre is being used by a person suffering from myopia for distant vision. He also needs to use separate reading glass of power + 2.0 dioptres when he turns old. Explain what may have happened.
Answer – We have that, A myopic person uses a spectacle of power for distant vision, P = -1.0 D Focal length of the given spectacles is then determined by – f = $\frac{1}{P}$ =...
Does the human eye partially lose its ability of accommodation when it undergoes short-sightedness (myopia) or long-sightedness (hypermetropia)? If not, what might cause these defects of vision?
Answer – Eye-lenses are utilised when a person suffers from myopia or hypermetropia. Myopia is a condition in which the eyeballs gradually lengthen from front to rear. When the eyeballs start to...
For a normal eye, the far point is at infinity and the near point of distinct vision is about 25cm in front of the eye. The cornea of the eye provides a converging power of about 40 dioptres, and the least converging power of the eye-lens behind the cornea is about 20 dioptres. From this rough data estimate the range of accommodation (i.e., the range of converging power of the eye-lens) of a normal eye.
Answer: We have –Least distance of distinct vision is d = 25 cmFar point of a normal eye is d’ = ∞Converging power of the cornea is $P_{c}=40D$Least converging power of the given eye-lens is...
You are given prisms made of crown glass and flint glass with a wide variety of angles.
Suggest a combination of prisms which will
(i) deviate a pencil of white light without much dispersion,
(ii) disperse (and displace) a pencil of white light without much deviation.
Answer - (i) The two prisms must be in close proximity to one another. The bases of these two prisms must be on opposite sides of the white light that is incident. White light is dispersed for the...
At what angle should a ray of light be incident on the face of a prism of refracting angle
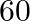 so that it just suffers total internal reflection at the other face? The refractive index of the material of the prism is 1.524.
so that it just suffers total internal reflection at the other face? The refractive index of the material of the prism is 1.524.
Answer –The incident, refracted and emergent rays associated with a glass prism ABC are shown in the given figure. Angle of prism, therefore is A = $60°$Refractive index of the prism is $\mu$=...
Answer the following questions –
(i) Determine the ‘effective focal length’ of the combination of the two lenses inExercise 9.10, if they are placed 8.0 cm apart with their principal axes coincident. Doesthe answer depend on which...
A screen is placed 90 cm from an object. The image of the object on the screen is
formed by a convex lens at two different locations separated by 20 cm. Determine the
focal length of the lens.
Answer: We are given that –Distance between the object and the image (screen) is D = 90 cmDistance between two locations of the given convex lens is d = 20 cmFocal length of the lens is fFocal...
The image of a small electric bulb fixed on the wall of a room is to be obtained on the
opposite wall 3 m away by means of a large convex lens. What is the maximum
possible focal length of the lens required for the purpose?
Answer: We are given that –Distance between the object and the image is d = 3 m Maximum focal length of the convex lens is = $f_{max}$The maximum focal length, for real images is given as:...
Answer the following questions:
(i) You have learnt that plane and convex mirrors produce virtual images of objects.Can they produce real images under some circumstances? Explain.(ii) A virtual image, we always say, cannot be...
Discuss the nature of bonding in the following coordination entities on the basis of valence bond theory: ![Rendered by QuickLaTeX.com \[\left( i \right)\text{ }{{[Co{{({{C}_{2}}{{O}_{4}})}_{3}}]}^{3}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-efe43055f50042489c9d11ee26754efd_l3.png)
![Rendered by QuickLaTeX.com \[\left( ii \right)\text{ }{{[Co{{F}_{6}}]}^{3-}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a46576bdb43c6d6939fb964ada245115_l3.png)
(i) Here, the oxidation state of cobalt is +3.Orbitals of Co 3+ ion : Oxalate is a field ligand with a low affinity. As a result, the electrons in the 3d orbital will not pair.Because there are six...
(i) Figure below shows a cross-section of a ‘light pipe’ made of a glass fiber of refractive
index 1.68. The outer covering of the pipe is made of a material of refractive index
1.44. What is the range of the angles of the incident rays with the axis of the pipe
for which total reflections inside the pipe take place, as shown in the figure.
(ii) What is the answer if there is no outer covering of the pipe?
Answer – (i) Refractive index of the glass fibre is = $\mu_{2}$= 1.68 Refractive index of the outer covering of thr pipe is = $\mu_{1}$= 1.44Angle of incidence is iAngle of refraction is rAngle...
Discuss the nature of bonding in the following coordination entities on the basis of valence bond theory: ![Rendered by QuickLaTeX.com \[\left( i \right)\text{ }{{[Fe{{\left( CN \right)}_{6}}]}^{4}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c4239d4c20c9394384021fc803fa63f1_l3.png)
![Rendered by QuickLaTeX.com \[\left( ii \right)\text{ }{{[Fe{{F}_{6}}]}^{3}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-2cec302fc55db082efb330ec046bbe8e_l3.png)
(i) Here, the oxidation state of Fe is +3.Fe 2+ : Electronic configuration is 3d6Orbitals of Fe2+ ion : CN− is a strong field ligand, so it causes the unpaired 3d electrons to pair up: There are six...
What is the coordination entity formed when an excess of aqueous KCN is added to an aqueous solution of copper sulfate? Why is it that no precipitate of copper sulphide is obtained when ![Rendered by QuickLaTeX.com \[{{H}_{2}}S\left( g \right)\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-c8bf88a9a8a76dacccc6552af3570fa6_l3.png)
is passed through this solution?
As a result of the foregoing procedure, the coordination entity obtained is K2[Cu(CN)4 ].The above coordination entity does not ionize to produce Cu2+ ions since it is extremely stable. When...
Aqueous copper sulfate solution (blue in color) gives: (i) a green precipitate with aqueous potassium fluoride and (ii) a bright green solution with aqueous potassium chloride. Explain these experimental results.
The presence of [Cu( H2O)4]2+ ions in an aqueous CuSO4 solution gives it a blue colour. (i) As a result, when KF is introduced, H2O ligands are replaced by F- ligands, resulting in green [ CuF4 ]2+...
Write all the geometrical isomers of [Pt(NH3)(Br)(Cl)(py)] and how many of these will exhibit optical isomers?
None of the isomers exhibit optical isomerism.
Draw all the isomers (geometrical and optical) of ![Rendered by QuickLaTeX.com \[{{[Co{{(N{{H}_{3}})}_{2}}C{{l}_{2}}\left( en \right)]}^{+}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-3d4533747f86351d7cf03af6d4d2667f_l3.png)
Draw all the isomers (geometrical and optical) of: (i) ![Rendered by QuickLaTeX.com \[{{[CoC{{l}_{2}}{{\left( en \right)}_{2}}]}^{+}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-a90cf5dd5a070919003d67e7a6ddfc4f_l3.png)
(ii) ![Rendered by QuickLaTeX.com \[{{[Co(N{{H}_{3}})Cl{{\left( en \right)}_{2}}]}^{2+}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-4265017b2224d863ef19cf033ef31c23_l3.png)
(i) (ii)
Draw the structures of optical isomers of ![Rendered by QuickLaTeX.com \[{{[Cr{{(N{{H}_{3}})}_{2}}C{{l}_{2}}\left( en \right)]}^{+}}\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-826b87fdd17fb106f6e60bd3b147a0cf_l3.png)
A small pin fixed on a tabletop is viewed from above from a distance of 50 cm. By
what distance would the pin appear to be raised if it is viewed from the same point
through a 15 cm thick glass slab held parallel to the table? Refractive index of glass =
1.5. Does the answer depend on the location of the slab?
Answer:
According to the question,The actual depth of the pin is d = 15 cmApparent depth of the pin is = d’Refractive index of glass is $\mu$=1.5 here, the ratio of actual depth to the apparent depth and...
Use the mirror equation to deduce that:
(i) an object placed between f and 2f of a concave mirror produces a real imagebeyond 2f.(ii) a convex mirror always produces a virtual image independent of the locationof the object.(iii) the...
Draw the structures of optical isomers of:
(i) [Cr(C2O4)3]3– (ii) [PtCl2(en)2]2+ Solution: (i) (ii)
How many geometrical isomers are possible in the following coordination entities? (i) [Cr(C2O4)3]3– (ii) [Co(NH3)3Cl3]
( i ) In [ Cr(C2O4)3] 3− no geometric isomers are present because it is a bidentate ligand. ( ii ) In [ Co( NH3)3 Cl3 ]two isomers are possible.
(i) A giant refracting telescope at an observatory has an objective lens of focal
length 15 m. If an eyepiece of focal length 1.0 cm is used, what is the angular
magnification of the telescope?
(ii)If this telescope is used to view the moon, what is the diameter of the imageof the moon formed by the objective lens? The diameter of the moon is $3.48 \times 10^{6} m$,and the radius of lunar...
A small telescope has an objective lens of focal length 144 cm and an eyepiece of focal
length 6.0 cm. What is the magnifying power of the telescope? What is the separation
between the objective and the eyepiece?
Answer:
Answer - According to the question –Focal length of the objective lens is $f_{o}$= 144 cmFocal length of the eyepiece is $f_{e}$= 6.0 cmThe magnifying power of the telescope is given by the...
A person with a normal near point (25 cm) using a compound microscope with objective of focal length 8.0 mm and an eyepiece of focal length 2.5cm can bring an object placed at 9.0 mm from the objective in sharp focus. What is the separation between the two lenses? Calculate the magnifying power of the microscope,
Answer: According to the question, Focal length of the given objective lens is $ f_{o}$= 8 mm = 0.8 cm Focal length of the eyepiece is $ f_{e}$= 2.5 cm Object distance for the given objective lens...
A compound microscope consists of an objective lens of focal length 2.0cm and an eyepiece of focal length 6.25cm separated by a distance of 15 cm. How far from the objective should an object be placed in order to obtain the final image at (a) the least distance of distinct vision (25 cm), and (b) at infinity? What is the magnifying power of the microscope in each case?
Answer: According to the question, we have – Focal length of the given objective lens is f1=2.0 cm Focal length of the given eyepiece is f2=6.25 cm Distance between the eyepiece and the objective...
What is the focal length of a convex lens of focal length 30cm in contact with a concave lens of focal length 20cm? Is the system a converging or a diverging lens? Ignore the thickness of the lenses.
Answer: According to the question – Focal length of the convex lens is f1=30cm Focal length of the concave lens is f2= -20cm Focal length of the system of lenses is denoted by f The equivalent...
An object of size 3.0cm is placed 14cm in front of a concave lens of focal length 21cm. Describe the image produced by the lens. What happens if the object is moved further away from the lens?
Answer: According to the question, Size of the object is $h_{1} = 3 cm$ Object distance is u = -14 cm Focal length of the given concave lens is f = -21 cm Image distance is denoted by v...
A beam of light converges at a point P. Now a lens is placed in the path of the convergent beam 12cm from P. At what point does the beam converge if the lens is (a) a convex lens of focal length 20cm, and (b) a concave lens of focal length 16cm?
Answer : According to the question, the object given is virtual and the image formed is real. Object distance is u= +12 cm (i) The focal length of the convex lens is f =20 cm Image distance is...
Double-convex lenses are to be manufactured from a glass of refractive index 1.55, with both faces of the same radius of curvature. What is the radius of curvature required if the focal length is to be 20cm?
Answer: We are given that, Refractive Index of glass is $\mu=1.55$ Focal length of the given double-convex lens is f= 20 cm Radius of curvature of one face of the given lens is =R1 Radius of...
A prism is made of glass of unknown refractive index. A parallel beam of light is incident on the face of the prism. The angle of minimum deviation is measured to be 40°. What is the refractive index of the
material of the prism? The refracting angle of the prism is 60°. If the prism is placed in water (refractive index 1.33), predict the new angle of minimum deviation of a parallel beam of light.
Answer: We are given that – Angle of minimum deviation is $\delta_{m}=40°$ Angle of the prism is A = $60°$ Refractive Index of water is given by, $\mu=1.33$ Material’s refractive Index =...
A small bulb is placed at the bottom of a tank containing water to a depth of 80cm. What is the area of the surface of water through which light from the bulb can emerge out? Refractive index of water is 1.33. (Consider the bulb to be a point source.)
Answer : We are given the following information – Bulb’s actual depth in water is d1=80 cm =0.8 m Refractive Index of water is $\mu$= 1.33 I is the Angle of incidence r is the Angle of refraction –...
List various types of isomerism possible for coordination compounds, giving an example of each.
The various types of isomerism that can be observed in coordination compounds are 🙁 i ) Geometrical isomerism : ( ii ) Optical isomerism : (iii) Coordination isomerism :This kind of isomerism...
Using IUPAC norms write the systematic names of the following: (i) [Ni(NH3)6]Cl2 (ii) [Co(en)3]3+ (iii) [Ni(CO)4]
( i ) Hexaamminenickel(II) chloride ( ii ) Tris(ethane-1, 2-diammine) cobalt(III) ion ( iii ) Tetracarbonylnickel(0)
Using IUPAC norms write the systematic names of the following: (i) [Mn(H2O)6]2+ (ii) [NiCl4]2–
(i ) Hexaquamanganese(II) ion ( ii) Tetrachloridonickelate(II) ion
. Using IUPAC norms write the systematic names of the following: (i) [Ti(H2O)6]3+ (ii) [Co(NH3)4Cl(NO2)]Cl
(i ) Hexaquatitanium(III) ion ( ii ) Tetraamminichloridonitrito-N-Cobalt(III) chloride
The figures above show the refraction of a ray in air incident at 60° with the normal to a glass-air and water-air interface, respectively. Predict the angle of refraction in glass when the angle of incidence
in water is 45° with the normal to a water-glass interface.
For the glass-air interface, we are given that – Angle of incidence is i =60° Angle of refraction is r=35° Using Snell’s law, the refractive index of the glass with respect to air becomes –...
Using IUPAC norms write the systematic names of the following: (i) [Co(NH3)6]Cl3 (ii) [Pt(NH3)2Cl(NH2CH3)]Cl
( i ) Hexaamminecobalt(III) chloride ( ii ) Diamminechlorido(methylamine) platinum(II) chloride
Using IUPAC norms write the formulas for the following: (i) Tetrabromidocuprate(II) (ii) Pentaamminenitrito-N-cobalt(III)
( i ) [ Cu (Br)4] 2− ( ii ) [Co ( ONO )( NH3)5] 2+
Using IUPAC norms write the formulas for the following: (i) Potassium tri(oxalato)chromate(III) (ii) Hexaammineplatinum(IV)
(i ) K3 [ Cr ( C2O4)3] ( ii ) [ Pt (NH3)6] 4+
Using IUPAC norms write the formulas for the following: (i) Pentaamminenitrito-O-cobalt(III) (ii) Hexaamminecobalt(III) sulphate
(i ) [ Co (NO2) ( NH3)5] 2+ ( ii) [ Co( NH3)6]2 (SO4)3
Using IUPAC norms write the formulas for the following: (i) Diamminedichloridoplatinum(II) (ii) Potassium tetracyanidonickelate(II)
( i ) [ Pt ( NH3)2Cl2] ( ii) K2[ Ni(CN )4]
Using IUPAC norms write the formulas for the following: (i) Tetrahydroxidozincate(II) (ii) Potassium tetrachloridopalladate(II)
( i ) [Zn(OH)4]2 – ( ii ) K2[ Pd Cl4]
Specify the oxidation numbers of the metals in the following coordination entities:
Solution: (i) (ii) (iii)
Specify the oxidation numbers of the metals in the following coordination entities:
(i) 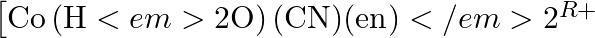
(ii) 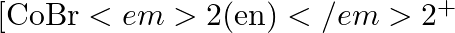
A tank is filled with water to a height of 12.5 cm. The apparent depth of a needle lying at the bottom of the tank is measured by a microscope to be 9.4 cm. What is the refractive index of water? If
water is replaced by a liquid of refractive index 1.63 up to the same height, by what distance would the microscope have to be moved to focus on the needle again?
Answer: We are given that Actual depth of the needle in water here is h1=12.5cm Apparent depth in water is h2 =9.4 cm Refractive Index of water is given by – $\mu$ The value of $\mu$ can be...
A 4.5 cm needle is placed 12 cm away from a convex mirror of focal length 15 cm. Give the location of the image and the magnification. Describe what happens as the needle is moved farther from the mirror.
Answer : We are given, Size of the needle is h1=4.5 cm Distance of object is u=-12 cm Focal length of the convex mirror is f= 15 cm Image distance is represented by v $\frac{1}{u}$+ $\frac{1}{v}$=...
What is meant by unidentate, bidentate, and ambidentate ligands? Give two examples for each.
(a) Unidentate ligands: these are ligands with one donor site. Example Cl–, NH3 (b) Bidentate – these are ligands with two donor sites.Example – Ethane-1,2-diamine, Oxalate ion ( C2O42- ) (c)...
Collect 5 examples of palindromic DNA sequences by consulting your teacher. Better try to create a palindromic sequence by following base-pair rules.
◆A sequence of DNA that reads the same whether read from 5’ -> 3’ or from 3’ -> 5’direction is a palindromic sequence. ◆These are the sites for action of restriction enzymes. Almost all...
Explain with two examples each of the following: coordination entity, ligand, coordination number, coordination polyhedron, homoleptic and heteroleptic.
(a) Ligands are neutral chemicals or negative ions that are bonded to a metal atom in a coordination entity. Cl-, –OH as an exampleThey are electrically charged radicals or species. ( b )...
Discuss with your teacher and find out how to distinguish between
(a) Plasmid DNA and Chromosomal DNA (b) RNA and DNA (c) Exonuclease and Endonuclease A) Plasmid DNA ◆Plasmid DNA Chromosomal DNA It is an extra chromosomal DNA molecule found...
A small candle, 2.5 cm in size is placed at 27 cm in front of a concave mirror of radius of curvature 36 cm. At what distance from the mirror should a screen be placed in order to obtain a sharp image? Describe the nature and size of the image. If the candle is moved closer to the mirror, how would the screen have to be moved?
Answer – We are given that, Height of the candle is h =2.5 cm Let the image size be h’ Object distance is u = -27 cm Radius of the concave mirror, is R = -36 cm Focal length of the concave mirror...
Explain briefly
(a) PCR (b) Restriction enzymes and DNA (c) Chitinase Solution: 1.PCR ◆PCR or polymerase chain reaction is a technique to amplify a gene or a DNA fragment in...
FeSO4 solution mixed with (NH4)2SO4 solution in 1:1 molar ratio gives the test of Fe2+ ion but CuSO4 solution mixed with aqueous ammonia in 1:4 molar ratio does not give the test of Cu2+ ion. Explain why.
When FeSO4is combined with (NH4)2SO4 in a 1: 1 molar ratio, a double salt is formed. FeSO4 (NH4)2SO4 .6H2O. This salt is in charge of supplying Fe 2+ . CuSO 4 in a 1:4 ratio with aqueous ammonia...
Explain the bonding in coordination compounds in terms of Werner’s postulates.
( a ) There are two types of valencies in metals: primary and secondary valencies. Primary valencies are satisfied by negative ions, and secondary valencies are filled by both neutral and negative...
Can you think and answer how a reporter enzyme can be used to monitor transformation of host cells by foreign DNA in addition to a selectable marker?
DNA CONSTRUCT ●A collective DNA construct is introduced into the cell where the reporter gene is used as a selectable marker to discover the successful uptake of foreign genes. ● Example...
Can you recall meiosis and indicate at what stage a recombinant DNA is made?
◆Meiosis is a process involving a reduction in the quantity of genetic material . ◆ type of cell division- It occurs in two phases are - 1.meiosis I 2.meiosis II ◆ pachytene event...
Give examples and suggest reasons for the following features of the transition metal chemistry:
(i) The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
(ii) A transition metal exhibits highest oxidation state in oxides and fluorides.
(i) In the case of a lower oxide of a transition metal, the metal atom has a low oxidation state. This means that some of the valence electrons of the metal atom are not involved in bonding. As a...
Calculate the number of unpaired electrons in the following gaseous ions: Mn3+, Cr3+, V3+ and Ti3+. Which one of these is the most stable in aqueous solution?
Cr3+ is the most stable in aqueous solutions owing to a t2g3 configuration.
Which metal in the first series of transition metals exhibits +1 oxidation state most frequently and why?
In the first transition metal series, Cu exhibits +1 oxidation state very frequently because Cu has an electronic configuration of [Ar] 3d 10, that is, the completely filled d-orbital makes it...
What is meant by ‘disproportionation’? Give two examples of disproportionation reaction in aqueous solution.
It is found that sometimes a relatively less stable oxidation state undergoes an oxidation−reduction reaction in which it is simultaneously oxidised and reduced. This is called disproportionation....
The decomposition of 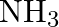 on platinum surface is zero order reaction. What are the rates of production of
on platinum surface is zero order reaction. What are the rates of production of 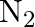 and
and 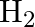 if
if 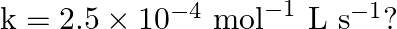
How would you account for the following? (i) The d1 configuration is very unstable in ions.
(i) The ions in d1 configuration tend to lose one more electron to get into stable d0 configuration. Also, the hydration or lattice energy is more than sufficient to remove the only electron present...
How would you account for the following?
(i) Of the d4 species, Cr2+ is strongly reducing while manganese(III) is strongly oxidising.
(ii) Cobalt(II) is stable in aqueous solution but in the presence of complexing reagents it is easily oxidised.
(i) Cr2+ is strongly reducing in nature. It has a d4 configuration. While acting as a reducing agent, it gets oxidized to Cr3+ (electronic configuration, d3). This...
![Rendered by QuickLaTeX.com \[\text { For the reaction: } 2 A+B \rightarrow A_{2} B \text { is } k[A][B]^{2} \text { with } k=2.0 \times 10^{-6} \mathrm{~mol}^{-2} L^{2} \mathrm{~s}^{-1} \text {. }\]](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-e9fea1165ffad66e28f91d69315a1de4_l3.png)
Calculate the initial rate of the reaction when ![Rendered by QuickLaTeX.com [\mathrm{A}]=0.1 \mathrm{~mol} \mathrm{~L}^{-1},[\mathrm{~B}]=0.2 \mathrm{~mol} \mathrm{~L}^{-1}](https://www.learnatnoon.com/s/wp-content/ql-cache/quicklatex.com-d19cf8e34beaaae48c1bfcd427f9aa96_l3.png) . Calculate the rate of reaction after [A] is reduced to
. Calculate the rate of reaction after [A] is reduced to 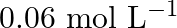
From the rate expression for the following reactions, determine their order of reaction and the dimensions of the rate constants.
(i) (ii) Solution: (i) (ii)
From the rate expression for the following reactions, determine their order of reaction and the dimensions of the rate constants.
Besides better aeration and mixing properties, what other advantages do stirred tank bioreactors have over shake flasks?
◆Stirred tank bioreactors are developed for a large-scale production of biotechnology products but the shake flask method is used for a small-scale production of biotechnological products carried...
Do eukaryotic cells have restriction endonucleases? Justify your answer.
● Eukaryotic cells do not have restriction endonucleases as the DNA of eukaryotes cells is highly methylated by methylase – a modification enzyme. ● Methylation safeguards the DNA from the...
What would be the molar concentration of human DNA in a human cell? Consult your teacher.
◆ Molar concentration of human being DNA in a human is : ◆6.023 x 1023 x Total number of chromosomes ◆6.023 x 1023 x 46 ◆2.77 x 1023 moles ◆ 2.77 x 1023 moles is the molar concentration of DNA...
From what you have learnt, can you tell whether enzymes are bigger or DNA is bigger in molecular size? How did you know?
●Compared to DNA molecules, enzymes are smaller in size. ● DNA comprises genetic material, which is essential for the normal development and functioning of living cells. ●A DNA molecule...
Write the structure of the major organic product in each of the following reactions:
Solution:
Write the structure of the major organic product in each of the following reactions:
Solution: (i) (ii)
1. Make a chart (with diagrammatic representation) showing a restriction enzyme, the substrate DNA on which it acts, the site at which it cuts DNA and the product it produces.
●Steps in the formation of recombinant DNA by action of restriction endonuclease enzyme written as – EcoRI ●It can be diagrammatically represent as -
What happens when (i) methyl bromide is treated with sodium in the presence of dry ether, (Ii) methylchloride is treated with KCN.
(i) Ethane is generated when methyl bromide is reacted with sodium in the presence of dry ether. The Wurtz reaction is the name for this response. (ii)
What happens when (i) chlorobenzene is subjected to hydrolysis, (ii) ethyl chloride is treated with aqueous KOH
(i) Under normal circumstances, chlorobenzene does not hydrolyze. When heated in an aqueous sodium hydroxide solution at a temperature of 623 K and a pressure of 300 atm, it hydrolyzes to produce...
Can you list 10 recombinant proteins which are used in medical practice? Find out where they are used as therapeutics (use the internet).
●Recombinant proteins used in medical practice which are obtained from the recombinant DNA technique . ●In this technology a particular gene is transferred from one organism to...
What happens when (i) n-butyl chloride is treated with alcoholic KOH, (ii) bromobenzene is treated with Mg in the presence of dry ether.
(i) The production of but-1-ene occurs when n-butyl chloride is treated with alcoholic KOH. A dehydrohalogenation process is what this is. (ii) Phenylmagnesium bromide is generated when bromobenzene...
Primary alkyl halide C4H9Br (a) reacted with alcoholic KOH to give compound (b). Compound (b) is reacted with HBr to give (c) which is an isomer of (a). When (a) is reacted with sodium metal it gives compound (d), C8H18 which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (a) and write the equations for all the reactions.
The formula C4H9Br is used to make two main alkyl halides. They're n-butyl bromide and isobutyl bromide, respectively. Compound (a) is therefore either nbutyl bromide or isobutyl bromide. Compound...
The treatment of alkyl chlorides with aqueous KOH leads to the formation of alcohols but in the presence of alcoholic KOH, alkenes are major products. Explain.
KOH almost entirely ionizes in an aqueous solution, yielding OH ions. Because the OH ion is a powerful nucleophile, it causes the alkyl chloride to undergo a substitution reaction, resulting in the...
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
The symmetrical nature of p-Dichlorobenzene is superior to that of the o- and m-isomers. As a result, it matches the crystal lattice better than the o- and m-isomers. As a result, breaking the...
Out of C6H5CH2Cl and C6H5CHClC6H5, which is more easily hydrolyzed by aqueous KOH?
The production of carbocation occurs during aqueous KOH hydrolysis. If the carbocation is stable, aqueous KOH can easily hydrolyze the compound. Now, C6H5 CH2Cl creates 1o– carbohydrate, whereas...
Arrange the compounds of each set in order of reactivity towards SN2 displacement: (i) 2-Bromo-2-methylbutane, 1-Bromopentane, 2-Bromopentane (ii) 1-Bromo-3-methylbutane, 2-Bromo-2-methylbutane, 3-Bromo-2- methylbutane (iii) 1-Bromobutane, 1-Bromo-2,2-dimethylpropane, 1-Bromo-2-methylbutane, 1Bromo-3- methylbutane.
(i) The nucleophile approaches the carbon atom to which the leaving group is linked in an SN2 reaction. The reactivity for SN2 displacement reduces when the nucleophile is sterically inhibited. The...
What will be the mechanism for the following reaction?
Solution: The given reaction is CN- functions as a nucleophile, attacking the carbon atom that the Br is bonded to. The CN– ion is a nucleophile that can attack at both the C and N sites. In this...
Write the structure of the major organic product in each of the following reactions:
Solution:
Write the structure of the major organic product in each of the following reactions:
Solution:
Give the uses of freon 12, DDT, carbon tetrachloride and iodoform.
Freon -12 has a variety of applications. Freon-12 (dichlorodifluoromethane, CF2Cl2) is also known as CFC. It's found in deodorants, hair sprays, and other aerosol spray propellants. It's also...
Explain why (i) the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride? (ii) alkyl halides, though polar, are immiscible with water? (iii) Grignard reagents should be prepared under anhydrous conditions?
(i) The Cl- atom in chlorobenzene is coupled to an sp2 hybridized carbon atom, whereas it is linked to an sp3 hybridized carbon atom in cyclohexyl chloride. The sp2 hybridized carbon atom is now...
How will you bring about the following conversions? (i) 1-Chlorobutane to n-octane (ii) Benzene to biphenyl.
(i) (ii)
How will you bring about the following conversions? (i) Bromomethane to propanone (ii) But-1-ene to but-2-ene
(i) (ii)
How will you bring about the following conversions? (i) Propene to propyne (ii) Ethanol to ethyl fluoride
(i) (ii)
How will you bring about the following conversions? (i) Propene to 1-nitropropane (ii) Toluene to benzyl alcohol
(i) (ii)
How will you bring about the following conversions? (i) Ethanol to but-1-yne (ii) Ethane to bromoethene
(i) (ii)
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene: (i) 1-Bromo-1-methylcyclohexane (ii) 2-Chloro-2-methyl butane (iii) 2,2,3-Trimethyl-3-bromopentane.
(i) 1−bromo−1−methylcyclohexane In the given molecule, all β-hydrogens are equivalent. As a result, only one alkene is produced when the given molecule is dehydrogenated. (ii) Different sets of...
Compare the chemistry of actinoids with that of the lanthanoids with special reference to: (i) oxidation state (ii) chemical reactivity.
(i) Oxidation states : The major oxidation state of lanthanoids is (+3). However, sometimes we also find oxidation states of +2 and +4. This occurs because of the extra stability of fully-filled and...
Which compound in each of the following pairs will react faster in SN2 reaction with –OH? (i) CH3Br or CH3I (ii) (CH3)3CCl or CH3Cl
(i) The order in which the halides react to an alkyl group is constant in the SN2 process. This is because as the size of the halide ion rises, it becomes a better leaving group. R-F << R-Cl...
Compare the chemistry of actinoids with that of the lanthanoids with special reference to:
(i) electronic configuration
(ii) atomic and ionic sizes
(i) Electronic configuration: The general electronic configuration for lanthanoids is [Xe]54 4f0-14 5d0-1 6s2 and that for actinoids is [Rn]86 5f1-14 6d0-1 7s2....
Compare the stability of +2 oxidation state for the elements of the first transition series.
From the table shown above we can deduce the following points: Mn shows maximum number of oxidation states, that ranges between +2 to +7.The number of oxidation states increases as we move on from...
What are ambident nucleophiles? Explain with an example.
The term ambident nucleophile refers to a nucleophile with two nucleophilic sites. These nucleophilic sites are targets for them to attack. For example, the nitrite ion. When the nitrite ion can...
Predict which of the following will be coloured in aqueous solution? Ti3+, V3+, Cu+, Sc3+, Mn2+, Fe3+ and Co2+. Give reasons for each.
Ions that have electrons in d-orbital will be the only ones that will impart colour and the ions with vacant d-orbital will remain colourless. All other ions, except Sc3+, will be coloured in...
Write the equations for the preparation of 1−iodobutane from: (i) 1-butanol (ii) 1-chlorobutane (iii) but-1-ene
(i) (ii) (iii)
For M2+/M and M3+/M2+ systems the EV values for some metals are as follows:
Cr2+/Cr = -0.9V Cr3/Cr2+ = -0.4 V
Mn2+/Mn = -1.2V Mn3+/Mn2+ = +1.5 V Fe2+/Fe = -0.4V Fe3+/Fe2+ = +0.8 V Use this data to comment upon: (i) the stability of Fe3+ in acid solution as compared to that of Cr3+ or Mn3+ and (ii) the ease with which iron can be oxidised as compared to a similar process for either chromium or manganese metal.
(i) The reduction potentials for the given pairs increase in the given order: Mn2+ / Mn < Cr2+ / Cr < Fe2+ /Fe So, the oxidation of Fe to Fe2+ is not as easy as the...
Write the isomers of the compound having formula C4H9Br.
(i) 1−Bromobutane (ii) 2−Bromobutane (iii) 1−Bromo−2−methylpropane (iv) 2−Bromo−2−methylpropane
A hydrocarbon C5H10does not react with chlorine in dark but gives a single monochloro compound C5H9Cl in bright sunlight. Identify the hydrocarbon.
C5H10 is a hydrocarbon with the chemical formula CnH2n, which belongs to the CnH2n group of hydrocarbons. As a result, it could be an alkene or a cycloalkane. Hydrocarbon cannot be an alkene because...
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with (i) iron(II) ions (ii) SO2 and (iii) oxalic acid?
Write the ionic equations for the reactions. The preparation of potassium permanganate can be done from pyrolusite (MnO2). The ore is fused with KOH in the presence of either atmospheric oxygen or...
Give the IUPAC names of the following compounds:
(i) CH3 C (p – Cl C6 H4 )2 CH(Br) CH3 (ii) (CH3 )3 C CH = C Cl C6 H4 I – p (i) 2−Bromo−3, 3−bis(4 − chlorophenyl) butane (ii) 1−chloro−1−(4−iodophenyl)−3,...
Describe the oxidising action of potassium dichromate and write the ionic equations for its reaction with:
(i) iodide (ii) iron(II) solution and (iii) H2S
In acidic medium, K2Cr2O7 behaves as a very strong oxidising agent. \[{K_2}C{r_2}{O_7} + 4{H_{2}}S{O_4} \to {K_2}S{O_{4}} + C{r_{2}}{(S{O_4})_{3}} + 4{H_2}O + 3[O]\;\] K2Cr2O7 gains electrons...
Describe the preparation of potassium dichromate from iron chromite ore. What is the effect of increasing pH on a solution of potassium dichromate?
Potassium dichromate is prepared from iron chromite ore through the following steps: Step 1: Preparation of sodium chromate: \[FeC{r_2}{O_4} + 16NaOH + 7{O_2} \to 8N{a_2}Cr{O_4} + 2F{e_2}{O_3} +...
How is the variability in oxidation states of transition metals different from that of the non transition metals? Illustrate with examples.
In transition elements, the oxidation state can vary from +1 to the highest oxidation state by the removal of all their valence electrons. Along with that. in transition elements, the oxidation...
What are interstitial compounds? Why are such compounds well known for transition metals?
Transition metals are large in size and they also contain many interstitial sites. These interstitial sites can be used to trap atoms of other elements (that have small atomic size), such as H, C, N...
Explain giving reasons:
(i) The transition metals generally form coloured compounds.(ii) Transition metals and their many compounds act as good catalyst. (i) Most of the complexes of transition metals are coloured as...
Explain giving reasons: (i) Transition metals and many of their compounds show paramagnetic behaviour.(ii) The enthalpies of atomisation of the transition metals are high.
i) Transition metals show paramagnetic behaviour. Paramagnetism is a phenomenon that arises due to the presence of unpaired electrons with each electron having a magnetic moment associated with...
What are the different oxidation states exhibited by the lanthanoids?
The most common oxidation state in the case of lanthanoids is +3 oxidation state. Thus, Ln(III) compounds are predominant. However, +2 and +4 oxidation states can also be found in the solution or in...
In what way is the electronic configuration of the transition elements different from that of the non transition elements?
Transition metals have a partially filled or incomplete d−orbital. Hence, the electronic configuration of transition elements is (n − 1)d1-10 ns0-2. The non-transition elements either do not...
What are the characteristics of the transition elements and why are they called transition elements? Which of the d-block elements may not be regarded as the transition elements?
Transition elements are those elements in which the atoms or ions (in stable oxidation state) contain partially filled or incomplete d-orbital. These elements are found in the d-block (as the...
In deriving the single slit diffraction pattern, it was stated that the intensity is zero at angles of nλ/a. Justify this by suitably dividing the slit to bring out the cancellation.
Answer - Let “a” denote the width of a single slit. According to the question, the single slit is then further divided into n smaller slits of width a’. So, we can write – a’ = a / n For the...
Answer the following questions:
(a) When a low flying aircraft passes overhead, we sometimes notice a slight shaking of the picture on our TV screen. Suggest a possible explanation.
(b) As you have learnt in the text, the principle of linear superposition of wave displacement is basic to understanding intensity distributions in diffraction and interference patterns. What is the justification of this principle?
Answer – (a) The aircraft's weak radar emissions interfere with the antenna's reception of the TV signal. (b) This is because the linear aspect of a differential equation that drives wave motion...
A parallel beam of light of wavelength 500 nm falls on a narrow slit and the resulting diffraction pattern is observed on a screen 1 m away. It is observed that the first minimum is at a distance of 2.5 mm from the centre of the screen. Find the width of the slit.
Answer – We are given that, λ = 500 nm is the wavelength of the beam of light D= 1 m is the distance between the slit and the screen x = 2.5 mm = 2.5 x 10-3 m is the distance of the first...
Two towers on top of two hills are 40 km apart. The line joining them passes 50 m above a hill halfway between the towers. What is the longest wavelength of radio waves, which can be sent between the towers without appreciable diffraction effects?
Answer – We are given that, 40 km Is the distance between the towers d = 50 m is the height of the line joining the hills The radial spread of the radio waves can not exceed 50 m Aperture,...
Answer the following questions:
(a) In a single slit diffraction experiment, the width of the slit is made double the original width. How does this affect the size and intensity of the central diffraction band?
(b) In what way is diffraction from each slit related to the interference pattern in a double-slit experiment?
(c) When a tiny circular obstacle is placed in the path of light from a distant source, a bright spot is seen at the centre of the shadow of the obstacle. Explain why?
(d) Two students are separated by a 7 m partition wall in a room 10 m high. If both light and sound waves can bend around obstacles, how is it that the students are unable to see each other even though they can converse easily?
(e) Ray optics is based on the assumption that light travels in a straight line. Diffraction effects (observed when light propagates through small apertures/slits or around small obstacles) disprove this assumption. Yet the ray optics assumption is so commonly used in an understanding location and several other properties of images in optical instruments. What is the justification?
Answer – (a) In a single slit diffraction experiment, doubling the slit width reduces the size of the central diffraction band by half while increasing the intensity of the band by four times. (b)...
In a double-slit experiment using the light of wavelength 600 nm, the angular width of a fringe formed on a distant screen is 0.1°. What is the spacing between the two slits?
Answer – We are given that - Wavelength of the light is λ = 600 nm Θ is the angular width of the fringe formed, given equal to 0.10 = 0.1 π/180 Spacing between the slits is d...
Question 12.1: What is meant by the following terms? Give an example of the reaction in each case. (v) Hemiacetal (vi) Oxime
Solution: (v) Hemiacetals are a type of α−alkoxyalcohols. A hemiacetal's overall structure. Aldehyde interacts with one molecule of monohydrated alcohol in the presence of dry HCl gas. (vi) Oxime:...
For sound waves, the Doppler formula for frequency shift differs slightly between the two situations: (i) source at rest; observer moving, and (ii) source moving; observer at rest. The exact Doppler formulas for the case of light waves in vacuum are, however, strictly identical for these situations. Explain why this should be so. Would
you expect the formulas to be strictly identical for the two situations in the case of light travelling in a medium?
Answer – Because sound waves can only move through the medium, the Doppler formula differs somewhat between the two scenarios. In both circumstances, the observer's motion relative to the medium is...
Let us list some of the factors, which could possibly influence the
speed of wave propagation:
(i) nature of the source.
(ii) the direction of propagation.
(iii) the motion of the source and/or observer.
(iv) wavelength.
(v) the intensity of the wave. On which of these factors, if any, does (a) the speed of light in a vacuum,
(b) the speed of light in a medium (say, glass or water), depend?
Answer – (a) In the vacuum, none of the components listed affect the speed of light. (b) The wavelength of light in a media affects the speed of light in that medium.
You have learnt in the text how Huygens’ principle leads to the laws of reflection and refraction. Use the same principle to deduce directly that a point object placed in front of a plane mirror produces a virtual image whose distance from the mirror is equal to the object distance from the mirror.
Answer – Consider an item O placed at a distance r in front of the plane mirror MO'. The object is designated as point O, and a circle is drawn around it, just touching the plane mirror at point O'....
Explain how Corpuscular theory predicts the speed of light in a medium, say, water, to be greater than the speed of light in vacuum. Is the prediction confirmed by experimental determination of the speed of light in water? If not, which alternative picture of light is consistent with experiment?
Answer – The velocity of light in the denser medium (water) is greater than the velocity of light in the rarer medium (air), according to Newton's Corpuscular hypothesis (vacuum). This was erroneous...
The 6563 Å Hα line emitted by hydrogen in a star is found to be red-shifted by 15 Å. Estimate the speed with which the star is receding from the Earth.
Answer – It is given in the question – λ = 6563 Å and, Δλ = 15 Å Since the star is receding, the velocity (v) will be negative. We know that, Δλ = – vλ/c Therefore, v = – cΔλ/λ = – (3 x...
Estimate the distance for which ray optics is a good approximation for an aperture of 4 mm and wavelength 400 nm.
Answer: The distance which is used in ray optics for a good approximation is known as Fresnel’s distance ZF, relation for which is given as follows – \[{{Z}_{F}}=\frac{{{a}^{2}}}{\lambda }\] Where...
Light of wavelength 5000 Armstrong falls on a plane reflecting surface. What are the wavelength and frequency of the reflected light? For what angle of incidence is the reflected ray normal to the incident ray?
Answer – We are given, Wavelength of incident light is λ = 5000 Armstrong = 5000 x 10-10 m Speed of light is known as c =3 x 108 m The relation for the frequency of...
What is the Brewster angle for air to glass transition? (Refractive index of glass=1.5.)
Answer – We are given that Refractive index of glass is μ = 1.5 Let the Brewster angle be θ The relation between the Brewster angle and the refractive index is given as follows – \[\tan \theta =\mu...
In a double-slit experiment, 0.2° is found to be the angular width of a fringe on a screen placed 1 m away. The wavelength of light used is 600 nm. What will be the angular width of the fringe if the entire experimental apparatus is immersed in water? Take refractive index of water to be ¾.
Answer: We are given, Distance of the screen from the slits is D = 1m Wavelength of light used is λ1 = 600 nm Angular width of the fringe in air is θ1 = 0.2° θ2 is the angular...
In Young’s double-slit experiment using the monochromatic light of wavelength λ, the intensity of light at a point on the screen where path difference is λ, is K units. What is the intensity of light at a point where path difference is λ/3?
Answer – Let I1 and I2 represent the intensity of the two light waves. Then, their resultant intensities can be determined as follows –...
In Young’s double-slit experiment, 0.28mm separation between the slits and the screen is placed 1.4m away. 1.2cm is the distance between the central bright fringe and the fourth bright fringe. Determine the wavelength of light used in the experiment.
Answer – We are given, Distance between the screen and the slits, D = 1.4 m Distance between the slits is d = 0.28 mm = 0.28 x 10-3 m Also, the distance between the central fringe and the...
(i) The refractive index of glass is 1.5. What is the speed of light in glass? Speed of light in a vacuum is ( 3.0 x 108 m s-1 )
(ii) Is the speed of light in glass Independent of the colour of light? If not, which of the two colours red and violet travels slower in a glass prism? Answer – (i) We are given, Refractive...
What is the shape of the wavefront in each of the following cases:
(i) Light diverging from a point source. (ii) Light emerging out of a convex lens when a point source is placed at its focus. (iii) The portion of the wavefront of the light from a distant star...
Monochromatic light having a wavelength of 589nm from the air is incident on a water surface. Find the frequency, wavelength and speed of (i) reflected and (ii) refracted light? [1.33 is the Refractive index of water]
Answer – We are given, Wavelength of monochromatic light incident is , λ = 589 nm = 589 x 10-9 m Speed of light in air is c = 3 x 108 m s-1 Refractive index of water is...
